Advanced Homogeneous Alkylation Strategy for Scalable Dipropylmalonic Acid Diester Production
Advanced Homogeneous Alkylation Strategy for Scalable Dipropylmalonic Acid Diester Production
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates, and the preparation of dipropylmalonic acid diester stands as a pivotal step in the manufacture of antiepileptic agents like sodium valproate. Patent CN103183612A introduces a transformative methodology that shifts the paradigm from cumbersome heterogeneous systems to efficient homogeneous alkylation processes. This innovation addresses long-standing challenges regarding reaction time, reagent stoichiometry, and environmental safety, providing a blueprint for modern chemical manufacturing. By leveraging specific alkoxide bases in compatible alcohol solvents, the process achieves remarkable efficiency without the need for phase transfer catalysts. This technical breakthrough not only enhances the purity profile of the resulting intermediate but also streamlines the operational workflow, making it an attractive option for large-scale production facilities aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dialkyl malonates has been plagued by inefficiencies inherent in biphasic reaction systems. Traditional methods, such as those described in US Patent 5856569, rely heavily on phase transfer catalysts like tetrabutylammonium bromide to facilitate reactions between organic halides and aqueous base solutions. These processes often require a massive excess of alkylating agents, sometimes up to fifteen times the stoichiometric amount, leading to significant raw material waste and complicated downstream purification steps to remove unreacted halides. Furthermore, the reaction times in these legacy systems are prohibitively long, frequently exceeding forty-five hours, which severely bottlenecks production capacity. The use of hazardous inorganic bases like cesium hydroxide or reactive metals like sodium in other prior art methods introduces severe safety risks and corrosion issues, while solvents like DMF pose difficulties in removal due to their high boiling points, ultimately inflating production costs and environmental footprints.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a homogeneous organic system where both the base and the solvent are chemically compatible, typically pairing an alkoxide base with its corresponding alcohol solvent. This strategic alignment ensures that the reaction proceeds in a single phase, eliminating mass transfer limitations and removing the necessity for phase transfer catalysts entirely. The reaction kinetics are dramatically accelerated, with completion achievable within nine hours, representing a substantial improvement over conventional timelines. Moreover, the stoichiometry is tightly controlled, requiring only a modest excess of the halopropane reactant, which minimizes waste and simplifies the isolation of the final product. This method allows for the direct use of the crude product in subsequent steps, thereby reducing the overall number of unit operations and enhancing the economic viability of the synthesis.
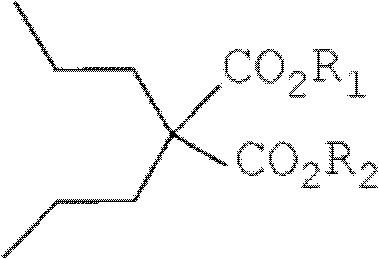
Mechanistic Insights into Alkoxide-Catalyzed Homogeneous Alkylation
The core of this technological advancement lies in the generation of a highly reactive enolate species within a homogeneous medium. When an alkoxide base, such as potassium tert-butoxide, is dissolved in an alcohol solvent like tert-butanol, it creates a uniform solution that readily deprotonates the active methylene group of the malonic acid diester. This deprotonation occurs rapidly and efficiently due to the lack of phase boundaries, ensuring that the nucleophilic enolate is immediately available to attack the electrophilic carbon of the 1-halopropane. The reaction follows a classic SN2 mechanism, where the steric environment is optimized by the choice of base and solvent to favor double alkylation at the central carbon atom. The homogeneity of the system prevents the localized high concentrations of base that can lead to side reactions or decomposition, thereby preserving the integrity of the ester groups and ensuring a clean impurity profile.
Impurity control is inherently managed through the precise selection of reaction parameters and the inherent stability of the homogeneous system. In heterogeneous systems, incomplete mixing often leads to pockets of unreacted starting material or over-alkylated byproducts, necessitating rigorous purification. However, in this novel process, the solubility of the base ensures consistent reactivity throughout the vessel, minimizing the formation of mono-alkylated intermediates or elimination byproducts. The use of potassium tert-butoxide in tert-butanol is particularly effective, as the bulky tert-butoxide ion acts as a strong base but a poor nucleophile, reducing the risk of nucleophilic attack on the ester carbonyls themselves. This selectivity is crucial for maintaining high product quality, allowing the crude material to meet stringent specifications with minimal processing, which is a key consideration for regulatory compliance in pharmaceutical manufacturing.
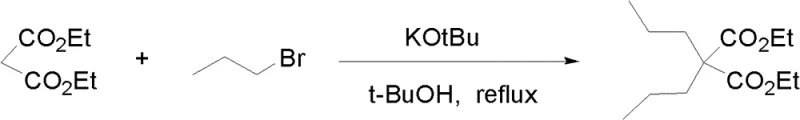
How to Synthesize Dipropylmalonic Acid Diester Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing, heating, and workup steps that are easily adaptable to standard reactor configurations. The process begins with the dissolution of the malonic acid diester in the chosen alcohol solvent, followed by the addition of the alkoxide base and the alkyl halide. A split-addition strategy is often employed, where a portion of the reagents is added initially, and the remainder is introduced after a set period to maintain optimal reaction conditions and drive the equilibrium towards the fully alkylated product. This operational simplicity, combined with the ability to filter off inorganic salts and concentrate the filtrate, makes the process highly amenable to automation and scale-up. For a detailed breakdown of the specific operational parameters and stoichiometric ratios required for optimal results, please refer to the standardized synthesis guide below.
- Prepare a homogeneous reaction mixture by dissolving malonic acid diester in an alcohol solvent such as tert-butanol, then add an alkoxide base like potassium tert-butoxide.
- Introduce 1-halopropane (preferably 1-bromopropane) to the mixture and heat to reflux conditions to initiate the double alkylation reaction.
- After approximately 5 hours, supplement with additional halopropane and base to drive completion, then filter and concentrate the filtrate to isolate the crude product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers profound benefits that directly address the pain points of procurement managers and supply chain directors. The elimination of phase transfer catalysts and the reduction in excess reagent usage translate into significant cost savings on raw materials, which is a critical factor in maintaining competitive pricing for high-volume intermediates. Additionally, the shortened reaction time drastically improves asset utilization, allowing manufacturing facilities to increase throughput without capital investment in new equipment. The simplified workup procedure reduces the consumption of solvents and utilities associated with extensive washing and distillation steps, further lowering the operational expenditure. These efficiencies collectively contribute to a more resilient supply chain capable of meeting tight delivery schedules while adhering to increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the elimination of expensive phase transfer catalysts and the minimization of raw material waste. By avoiding the use of fifteen-fold excesses of alkyl halides seen in older methods, the material cost per kilogram of product is substantially reduced. Furthermore, the ability to use the crude product directly in the next synthesis step removes the need for energy-intensive purification processes like column chromatography or multiple recrystallizations. This streamlined approach lowers the overall cost of goods sold, providing a distinct economic advantage in the competitive market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The use of stable, commercially available reagents like potassium tert-butoxide and common alcohol solvents ensures a secure supply of inputs, mitigating the risk of production delays caused by specialty chemical shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, leading to consistent batch-to-batch performance. This reliability is essential for maintaining continuous production schedules and fulfilling long-term supply agreements with downstream API manufacturers. The reduced reaction time also allows for greater flexibility in scheduling, enabling quicker response times to unexpected changes in demand.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction facilitates easy scale-up from laboratory to industrial production, as heat and mass transfer issues are minimized compared to slurry-based systems. The absence of hazardous reagents like metallic sodium or corrosive cesium hydroxide improves workplace safety and reduces the regulatory burden associated with handling dangerous goods. Moreover, the reduction in solvent usage and waste generation aligns with green chemistry principles, helping companies meet their sustainability goals and comply with environmental discharge standards. This eco-friendly profile enhances the corporate image and ensures long-term operational viability in a regulated industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs. The answers highlight the balance between chemical efficiency and operational practicality that defines this innovation.
Q: Why is the homogeneous alkoxide system superior to traditional phase transfer catalysis for this synthesis?
A: The homogeneous system eliminates the need for expensive and environmentally burdensome phase transfer catalysts, significantly simplifying post-reaction workup and reducing purification costs while accelerating reaction kinetics.
Q: What are the safety advantages of using potassium tert-butoxide over metallic sodium in this process?
A: Potassium tert-butoxide is safer to handle and store compared to pyrophoric metallic sodium, reducing operational risks and facilitating easier scale-up in industrial manufacturing environments.
Q: How does this method impact the overall yield and purity of the pharmaceutical intermediate?
A: By optimizing the base-solvent pairing, specifically using potassium tert-butoxide in tert-butanol, the method achieves crude yields exceeding 90 percent with high purity suitable for direct use in subsequent synthetic steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dipropylmalonic Acid Diester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch supports the efficacy and safety of the final therapeutic agent. Our expertise in homogeneous alkylation technologies allows us to offer a reliable supply of dipropylmalonic acid diester that is both cost-effective and consistently available.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this superior method. We encourage you to contact us for specific COA data and route feasibility assessments to ensure that our capabilities align perfectly with your development timelines and quality standards. Let us collaborate to drive efficiency and innovation in your supply chain.
