Advanced Butterfly-Structure OLED Emitters: Scalable Synthesis and Commercial Viability
The rapid evolution of the display industry demands organic electroluminescent materials that combine high efficiency with exceptional thermal stability. Patent CN112062775B introduces a groundbreaking class of organic photoelectric materials characterized by a unique three-dimensional butterfly structure. Unlike traditional planar molecules that suffer from aggregation-caused quenching, these compounds feature a rigid, symmetric D3h steric aromatic structure that effectively inhibits intermolecular crystallization. This structural innovation results in amorphous materials with superior film-forming properties, making them ideal candidates for the emissive layers of next-generation OLED displays. The patent discloses a series of compounds, designated D01 through D52, which demonstrate remarkable improvements in device performance metrics.
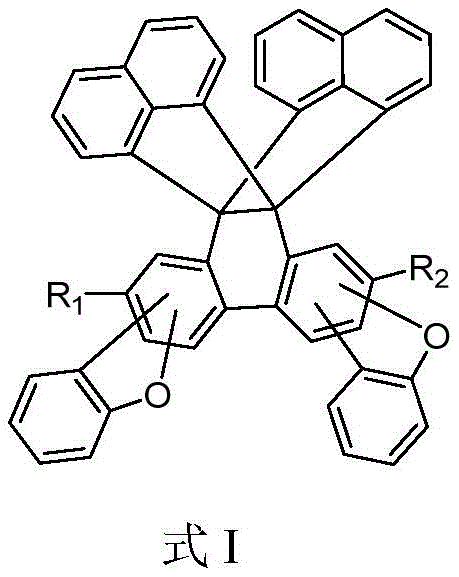
As a reliable OLED material supplier, understanding the underlying architecture is crucial. The core of these molecules consists of three aromatic rings forming well-defined angles, creating open electron-rich cavities and significant internal free volume. This design not only enhances the glass transition temperature but also optimizes the charge transport balance within the device. For procurement managers and supply chain heads, this translates to a high-purity OLED material that reduces the risk of device failure due to thermal degradation, thereby lowering the total cost of ownership for display manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic light-emitting diodes has been hindered by the inherent limitations of planar conjugated systems. Conventional emitters often exhibit strong pi-pi stacking interactions, which lead to undesirable crystallization during device operation or storage. This aggregation phenomenon drastically reduces luminescence efficiency and shortens the operational lifespan of the display panel. Furthermore, many existing synthetic routes rely on harsh conditions or complex purification steps to achieve the necessary purity levels, resulting in low overall yields and increased production costs. The inability to maintain an amorphous state under thermal stress remains a critical bottleneck in the commercial scale-up of complex polymer additives and small molecule emitters alike.
The Novel Approach
The novel approach detailed in the patent overcomes these challenges by engineering a non-planar, twisted molecular geometry. By incorporating a butterfly-shaped rigid group, the synthesis creates a spatial barrier that physically prevents close packing of the molecules. This structural modification ensures that the material remains amorphous even at elevated temperatures, as evidenced by glass transition temperatures exceeding 160°C. The preparation method is designed to be simple and convenient, utilizing accessible raw materials and standard reaction conditions. This facilitates cost reduction in electronic chemical manufacturing by minimizing waste and simplifying the downstream processing requirements, ultimately providing a more robust supply chain for high-performance display components.
Mechanistic Insights into Palladium-Catalyzed Cyclization and Coupling
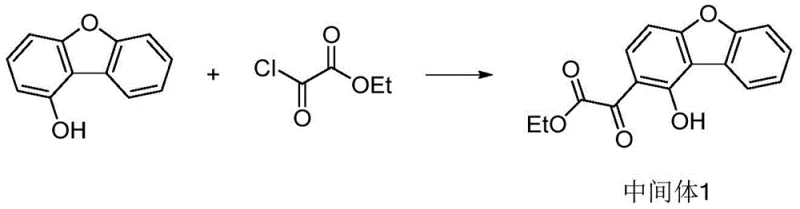
The synthesis of these advanced materials relies on a sophisticated sequence of organic transformations, prominently featuring palladium-catalyzed cross-coupling and Lewis acid-mediated cyclization. The process begins with the functionalization of a dibenzofuran derivative, where precise control over regioselectivity is paramount. For instance, the initial acylation step utilizes titanium tetrachloride to activate the aromatic ring, allowing for the introduction of the oxalate moiety which serves as a precursor for the central rigid core. Subsequent bromination and triflation steps activate specific positions on the aromatic ring, setting the stage for the critical carbon-carbon bond-forming events that define the molecular skeleton.
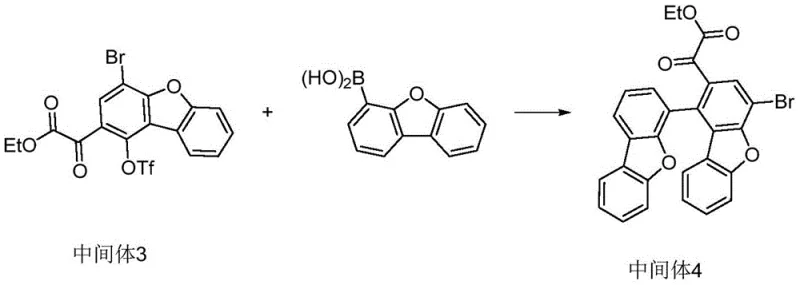
The construction of the butterfly framework is achieved through a strategic Suzuki-Miyaura coupling followed by an intramolecular cyclization. In this phase, a palladium catalyst facilitates the coupling of the activated aryl triflate with a boronic acid derivative, linking two aromatic systems with high fidelity. Following hydrolysis, titanium tetrachloride is employed again to induce cyclization, closing the ring to form the rigid central unit. This step is crucial for establishing the 3D geometry that imparts the desired thermal properties. The final stages involve Grignard addition to introduce naphthyl groups and a Buchwald-Hartwig amination to attach the charge-transporting amine substituents, completing the complex architecture.
How to Synthesize Butterfly-Structure OLED Material Efficiently
The preparation of compounds such as D01 involves a nine-step synthetic route that balances chemical complexity with operational feasibility. The process starts with readily available 1-hydroxydibenzofuran and proceeds through sequential functionalization, coupling, and cyclization reactions. Each step is optimized to maximize yield and purity, with specific attention paid to temperature control and reagent stoichiometry. The detailed standardized synthesis steps provided in the patent serve as a robust blueprint for industrial production, ensuring consistent quality across batches. For a comprehensive guide on executing this synthesis, please refer to the structured protocol below.
- Perform Friedel-Crafts acylation of 1-hydroxydibenzofuran with ethoxyoxalyl chloride using TiCl4 to form Intermediate 1.
- Execute bromination with NBS followed by triflation to activate the aromatic ring for cross-coupling.
- Conduct Suzuki coupling with dibenzofuran boronic acid, followed by hydrolysis and TiCl4-mediated cyclization to build the rigid core.
- Complete the synthesis via Grignard addition, acid treatment, and final Buchwald-Hartwig amination with aromatic amines.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this butterfly-structure technology offers significant strategic advantages for the supply chain. The synthesis route avoids the use of exotic or prohibitively expensive reagents, relying instead on commodity chemicals like N-bromosuccinimide and standard palladium catalysts. This accessibility ensures a stable supply of raw materials, mitigating the risk of production delays caused by sourcing bottlenecks. Furthermore, the high thermal stability of the final product reduces the need for aggressive cooling systems in the final display assembly, indirectly contributing to energy savings and manufacturing efficiency.
- Cost Reduction in Manufacturing: The synthetic pathway is designed for high atom economy and operational simplicity. By eliminating the need for complex chiral resolutions or extreme cryogenic conditions often required for sensitive intermediates, the process significantly lowers energy consumption and equipment wear. The use of recyclable solvents such as toluene and dichloromethane further optimizes the cost structure. Additionally, the high yields reported in the experimental examples suggest that waste generation is minimized, leading to substantial cost savings in waste disposal and raw material procurement.
- Enhanced Supply Chain Reliability: The reliance on established chemical transformations such as Suzuki coupling and Friedel-Crafts reactions means that the process can be easily transferred between different manufacturing sites without significant re-engineering. This flexibility enhances supply chain resilience, allowing for multi-sourcing strategies that protect against regional disruptions. The robustness of the intermediates, which can often be carried forward without rigorous purification, streamlines the production timeline and reduces lead time for high-purity OLED materials.
- Scalability and Environmental Compliance: The reaction conditions, ranging from ambient to moderately elevated temperatures, are well-suited for large-scale reactor operations. The process avoids the generation of hazardous byproducts that would require specialized treatment, aligning with modern environmental compliance standards. The ability to scale from laboratory gram quantities to multi-ton annual production volumes without compromising purity specifications makes this technology highly attractive for meeting the growing global demand for high-resolution displays.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these organic electroluminescent materials. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for R&D teams evaluating the material for integration into existing device architectures.
Q: What are the thermal stability characteristics of these butterfly-structure materials?
A: The materials exhibit high glass transition temperatures (Tg) ranging from 163°C to 171°C and decomposition temperatures (Td) exceeding 469°C, ensuring robust performance in OLED devices.
Q: How does the 3D structure improve device lifetime?
A: The unique 3D rigid butterfly structure prevents intermolecular crystallization and aggregation, leading to superior film-forming properties and significantly extended operational lifetimes compared to planar analogs.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation method utilizes standard organic solvents and common catalysts like palladium acetate, with reaction conditions suitable for large-scale manufacturing from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Butterfly-Structure OLED Material Supplier
The technical potential of the butterfly-structure compounds described in CN112062775B represents a significant leap forward in OLED emitter technology. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such complex molecules to market. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of material performs consistently in your high-value display applications. We understand the critical nature of supply continuity in the electronics sector and are committed to being a dependable partner.
We invite you to initiate a dialogue regarding your specific material requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes. By collaborating with us, you can access specific COA data and route feasibility assessments that will help you optimize your supply chain and accelerate your product development cycles. Let us help you engineer the future of display technology with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
