Advanced Iodine-Catalyzed Synthesis of 3-Substituted 4-Hydroxycoumarins for Commercial Scale-Up
Introduction to Novel Catalytic Methodology in Coumarin Chemistry
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the urgent need for greener, more cost-effective, and operationally simpler methodologies. Patent CN101613336B introduces a transformative approach to the preparation of 3-substituted 4-hydroxycoumarin derivatives, a class of compounds serving as critical scaffolds for anticoagulants and other bioactive agents. This technology leverages molecular iodine as a highly efficient, non-metallic catalyst to facilitate the direct alkylation of 4-hydroxycoumarin with various benzyl alcohols. Unlike traditional methods that often rely on toxic heavy metals or expensive rare earth catalysts, this iodine-catalyzed protocol operates under remarkably mild thermal conditions, typically ranging from 50°C to 80°C, thereby preserving the structural integrity of sensitive functional groups. The strategic implementation of this chemistry not only addresses environmental concerns regarding heavy metal waste but also streamlines the production workflow by eliminating the need for rigorous inert atmosphere protection. For R&D directors and process chemists, this represents a significant opportunity to optimize existing synthetic routes for high-value coumarin derivatives, ensuring both economic viability and regulatory compliance in the manufacture of complex pharmaceutical intermediates.
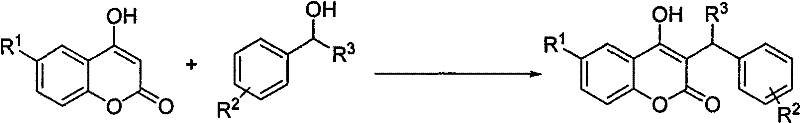
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted 4-hydroxycoumarins has been plagued by significant technical and economic hurdles that hinder efficient large-scale production. Conventional protocols frequently employ stoichiometric amounts of toxic heavy metal salts, such as lead acetate, which pose severe environmental disposal challenges and introduce risks of residual metal contamination in the final active pharmaceutical ingredient. Furthermore, alternative catalytic systems utilizing strong Lewis acids like ytterbium trifluoroacetate or acidic cation exchange resins often necessitate harsh reaction conditions, including elevated temperatures and strictly anhydrous environments, which can lead to the degradation of the coumarin core or unwanted side reactions. These legacy methods also tend to suffer from limited substrate scope, struggling to accommodate diverse benzyl alcohol derivatives without significant loss in yield or selectivity. The operational complexity associated with removing trace metal catalysts to meet stringent pharmaceutical purity standards adds substantial downstream processing costs, making these traditional routes increasingly unattractive for modern, sustainability-focused manufacturing facilities seeking to reduce their ecological footprint while maintaining high productivity.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN101613336B utilizes molecular iodine as a benign, inexpensive, and highly effective catalyst to drive the alkylation reaction with exceptional efficiency. This innovative approach capitalizes on the unique electrophilic properties of iodine to activate the hydroxyl group of the benzyl alcohol, generating a reactive intermediate that readily undergoes nucleophilic attack by the electron-rich 4-hydroxycoumarin scaffold. The reaction proceeds smoothly in polar organic solvents such as nitromethane or nitroethane, eliminating the necessity for exotic or hazardous reagents that complicate supply chain logistics. Crucially, the process tolerates ambient atmospheric conditions, removing the capital expenditure and operational burden associated with maintaining nitrogen or argon blankets throughout the reaction cycle. By shifting from toxic heavy metals to a simple halogen catalyst, this method drastically simplifies the workup procedure, allowing for easy removal of the catalyst via sodium sulfite quenching, which translates directly into higher overall process throughput and reduced waste generation for procurement and supply chain teams managing complex chemical inventories.
Mechanistic Insights into Iodine-Catalyzed Electrophilic Substitution
The mechanistic pathway of this iodine-catalyzed transformation involves a sophisticated interplay of electrophilic activation and nucleophilic substitution that ensures high regioselectivity at the 3-position of the coumarin ring. Initially, the molecular iodine interacts with the hydroxyl group of the benzyl alcohol substrate, facilitating the departure of the hydroxyl moiety and generating a stabilized benzylic carbocation or an iodonium intermediate species. This activated electrophile is then strategically attacked by the enol form of the 4-hydroxycoumarin, which acts as a potent nucleophile due to the electron-donating resonance effects of the adjacent phenolic hydroxyl group. The reaction kinetics are finely tuned by the choice of solvent, with nitromethane providing an optimal dielectric environment that stabilizes the charged transition states without promoting excessive side reactions. This precise control over the reaction mechanism allows for the successful incorporation of a wide variety of substituents, including halogens, alkoxy groups, and styryl moieties, without compromising the yield or purity of the final product. For technical teams, understanding this mechanism is vital for troubleshooting potential scale-up issues, as it highlights the importance of maintaining optimal catalyst loading ratios between 0.08 and 0.20 molar equivalents to ensure complete conversion while minimizing iodine-related impurities.
Impurity control in this synthesis is inherently robust due to the mild nature of the catalytic cycle, which avoids the aggressive conditions that typically lead to polymeric byproducts or ring-opening degradation of the lactone moiety. The use of molecular iodine, rather than strong Brønsted or Lewis acids, prevents the protonation of the coumarin carbonyl oxygen, a common pathway for decomposition in acidic media. Furthermore, the quenching step utilizing saturated sodium sulfite solution effectively reduces any residual molecular iodine to water-soluble iodide salts, ensuring that the organic phase remains free from colored contaminants that could interfere with downstream crystallization or purification steps. This inherent cleanliness of the reaction profile means that the crude product often requires minimal purification, typically achievable through standard column chromatography or recrystallization techniques. For quality assurance professionals, this translates to a more predictable impurity profile, simplifying the validation of analytical methods and ensuring consistent batch-to-batch reproducibility essential for regulatory filings and commercial supply agreements.
How to Synthesize 3-Substituted 4-Hydroxycoumarin Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the iodine catalysis system. The process begins by dissolving equimolar amounts of 4-hydroxycoumarin and the selected benzyl alcohol derivative in a suitable volume of nitromethane, ensuring complete solubilization before the introduction of the catalyst. Once the homogeneous solution is established, molecular iodine is added, and the mixture is heated to the target temperature range of 50°C to 80°C, where it is maintained for a duration of 1 to 12 hours depending on the specific electronic nature of the substrates involved. Monitoring the reaction progress via thin-layer chromatography or HPLC is recommended to determine the optimal endpoint, preventing over-reaction that could lead to minor byproduct formation. Upon completion, the reaction is carefully quenched to neutralize the catalyst, followed by a standard extraction and drying protocol to isolate the crude material. The detailed standardized synthesis steps, including specific molar ratios, exact temperature ramps, and purification parameters for various derivatives, are outlined in the guide below.
- Dissolve 4-hydroxycoumarin and the corresponding benzyl alcohol derivative in an organic solvent such as nitromethane or nitroethane under standard atmospheric conditions.
- Add molecular iodine as the catalyst (0.08-0.20 molar equivalents) and heat the reaction mixture to a temperature range of 50-80°C for 1 to 12 hours to facilitate the alkylation.
- Quench the reaction with saturated sodium sulfite solution, extract the product with ethyl acetate, dry the organic phase, and purify via column chromatography to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iodine-catalyzed methodology offers profound advantages for procurement managers and supply chain heads tasked with optimizing the cost structure and reliability of pharmaceutical intermediate sourcing. The substitution of expensive and regulated heavy metal catalysts with commodity-grade molecular iodine results in a drastic reduction in raw material costs, directly impacting the bottom line of manufacturing operations without sacrificing product quality. Moreover, the elimination of toxic metals simplifies the regulatory compliance landscape, reducing the administrative burden and costs associated with environmental health and safety reporting, waste disposal, and residual metal testing. The operational simplicity of the process, which does not require specialized inert atmosphere equipment or extreme temperature controls, lowers the barrier to entry for contract manufacturing organizations, thereby increasing the pool of qualified suppliers and enhancing supply chain resilience against disruptions. This flexibility allows for more agile response to market demand fluctuations, ensuring continuous availability of critical coumarin intermediates for downstream drug production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost catalytic systems with inexpensive molecular iodine, which is globally available and priced significantly lower than rare earth triflates or organometallic complexes. By removing the need for expensive metal scavengers and complex purification trains required to meet strict heavy metal limits, the overall processing cost per kilogram is substantially decreased. Additionally, the high atom economy and excellent yields reported in the patent examples mean that less raw material is wasted, further driving down the cost of goods sold. This cost efficiency is compounded by the ability to run reactions at moderate temperatures, which reduces energy consumption for heating and cooling compared to processes requiring cryogenic conditions or high-temperature reflux, delivering comprehensive savings across the entire manufacturing value chain.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of widely available, non-strategic raw materials such as nitromethane and molecular iodine, which are not subject to the same geopolitical supply constraints as specialty catalysts or precious metals. The robustness of the reaction conditions, which tolerate ambient air and moisture to a reasonable extent, reduces the risk of batch failures due to minor deviations in operational parameters, leading to more predictable production schedules. This reliability is crucial for long-term supply agreements, as it minimizes the likelihood of delays caused by equipment maintenance for inert gas systems or the sourcing of hard-to-find reagents. Consequently, procurement teams can negotiate more favorable terms with suppliers who adopt this technology, secure in the knowledge that production continuity is less vulnerable to external logistical shocks or raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the straightforward engineering requirements, as the reaction does not involve exothermic hazards or the generation of toxic gaseous byproducts. The environmental profile is markedly improved by the absence of heavy metal waste streams, aligning with increasingly stringent global regulations on pharmaceutical manufacturing emissions and effluent discharge. The simplified workup procedure generates less solvent waste and reduces the volume of hazardous waste requiring specialized treatment, contributing to a smaller carbon footprint and lower disposal costs. For companies committed to sustainable chemistry principles, this methodology offers a clear pathway to greener manufacturing practices, enhancing corporate reputation and meeting the sustainability criteria often demanded by major pharmaceutical clients in their vendor assessment protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-catalyzed synthesis route, providing clarity for stakeholders evaluating its adoption for their specific supply chains. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of integrating this technology into existing production lines or for qualifying new suppliers capable of executing this advanced chemistry. We encourage technical teams to review these points closely to identify opportunities for process optimization and cost savings within their current intermediate sourcing strategies.
Q: What are the primary advantages of using molecular iodine over traditional Lewis acid catalysts for coumarin alkylation?
A: Molecular iodine offers a significantly cheaper and less toxic alternative to heavy metal catalysts like lead acetate or rare earth triflates. It operates under mild conditions (50-80°C) without requiring strict air exclusion, simplifying the operational complexity and reducing safety hazards associated with handling corrosive strong acids or toxic heavy metals.
Q: How does this process ensure high purity for pharmaceutical intermediate applications?
A: The mild reaction conditions minimize side reactions such as polymerization or decomposition of the sensitive coumarin scaffold. Furthermore, the straightforward workup procedure involving sodium sulfite quenching and ethyl acetate extraction effectively removes iodine residues and unreacted starting materials, facilitating high-purity isolation suitable for downstream API synthesis.
Q: Is this synthesis method scalable for industrial production of anticoagulant precursors?
A: Yes, the process demonstrates excellent scalability potential due to its reliance on inexpensive, commercially available reagents and the absence of stringent anhydrous or inert atmosphere requirements. The use of common solvents like nitromethane and simple purification techniques makes it highly adaptable for large-scale manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted 4-Hydroxycoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to deliver high-quality pharmaceutical intermediates that meet the rigorous demands of the global healthcare industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of the iodine-catalyzed coumarin synthesis can be fully realized at an industrial level. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-substituted 4-hydroxycoumarin delivered meets the highest standards for downstream API synthesis. Our commitment to process excellence means we can seamlessly transition this novel chemistry from the lab bench to full-scale manufacturing, providing our partners with a reliable source of complex intermediates that are both cost-effective and environmentally responsible.
We invite procurement leaders and R&D directors to engage with us to explore how this advanced synthesis route can optimize your supply chain and reduce overall manufacturing costs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible benefits of partnering with a supplier who prioritizes innovation and operational excellence. Let us collaborate to secure a sustainable and efficient supply of high-purity coumarin derivatives for your next generation of therapeutic products.
