Advanced Cerium-Catalyzed Oxidation Technology for High-Purity Sulfoxide Pharmaceutical Intermediates
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for greener, more selective, and cost-effective methodologies. A pivotal advancement in this domain is detailed in patent CN102161633A, which discloses a novel preparation method for sulfoxide-type organic compounds. Sulfoxides are not merely academic curiosities; they are foundational building blocks in the pharmaceutical industry, serving as critical chiral ligands in asymmetric catalysis and as key structural motifs in bioactive natural products and drugs, such as proton pump inhibitors. The traditional reliance on harsh stoichiometric oxidants has long plagued the supply chain with toxicity and waste issues. This patent introduces a paradigm shift by utilizing cerium-based catalysts in conjunction with environmentally benign oxidants like atmospheric air or oxygen. This approach not only aligns with the principles of Green Chemistry but also addresses the critical pain points of selectivity and scalability that R&D directors and procurement managers face daily when sourcing reliable sulfoxide supplier materials.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of sulfides to sulfoxides has been fraught with significant technical and environmental challenges that hinder efficient commercial scale-up of complex organic sulfides. Conventional protocols frequently employ stoichiometric amounts of strong oxidizing agents such as chromium trioxide, potassium permanganate, sodium periodate, or meta-chloroperbenzoic acid (mCPBA). While effective in small-scale laboratory settings, these reagents present severe drawbacks for industrial application. Firstly, they are inherently hazardous, posing significant safety risks during handling and storage. Secondly, and perhaps more critically for supply chain heads, they generate substantial quantities of toxic heavy metal waste or organic byproducts that require costly and complex disposal procedures. Furthermore, a persistent chemical challenge with these strong oxidants is the difficulty in controlling the reaction at the sulfoxide stage; over-oxidation to the corresponding sulfone is a common side reaction, drastically reducing the yield of the desired target and complicating the purification process, thereby inflating the overall cost of goods.
The Novel Approach
In stark contrast to the brute-force chemistry of the past, the methodology outlined in CN102161633A offers a sophisticated, catalytic solution that elegantly circumvents these legacy issues. By employing cerium salts—specifically cerium trifluoromethanesulfonate, cerium chloride heptahydrate, ammonium cerium nitrate, cerium nitrate hexahydrate, or cerium(III) sulfate—as catalysts, the reaction achieves high conversion under remarkably mild conditions. The process operates at temperatures ranging from room temperature to 100°C, utilizing molecular oxygen, air, or tert-butyl hydroperoxide (TBHP) as the terminal oxidant. This shift from stoichiometric to catalytic oxidation fundamentally alters the economic and environmental profile of the synthesis. The use of air or oxygen as the oxidant is particularly transformative, as it is the cheapest and safest oxidant available, effectively eliminating the cost burden associated with purchasing expensive chemical oxidants. Moreover, the cerium catalyst system exhibits exceptional chemoselectivity, preferentially generating the sulfoxide while minimizing further oxidation to the sulfone, thus ensuring high-purity sulfoxide compounds are obtained with simplified downstream processing.
Mechanistic Insights into Cerium-Catalyzed Selective Oxidation
The efficacy of this cerium-catalyzed system lies in its unique ability to activate molecular oxygen or peroxides towards the sulfur center without degrading sensitive functional groups on the substrate. The general substrate scope is impressively broad, encompassing aryl, substituted aryl, alkyl, and alkenyl groups attached to the sulfur atom. As illustrated in the reaction schemes, the catalyst likely facilitates a single-electron transfer or a coordination mechanism that lowers the activation energy for the formation of the S=O bond. For instance, when using cerium nitrate hexahydrate in dimethoxyethane (DME) at 60°C under air, methyl phenyl sulfide is smoothly converted to methyl phenyl sulfoxide with high efficiency. This mechanistic pathway is robust enough to tolerate a wide array of electronic environments on the aromatic ring, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as nitro, cyano, and halogens.
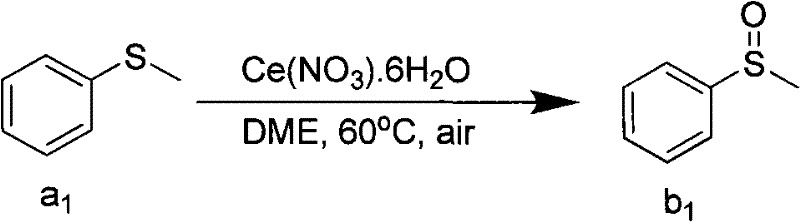
Beyond simple acyclic sulfides, the catalytic system demonstrates remarkable versatility with cyclic and heteroatom-containing substrates, which are often prone to ring-opening or decomposition under harsher oxidative conditions. The patent data highlights successful transformations of cyclic sulfides such as tetrahydrothiophene and 1,3-dithiane into their corresponding sulfoxides. For example, the oxidation of tetrahydrothiophene proceeds cleanly to yield tetrahydrothiophene sulfoxide, a valuable intermediate, using cerium nitrate and oxygen at 100°C. This capability is crucial for R&D teams working on complex molecule synthesis where the sulfur moiety is part of a larger, sensitive ring system. The impurity control mechanism is inherent to the mildness of the cerium catalyst; unlike ruthenium or vanadium systems that might require excess peroxide and risk over-oxidation, the cerium system appears to self-limit or be easily tuned to stop at the sulfoxide stage. This selectivity minimizes the formation of sulfone impurities, which are notoriously difficult to separate from sulfoxides due to similar polarity, thereby streamlining the purification workflow and enhancing the overall purity profile of the final API intermediate.
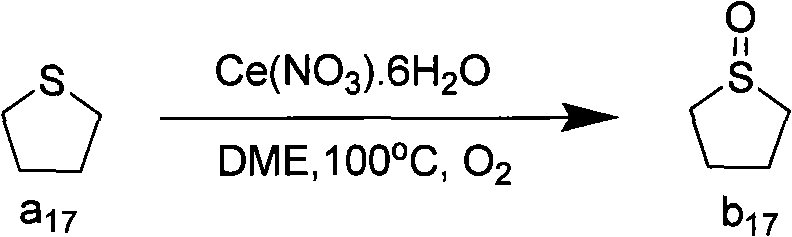
How to Synthesize Sulfoxide Efficiently
Implementing this technology requires a straightforward operational protocol that balances reaction kinetics with safety. The process begins by establishing the reaction system, comprising the thioether substrate, the chosen cerium catalyst, and an appropriate solvent if the substrate is not liquid at the reaction temperature. Solvents such as glycol dimethyl ether (DME), nitromethane, or acetonitrile are preferred for their ability to dissolve both the organic substrate and the inorganic catalyst while remaining stable under oxidative conditions. Once the mixture is prepared, the oxidant is introduced—either by bubbling oxygen/air through the solution or by adding a controlled amount of TBHP. The reaction is then heated to the specified range of 60°C to 100°C and stirred for a duration of 2 to 24 hours, depending on the specific substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by mixing the thioether substrate with a cerium catalyst (such as cerium nitrate hexahydrate or cerium chloride heptahydrate) in a suitable solvent like DME or acetonitrile.
- Introduce the oxidant, selecting from atmospheric air, pure oxygen, or tert-butyl hydroperoxide (TBHP), and heat the mixture to a temperature between 60°C and 100°C.
- Maintain the reaction for 2 to 24 hours, then perform an aqueous workup followed by extraction and silica gel chromatography to isolate the high-purity sulfoxide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cerium-catalyzed oxidation technology represents a strategic opportunity to optimize the supply base for sulfoxide intermediates. The shift away from hazardous stoichiometric oxidants towards catalytic air oxidation directly translates into significant operational efficiencies and risk mitigation. By eliminating the need for expensive and dangerous reagents like mCPBA or osmium tetroxide, manufacturers can drastically simplify their raw material inventory and reduce the regulatory burden associated with hazardous chemical storage. Furthermore, the high selectivity of the process means that less material is lost to over-oxidation byproducts, effectively increasing the mass balance and yield of the desired product without requiring complex recycling streams. This efficiency is a key driver for cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing structures in a tight market.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the replacement of expensive stoichiometric oxidants with virtually free atmospheric air or oxygen. In traditional processes, the cost of the oxidant and the subsequent waste treatment can constitute a major portion of the variable costs. By switching to a catalytic system where the cerium salt is used in sub-stoichiometric amounts (typically around 10-20 mol%), the direct material costs are significantly lowered. Additionally, the simplified workup procedure—often requiring only water washing and extraction rather than complex quenching of hazardous residues—reduces labor and utility costs. The avoidance of heavy metal contaminants like chromium or ruthenium also eliminates the need for expensive scavenging resins or specialized filtration steps, further contributing to substantial cost savings throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and commodity-grade reagents. Cerium salts are abundant lanthanides, and their supply is generally more stable compared to precious metals like ruthenium or palladium, which are subject to volatile geopolitical market fluctuations. The ability to use air or oxygen as the oxidant removes the dependency on specialized chemical suppliers for peracids or periodates, which can sometimes face availability bottlenecks. This decentralization of raw material sources ensures a more robust and continuous supply of high-purity sulfoxides, reducing lead time for high-purity sulfoxides and mitigating the risk of production stoppages due to reagent shortages. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors, avoiding the need for specialized corrosion-resistant equipment.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is vastly superior to conventional methods, facilitating easier regulatory approval and community acceptance. The generation of non-toxic byproducts and the absence of heavy metal waste streams simplify wastewater treatment and solid waste disposal, aligning with increasingly stringent global environmental regulations. The mild temperature range (60-100°C) and the use of air make the reaction inherently safer to scale up, reducing the risk of thermal runaways associated with concentrated peroxide oxidations. This safety profile allows for larger batch sizes and continuous flow processing opportunities, enabling the commercial scale-up of complex organic sulfides from pilot plant to multi-ton production with minimal engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this cerium-catalyzed oxidation technology. These insights are derived directly from the experimental data and claims within patent CN102161633A, providing a clear understanding of its capabilities and limitations for potential partners. Understanding these nuances is essential for evaluating the feasibility of this route for specific target molecules in your pipeline.
Q: Why is cerium catalysis preferred over traditional oxidants for sulfoxide synthesis?
A: Traditional oxidants like chromium trioxide or potassium permanganate are highly toxic and often lack selectivity, leading to over-oxidation into sulfones. The cerium-catalyzed method described in patent CN102161633A operates under mild conditions with air or oxygen, offering exceptional chemoselectivity that stops precisely at the sulfoxide stage without generating hazardous heavy metal waste.
Q: What types of thioether substrates are compatible with this oxidation method?
A: This catalytic system demonstrates broad substrate tolerance. It effectively oxidizes aromatic thioethers (such as methyl phenyl sulfide), aliphatic thioethers, benzyl sulfides, and even cyclic sulfides like tetrahydrothiophene. Functional groups such as methoxy, nitro, chloro, and cyano on the aromatic ring remain intact during the process.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable. It utilizes inexpensive and readily available oxidants like atmospheric air and avoids the need for cryogenic conditions or stoichiometric amounts of expensive reagents. The simple workup procedure involving water washing and standard extraction makes it ideal for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. We have closely analyzed the potential of the cerium-catalyzed oxidation pathway described in CN102161633A and are well-positioned to leverage this technology for our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the patent can be replicated and optimized on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of sulfoxide intermediate meets the exacting standards required for pharmaceutical applications. Our commitment to quality ensures that impurities, particularly the troublesome sulfone byproducts, are kept to negligible levels.
We invite you to collaborate with us to evaluate the feasibility of this green oxidation route for your specific projects. Whether you are looking to optimize an existing process or develop a new synthetic pathway for a challenging sulfoxide target, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out to our technical procurement team to obtain specific COA data for our reference standards and to discuss detailed route feasibility assessments. Together, we can drive down costs and enhance the sustainability of your supply chain while securing a reliable source of high-quality intermediates.
