Advanced Le-Phos Catalyst Technology for Scalable Pharmaceutical Intermediate Production
Introduction to Le-Phos Catalyst Technology
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. Patent CN109503660B introduces a groundbreaking class of chiral monophosphine catalysts known as Le-Phos, characterized by a unique cyclic phosphine skeleton. This innovation addresses critical bottlenecks in organocatalysis by providing a versatile platform capable of accessing all four stereoisomeric configurations with high optical purity. The core structure, designated as Compound 1, features a sophisticated arrangement of chiral centers including both carbon and phosphorus chirality, which is essential for inducing high stereoselectivity in downstream transformations.
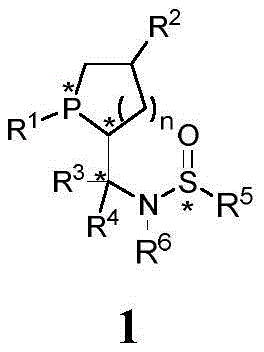
For R&D directors and process chemists, the significance of this technology lies in its ability to facilitate metal-free asymmetric reactions, specifically gamma-addition reactions involving nitrogen nucleophiles. Traditional methods often rely on expensive transition metals which pose contamination risks and environmental hazards. Le-Phos circumvents these issues by acting as a potent organocatalyst that forms zwitterionic intermediates, driving reactions with exceptional efficiency. The patent details a comprehensive preparation method that overcomes the limitations of previous generations of phosphine ligands, such as Ming-Phos or Xiao-Phos, by offering a more direct and cost-effective synthetic route. This makes Le-Phos an attractive candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their catalytic portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral phosphine ligands with P-centered chirality has been fraught with significant challenges that hinder their widespread industrial adoption. Conventional routes often involve multi-step sequences that utilize prohibitively expensive starting materials, leading to inflated production costs that are unsustainable for large-scale manufacturing. Furthermore, many existing protocols require harsh reaction conditions and toxic reagents, creating substantial safety and environmental compliance burdens for chemical plants. A major technical hurdle has been the difficulty in synthesizing specific enantiomers; separating diastereomers or resolving racemates often results in substantial material loss, capping overall yields at inefficient levels. These factors collectively contribute to extended lead times and supply chain vulnerabilities for high-purity chiral catalysts.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by streamlining the construction of the phosphine skeleton through a convergent strategy. By utilizing readily available ketones or aldehydes and tert-butylsulfinamide, the process establishes chirality early in the sequence via a condensation reaction. This is followed by a highly controlled lithiation and addition sequence that installs the phosphine moiety with precise stereochemical outcome. The novelty lies in the ability to access all four full configurations—(RP,S,S,RS), (SP,R,S,RS), (SP,R,R,SS), and (RP,S,R,SS)—from common precursors simply by varying the configuration of the starting materials. This flexibility eliminates the need for difficult resolution steps, thereby drastically simplifying the workflow. The resulting catalysts exhibit high reaction activity and stereoselectivity, making them powerful tools for cost reduction in API manufacturing where purity and efficiency are paramount.
Mechanistic Insights into Asymmetric Gamma-Addition Catalysis
Understanding the mechanistic underpinnings of Le-Phos is crucial for leveraging its full potential in complex molecule synthesis. The catalyst operates through a distinct organocatalytic cycle that avoids the use of transition metals entirely. Initially, the nucleophilic phosphorus atom of the Le-Phos catalyst attacks the electron-deficient central carbon of an allenoate substrate. This interaction generates a reactive quaternary phosphonium salt zwitterionic intermediate, which serves as the key activated species in the catalytic cycle. The rigidity and specific spatial arrangement of the cyclic phosphine skeleton in Le-Phos create a well-defined chiral environment around this intermediate.
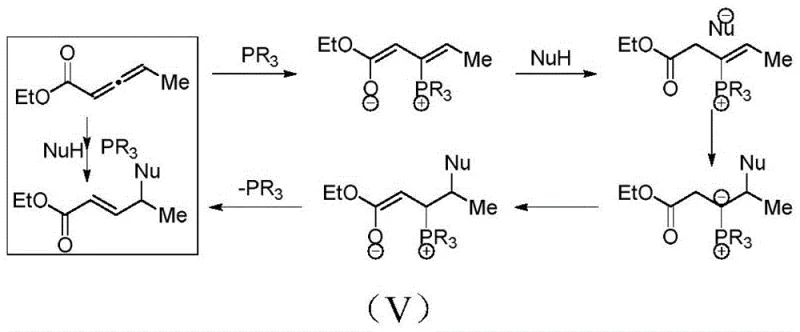
Subsequently, a nitrogen nucleophile, such as a cyclic imide or amine derivative, performs a nucleophilic attack on the activated allenoate system. The chiral pocket formed by the Le-Phos framework dictates the facial selectivity of this attack, ensuring that the new carbon-nitrogen bond is formed with high enantiomeric excess. Following the bond formation, the catalyst is eliminated from the adduct, regenerating the free phosphine species to enter another catalytic turnover. This mechanism not only ensures high atom economy but also minimizes the formation of unwanted byproducts. The ability to tune the steric and electronic properties of the R-groups on the phosphine ring allows chemists to fine-tune the catalyst for specific substrates, enhancing impurity control mechanisms and ensuring that the final gamma-amino acid derivatives meet stringent quality specifications required for pharmaceutical applications.
How to Synthesize Le-Phos Efficiently
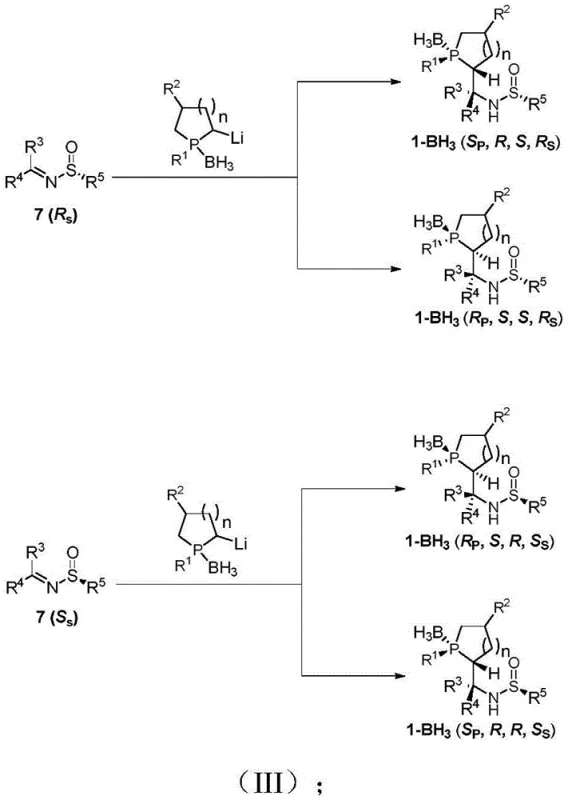
The preparation of Le-Phos is designed to be robust and adaptable, utilizing standard laboratory and plant equipment. The synthesis begins with the condensation of a carbonyl compound with a chiral sulfinamide to form a sulfinylimine, followed by the generation of a lithiated phosphine species from a cyclic phosphine-borane complex. These two key fragments are then coupled via a nucleophilic addition reaction to build the core scaffold. Finally, a deprotection step removes the borane and sulfinyl groups to reveal the active catalyst. This modular approach allows for the systematic variation of substituents to generate a library of catalysts tailored for specific reactions.
- Condense ketone/aldehyde compounds with tert-butylsulfinamide using a titanium ester condensing agent to form chiral sulfinylimines.
- Perform lithiation of cyclic phosphine-borane complexes using TMEDA-BuLi at low temperatures to generate reactive organolithium intermediates.
- Execute nucleophilic addition of the lithiated phosphine to the sulfinylimine to construct the carbon-phosphorus backbone with controlled stereochemistry.
- Remove the borane protecting group and sulfinyl auxiliary via deprotection reagents like diethylamine to yield the final free Le-Phos catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of Le-Phos technology offers tangible strategic benefits that extend beyond mere technical performance. The streamlined synthesis route directly translates to reduced manufacturing overheads. By eliminating the need for precious metal catalysts and complex resolution processes, the overall cost of goods sold (COGS) for the final chiral intermediates can be significantly lowered. This cost efficiency is critical for maintaining competitiveness in the global market for fine chemicals and active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The synthetic pathway for Le-Phos avoids the use of expensive transition metals and specialized chiral auxiliaries that are difficult to recover. Instead, it relies on commodity chemicals like benzaldehydes and amines, which are readily available from multiple global suppliers. The elimination of heavy metal removal steps, which often require specialized scavengers and extensive filtration, simplifies the downstream processing. This reduction in unit operations leads to substantial cost savings in terms of both material consumption and labor hours, making the process economically viable for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic catalysts poses a significant risk to production continuity. The Le-Phos synthesis utilizes widely accessible raw materials and standard solvents such as tetrahydrofuran and diethyl ether. This universality means that supply disruptions for any single component can be easily mitigated by sourcing from alternative vendors. Furthermore, the robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-high vacuum or inert gas manifolds beyond standard nitrogen protection, ensures that production can be maintained consistently across different manufacturing sites, reducing lead time for high-purity chiral catalysts.
- Scalability and Environmental Compliance: Modern chemical manufacturing is increasingly constrained by environmental regulations regarding waste disposal and solvent usage. The Le-Phos process generates fewer hazardous byproducts compared to traditional metal-catalyzed routes. The solvents used are common and can be efficiently recycled through distillation. Additionally, the high selectivity of the catalyst minimizes the formation of isomeric impurities, reducing the burden on purification systems and lowering the volume of waste solvent generated per kilogram of product. This alignment with green chemistry principles facilitates easier regulatory approval and supports sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of Le-Phos catalysts in industrial processes. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners.
Q: What are the primary advantages of Le-Phos over traditional phosphine ligands?
A: Le-Phos offers a unique cyclic skeleton with four chiral centers, allowing for the efficient synthesis of all four stereoisomers. Unlike prior art which often suffers from expensive raw materials and lengthy routes, Le-Phos utilizes a streamlined synthesis with yields ranging from 42% to 75%, significantly reducing production complexity and cost.
Q: Can Le-Phos be used for metal-free asymmetric catalysis?
A: Yes, Le-Phos is specifically designed for metal-free organocatalysis. It effectively catalyzes asymmetric gamma-addition reactions of nitrogen nucleophiles to allenoates, achieving high enantioselectivity (up to 95% ee) without the need for transition metals, aligning with green chemistry principles.
Q: Is the synthesis of Le-Phos scalable for industrial production?
A: The patent describes a robust four-step synthesis using common solvents like THF and ether under manageable temperature conditions (-78°C to 80°C). The avoidance of exotic reagents and the use of standard purification techniques like column chromatography indicate strong potential for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Le-Phos Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysts like Le-Phos in accelerating drug discovery and process development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive chemistry required for phosphine synthesis, ensuring stringent purity specifications are met for every batch. With rigorous QC labs and a commitment to quality, we guarantee that our Le-Phos catalysts deliver the high enantioselectivity and reproducibility necessary for your most challenging asymmetric syntheses.
We invite you to collaborate with us to unlock the full value of this technology for your pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with high-performance chiral solutions.
