Advanced Synthesis of Cyclopentenyl Lactone: A Scalable Route for Prostaglandin Manufacturing
Introduction to Advanced Lactone Synthesis Technology
The pharmaceutical industry constantly seeks robust pathways for constructing complex molecular scaffolds, particularly for vital classes like prostaglandins. Patent CN1732149A introduces a refined methodology for synthesizing 3,3a,6,6a-tetrahydro-2H-cyclopenta[b]furan-2-one, a pivotal intermediate in prostaglandin production. This specific lactone structure serves as a cornerstone for various bioactive molecules, necessitating a synthesis route that balances stereochemical integrity with operational simplicity. The disclosed process leverages a modified Claisen rearrangement utilizing amide acetals or aminoketene acetals, marking a significant departure from traditional high-energy protocols. By optimizing reaction conditions and reagent selection, this technology addresses long-standing challenges in scalability and purity that have historically plagued the manufacturing of such sensitive cyclic compounds.
![Chemical structure of the target lactone 3,3a,6,6a-tetrahydro-2H-cyclopenta[b]furan-2-one (Formula IV)](/insights/img/cyclopentenyl-lactone-synthesis-prostaglandin-intermediate-supplier-20260307235124-08.png)
For global procurement teams and R&D directors, the implications of this patent extend beyond mere academic interest; it represents a viable strategy for securing a reliable prostaglandin intermediate supplier. The ability to produce high-purity cyclopentenyl lactone with controlled stereochemistry is paramount for downstream drug efficacy. Furthermore, the process design inherently supports cost reduction in pharmaceutical intermediate manufacturing by simplifying purification steps and reducing energy inputs. As we delve deeper into the technical specifics, it becomes evident that this approach offers a compelling alternative for the commercial scale-up of complex pharmaceutical intermediates, ensuring a steady supply chain for critical therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 3,3a,6,6a-tetrahydro-2H-cyclopenta[b]furan-2-one has been fraught with significant technical hurdles that impede efficient large-scale production. One established route involves the reaction of dichloroketene with cyclopentadiene, followed by zinc dechlorination and Bayer-Villiger oxidation. While chemically feasible, this pathway is notorious for generating intractable black tar byproducts during the dichloroketene stage, complicating isolation and reducing overall yield. Additionally, this method typically produces a racemate, necessitating a subsequent resolution step to obtain the desired enantiomer, which inherently halves the theoretical yield and increases waste. Another approach utilizes asymmetric hydroboration of cyclopentadienylacetic acid, which avoids resolution but introduces operational complexities and limitations regarding scalability in industrial reactors.
Perhaps the most direct predecessor to the current invention is the Claisen rearrangement of 3-acyloxy-5-hydroxycyclopentenes. Literature precedents describe this transformation requiring extremely high temperatures, often reaching 160°C, to drive the reaction to completion. Such harsh thermal conditions pose severe risks for thermal degradation of sensitive functional groups and demand specialized, high-pressure equipment capable of withstanding intense heat. These factors collectively contribute to higher operational costs, increased safety risks, and difficulties in maintaining consistent product quality across large batches. Consequently, there has been a persistent industry demand for a simpler, more economical process that mitigates these thermal and purification challenges while preserving stereochemical fidelity.
The Novel Approach
The innovative process described in the patent fundamentally alters the reaction landscape by employing amide acetals or aminoketene acetals as key reagents in the Claisen rearrangement step. This strategic modification allows the rearrangement to proceed efficiently at significantly lower temperatures, specifically within the range of 90°C to 120°C. This reduction in thermal requirement is not merely incremental; it enables the use of standard production equipment and drastically lowers energy consumption, directly impacting the cost reduction in pharmaceutical intermediate manufacturing. The use of solvents with boiling points greater than 90°C, such as toluene or xylene, facilitates this温和er thermal profile while ensuring adequate solubility for the reactants.
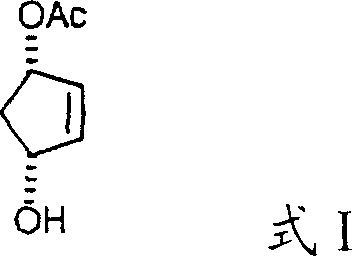
Furthermore, the novel approach incorporates a critical control parameter regarding the concentration of alcohol byproducts generated during the reaction. By maintaining the alcohol concentration at less than 3% by volume, the process effectively suppresses racemization, a common pitfall in high-temperature rearrangements. This ensures that the enantiomeric purity of the starting material, such as the 3S,5R isomer, is preserved throughout the transformation into the acyloxy(hydroxy)cyclopentene acetamide intermediate. This level of control over stereochemistry eliminates the need for difficult resolution steps post-reaction, streamlining the workflow and enhancing the overall economic viability of producing high-purity cyclopentenyl lactone for commercial applications.
Mechanistic Insights into Amide Acetal-Mediated Claisen Rearrangement
The core of this technological advancement lies in the mechanistic interaction between the 3-acyloxy-5-hydroxycyclopentene substrate and the amide acetal reagents. When compounds of Formula I react with amide acetals of Formula IIa or aminoketene acetals of Formula IIb, a transient intermediate is formed that lowers the activation energy required for the [3,3]-sigmatropic rearrangement. 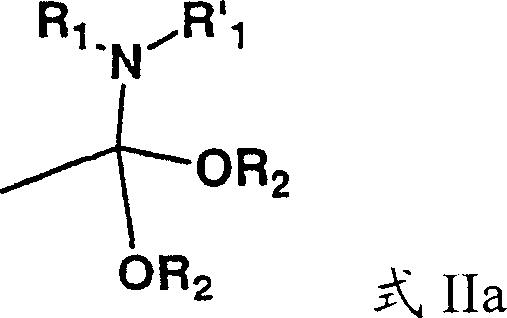 This catalytic-like effect allows the reaction to bypass the high energy barrier associated with traditional thermal Claisen rearrangements. The nitrogen-containing functionality of the acetal likely stabilizes the transition state through electronic effects, facilitating the migration of the allyl group and the formation of the new carbon-carbon bond at much milder temperatures. This mechanistic efficiency is crucial for preserving the integrity of the cyclopentene ring and preventing side reactions that typically occur under harsher thermal stress.
This catalytic-like effect allows the reaction to bypass the high energy barrier associated with traditional thermal Claisen rearrangements. The nitrogen-containing functionality of the acetal likely stabilizes the transition state through electronic effects, facilitating the migration of the allyl group and the formation of the new carbon-carbon bond at much milder temperatures. This mechanistic efficiency is crucial for preserving the integrity of the cyclopentene ring and preventing side reactions that typically occur under harsher thermal stress.
A critical aspect of the mechanism involves the management of the alcohol byproduct (R2OH) released during the transacetalization process. The patent data explicitly highlights that alcohol concentrations exceeding 3-5% lead to substantial racemization of the chiral centers. This phenomenon suggests that the alcohol may participate in a reversible equilibrium that scrambles the stereochemistry or catalyzes an unwanted epimerization pathway. By continuously distilling off the alcohol or using a nitrogen purge to maintain its concentration below 3%, the reaction equilibrium is driven forward towards the desired amide intermediate (Formula III) while locking in the stereochemical configuration. 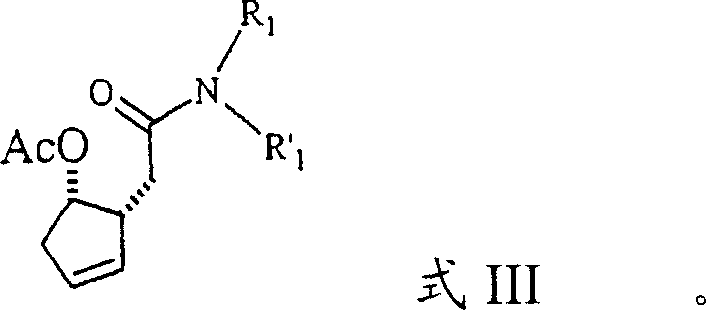 This precise control mechanism ensures that the final lactone product retains the high enantiomeric excess required for biological activity, demonstrating a sophisticated understanding of reaction kinetics and thermodynamics.
This precise control mechanism ensures that the final lactone product retains the high enantiomeric excess required for biological activity, demonstrating a sophisticated understanding of reaction kinetics and thermodynamics.
How to Synthesize 3,3a,6,6a-tetrahydro-2H-cyclopenta[b]furan-2-one Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and solvent management, to maximize yield and purity. The process begins with the heating of the starting cyclopentene derivative and the amide acetal in a suitable high-boiling solvent, followed by the continuous removal of volatile alcohol byproducts to drive the equilibrium. Once the rearrangement is complete, the crude amide intermediate undergoes hydrolysis using aqueous bases such as potassium hydroxide or sodium hydroxide, creating a biphasic or uniform mixture depending on the specific conditions. Finally, acidification with a strong mineral acid triggers the intramolecular cyclization to form the target lactone. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- React 3-acyloxy-5-hydroxycyclopentenes with amide acetals or aminoketene acetals at 90-120°C while maintaining alcohol concentration below 3%.
- Treat the resulting acyloxy(hydroxy)cyclopentene acetamide with a hydroxide or carbonate solution to form a uniform or biphasic mixture.
- Add a strong acid with a pKa value less than 2 to the mixture to effect cyclization and isolate the final lactone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative benefits for supply chain stability and cost management in the fine chemical sector. The elimination of extreme temperature requirements (dropping from 160°C to ~100°C) translates directly into reduced energy costs and decreased wear on reactor vessels, extending equipment lifespan and lowering maintenance overheads. Moreover, the avoidance of dichloroketene and the associated black tar byproducts simplifies the downstream purification process significantly. Traditional methods often require extensive chromatography or multiple crystallization steps to remove tarry impurities, which consumes time and solvents. By contrast, this cleaner reaction profile allows for more straightforward isolation techniques, thereby reducing lead time for high-purity prostaglandin precursors and accelerating time-to-market for finished drug products.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for expensive resolution steps that are typical in racemic synthesis routes. Since the reaction preserves enantiomeric purity through controlled conditions rather than correcting errors post-synthesis, the effective yield per batch is significantly higher. Additionally, the use of commodity chemicals like toluene and dimethylacetamide dimethyl acetal as reagents ensures that raw material costs remain stable and predictable. The simplified workup procedure further reduces labor hours and solvent consumption, contributing to a leaner manufacturing cost structure that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Robustness is a key feature of this synthesis, as it relies on stable reagents and moderate conditions that are less prone to batch-to-batch variability. The ability to use a wide range of solvents, including xylenes and various ethers, provides flexibility in sourcing raw materials, mitigating risks associated with supply shortages of specific chemicals. Furthermore, the scalability of the process from laboratory to industrial scale has been validated, ensuring that suppliers can meet fluctuating demand without compromising on quality. This reliability is crucial for pharmaceutical companies that require consistent supply of critical intermediates to maintain their own production schedules.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than conventional methods due to the absence of heavy metal catalysts and chlorinated byproducts. The reduction in hazardous waste generation simplifies compliance with increasingly stringent environmental regulations, reducing disposal costs and administrative burdens. The process is inherently designed for scale-up, with reaction conditions that are easily manageable in large-scale stirred tank reactors. This scalability ensures that production volumes can be increased to meet commercial demand, ranging from pilot plant quantities to multi-ton annual production, without encountering the engineering bottlenecks often seen in high-temperature or high-pressure processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of this critical lactone intermediate. These answers are derived directly from the experimental data and claims presented in the patent literature, providing clarity on the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions regarding technology adoption and supply partnerships. The focus is on practical implementation aspects that affect production planning and quality assurance protocols.
Q: Why is maintaining low alcohol concentration critical in this synthesis?
A: Keeping the alcohol concentration below 3% by volume is essential to minimize racemization of the chiral starting materials, ensuring high enantiomeric excess in the final prostaglandin intermediate.
Q: What are the temperature advantages of this new Claisen rearrangement method?
A: Unlike traditional methods requiring temperatures around 160°C, this process operates effectively between 90°C and 120°C, significantly reducing energy consumption and thermal stress on equipment.
Q: How does this process improve supply chain reliability compared to older methods?
A: By avoiding the formation of black tars associated with dichloroketene routes and eliminating the need for complex resolution steps, this method offers a more robust and predictable production timeline.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3a,6,6a-tetrahydro-2H-cyclopenta[b]furan-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of life-saving prostaglandin therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the enantiomeric excess and chemical purity of every batch, guaranteeing that our 3,3a,6,6a-tetrahydro-2H-cyclopenta[b]furan-2-one meets the highest industry standards. Our commitment to excellence extends beyond mere compliance; we actively optimize our processes to deliver superior value to our partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our efficient manufacturing methods can reduce your overall project costs. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey from research to commercial success with our reliable supply chain solutions.
