Scalable Synthesis of Asymmetric Ring-Opening Cucurbituril for Advanced Drug Delivery Systems
Scalable Synthesis of Asymmetric Ring-Opening Cucurbituril for Advanced Drug Delivery Systems
The landscape of supramolecular chemistry is undergoing a significant transformation with the introduction of asymmetric ring-opening cucurbiturils, as detailed in patent CN110724273A. This groundbreaking technology addresses critical limitations found in traditional macrocyclic hosts by introducing a versatile C-shaped architecture capable of encapsulating a broader range of guest molecules. Unlike their symmetric predecessors, these novel compounds incorporate specific molecular building blocks, including polymerized glycoluril and asymmetric hydroquinone or naphthalenediol derivatives, which impart unique curvature and binding properties. The presence of sulfonic acid groups on the side chains further revolutionizes their utility by providing exceptional water solubility, a prerequisite for many pharmaceutical and biological applications. This patent outlines a robust preparation method that leverages acid-catalyzed condensation to achieve high-purity products suitable for industrial deployment.
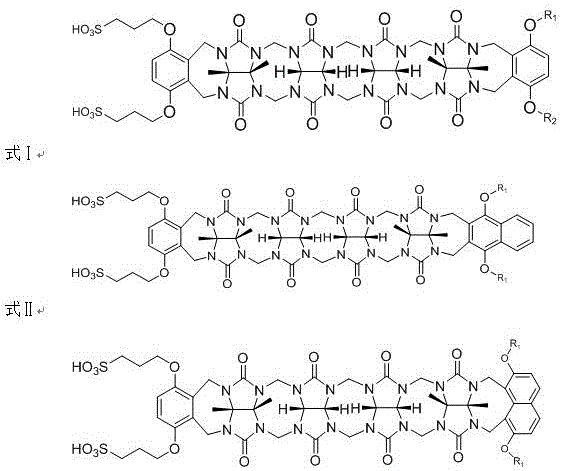
For R&D directors and procurement specialists in the fine chemical sector, this innovation represents a pivotal shift towards more adaptable supramolecular carriers. The ability to tune the cavity size and surface chemistry allows for precise host-guest recognition, which is essential for developing advanced drug delivery systems, solubilizing insoluble active pharmaceutical ingredients, and creating stable multi-molecular complexes. The synthetic route described avoids the complexities often associated with macrocycle functionalization, offering a direct path from readily available starting materials to high-value intermediates. As the demand for specialized excipients and functional materials grows in the pharmaceutical and agrochemical industries, mastering the production of these asymmetric hosts becomes a strategic imperative for maintaining competitive advantage in material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing cucurbituril derivatives have long been hindered by the inherent rigidity and symmetry of the macrocyclic framework. Conventional symmetric ring-opening cucurbiturils often suffer from limited adaptability, as their fixed cavity dimensions restrict the range of guest molecules they can effectively encapsulate. Furthermore, many existing synthesis protocols rely on harsh reaction conditions or complex multi-step functionalization processes that result in low overall yields and significant impurity profiles. The lack of solubilizing groups in standard architectures frequently necessitates additional derivatization steps to make the molecules compatible with aqueous environments, adding time and cost to the manufacturing process. These factors collectively create a bottleneck for the commercialization of supramolecular technologies in sectors requiring high purity and scalability, such as parenteral drug formulations.
The Novel Approach
The methodology presented in patent CN110724273A overcomes these historical barriers through a streamlined one-pot condensation strategy that directly integrates solubilizing and structural motifs. By reacting tetrameric glycoluril with 3,3'-(1,4-phenylenedioxy)-bis(1-propanesulfonic acid) and specific aromatic derivatives, the process inherently builds the asymmetric C-shape and the sulfonic acid functionality simultaneously. This convergent synthesis eliminates the need for post-synthetic modification to introduce solubility, drastically simplifying the workflow. The use of organic strong acids like methanesulfonic acid or trifluoroacetic acid as both solvent and catalyst creates a highly efficient reaction environment that promotes rapid bond formation at moderate temperatures. This approach not only enhances the structural diversity of the final product but also ensures that the resulting supramolecular carriers possess the necessary physicochemical properties for immediate application in complex biological matrices.
Mechanistic Insights into Acid-Catalyzed Condensation Polymerization
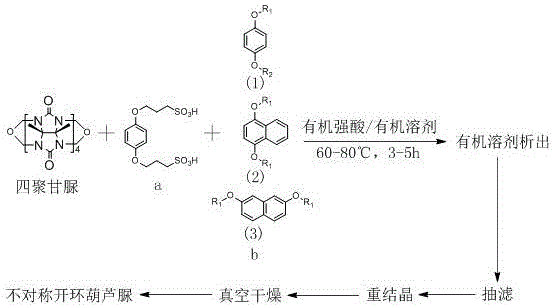
The core of this synthesis lies in the acid-catalyzed condensation mechanism, where the electrophilic activation of the glycoluril units facilitates nucleophilic attack by the hydroxyl groups of the aromatic derivatives. In the presence of strong organic acids, the carbonyl or imine functionalities within the glycoluril oligomers become highly reactive, driving the formation of methylene bridges that link the molecular building blocks into the characteristic semi-cyclic array. The stoichiometry plays a critical role here, with molar ratios of tetrameric glycoluril to the sulfonic acid linker and aromatic cap ranging from 5:6:6 to 5:8:8. This precise control over reactant ratios ensures that the polymerization terminates at the desired oligomeric state, preventing the formation of uncontrolled polymeric byproducts that would complicate purification. The reaction kinetics are optimized by maintaining temperatures between 60°C and 80°C, providing sufficient thermal energy to overcome activation barriers without degrading the sensitive functional groups.
Impurity control is intrinsically managed through the solubility characteristics of the intermediates and the final product. The sulfonic acid groups introduced via the 3,3'-(1,4-phenylenedioxy)-bis(1-propanesulfonic acid) linker render the growing oligomer soluble in the acidic reaction medium but allow for selective precipitation upon dilution with non-polar or less polar organic solvents. This phase behavior acts as a self-purification mechanism; as the reaction proceeds, the desired asymmetric product remains in solution while potential symmetric byproducts or unreacted starting materials may exhibit different solubility profiles. Subsequent workup steps involving dissolution in water followed by recrystallization with organic solvents like ethanol or acetone further refine the purity. This rigorous control over the crystallization process ensures that the final supramolecular carrier meets the stringent quality standards required for pharmaceutical intermediates, minimizing the risk of toxic impurities in the final drug product.
How to Synthesize Asymmetric Ring-Opening Cucurbituril Efficiently
The practical execution of this synthesis is designed for seamless translation from laboratory bench to pilot plant operations. The process begins with the precise weighing and mixing of the three key components: tetrameric glycoluril, the sulfonic acid linker, and the chosen aromatic derivative (hydroquinone or naphthalenediol based). These solids are suspended in a mixed solvent system comprising an organic strong acid and a co-solvent such as acetone or tetrahydrofuran, with volume ratios adjusted between 1:1 and 3:1 to optimize solubility and reaction rate. The mixture is then heated to the target temperature range of 60°C to 80°C and stirred continuously for a duration of 3 to 5 hours. Monitoring the reaction progress is straightforward, and upon completion, the mixture is cooled to room temperature to initiate the isolation phase.
- Combine tetrameric glycoluril, 3,3'-(1,4-phenylenedioxy)-bis(1-propanesulfonic acid), and hydroquinone or naphthalenediol derivatives in a mixture of organic strong acid and organic solvent.
- Heat the reaction mixture to 60-80°C and maintain stirring for 3-5 hours to facilitate the condensation polymerization.
- Cool the reaction, precipitate the product in an organic solvent, filter, dissolve in water, and recrystallize to obtain high-purity asymmetric cucurbituril.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this technology offers compelling advantages driven by its reliance on commodity chemicals and simplified processing. The elimination of transition metal catalysts, which are often expensive and subject to volatile market pricing, significantly reduces the raw material cost base. Furthermore, the absence of heavy metals removes the regulatory burden and operational cost associated with metal scavenging and residual metal testing, which are critical checkpoints in pharmaceutical manufacturing. The use of common organic solvents like acetone, ethanol, and methanol ensures that sourcing is reliable and不受 geopolitical supply shocks that often affect specialized reagents. This robustness in the supply chain translates to greater continuity of supply for downstream customers who depend on consistent availability of high-quality intermediates for their own production lines.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing a direct condensation route that avoids multi-step protection and deprotection sequences common in traditional macrocycle synthesis. The use of organic acids as catalysts is not only cheaper than noble metal catalysts but also easier to handle and dispose of, reducing waste management overheads. Additionally, the high efficiency of the reaction means that less raw material is wasted in the form of byproducts, improving the overall atom economy of the process. These factors combine to lower the cost of goods sold (COGS), allowing for more competitive pricing in the market for specialty supramolecular materials without compromising on margin.
- Enhanced Supply Chain Reliability: The starting materials, including glycoluril and various hydroquinone derivatives, are widely produced industrial chemicals with established global supply networks. This abundance mitigates the risk of shortages that can plague niche synthetic intermediates. The mild reaction conditions (60-80°C) also mean that the process can be run in standard glass-lined or stainless steel reactors without requiring exotic high-pressure or cryogenic equipment. This compatibility with existing infrastructure allows contract manufacturing organizations (CMOs) to adopt the technology quickly, expanding the available manufacturing capacity and ensuring that lead times remain short even during periods of high demand.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, as demonstrated by the straightforward workup procedure involving precipitation and filtration, which are unit operations easily adapted to large-scale production. The solvent systems used are largely recyclable, supporting green chemistry initiatives and reducing the environmental footprint of the manufacturing process. By avoiding chlorinated solvents and heavy metals, the process aligns with increasingly strict environmental regulations in major markets like Europe and North America. This compliance reduces the risk of regulatory delays and ensures long-term viability of the production route, making it a sustainable choice for companies aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these asymmetric supramolecular carriers. The answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing product pipelines or research programs.
Q: What are the key structural advantages of asymmetric ring-opening cucurbiturils over symmetric versions?
A: Asymmetric variants feature a flexible C-shaped cavity that can adapt to guest molecules of varying sizes, unlike rigid symmetric counterparts. Additionally, the incorporation of sulfonic acid side chains significantly enhances water solubility, making them superior for biological and pharmaceutical applications.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the process utilizes organic strong acids such as methanesulfonic acid or trifluoroacetic acid as catalysts. This eliminates the need for costly transition metals and the complex purification steps required to remove metal residues, thereby reducing overall production costs.
Q: What is the typical reaction temperature and time for this process?
A: The reaction operates under mild thermal conditions, typically between 60°C and 80°C, with a reaction duration of 3 to 5 hours. These moderate conditions contribute to energy efficiency and operational safety during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Ring-Opening Cucurbituril Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of asymmetric ring-opening cucurbiturils in next-generation drug delivery and material science applications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of residual solvents and impurity profiles, to guarantee that every batch meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems are designed to deliver that reliability batch after batch.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can reduce your overall procurement spend. Please contact our technical procurement team today to request specific COA data for our current inventory or to discuss route feasibility assessments for custom derivatives. Let us help you secure a stable, high-quality supply of these critical supramolecular intermediates for your future projects.
