Optimizing Ticagrelor Intermediate Purity: A Breakthrough in Industrial Purification Technology
Optimizing Ticagrelor Intermediate Purity: A Breakthrough in Industrial Purification Technology
The pharmaceutical landscape for antiplatelet therapies continues to evolve, with Ticagrelor standing as a cornerstone treatment for acute coronary syndromes. Central to the efficient manufacturing of this potent P2Y12 receptor antagonist is the availability of high-quality key intermediates, specifically 4,6-dichloro-2-(thiopropyl)-5-aminopyrimidine. Recent advancements detailed in patent CN111763175A introduce a robust purification methodology that addresses longstanding challenges in impurity profile management and product appearance. This technical insight report analyzes the novel five-step purification protocol, offering R&D and procurement leaders a comprehensive understanding of how this technology enhances supply chain reliability and cost-efficiency in the production of critical cardiovascular drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 4,6-dichloro-2-(thiopropyl)-5-aminopyrimidine, particularly those stemming from diethyl acetylaminomalonate precursors as disclosed in earlier patents like WO2014023681A1, often result in crude products with significant quality deficits. Following the final chlorination step using phosphorus oxychloride, standard workup procedures involving distillation and aqueous quenching frequently yield a brown oily substance rather than a crystalline solid. This crude material typically suffers from low purity and content, harboring difficult-to-remove impurities that persist even after attempted recrystallization with ethyl acetate and petroleum ether mixtures. The presence of these persistent impurities not only compromises the aesthetic quality of the intermediate but also poses severe risks for downstream coupling reactions required to build the triazolopyrimidine core of Ticagrelor.
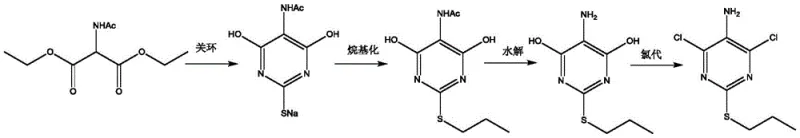
The Novel Approach
In stark contrast to these conventional limitations, the purification method described in CN111763175A implements a sophisticated sequence of oil removal, salt formation, dissociation, and recrystallization to transform low-grade crude material into a premium pharmaceutical intermediate. The process begins with a targeted adsorption step using silica gel in a low-polarity solvent like n-hexane, which effectively strips away colored oily byproducts that traditional washing cannot eliminate. Subsequent conversion of the intermediate into its hydrochloride salt allows for a powerful purification checkpoint, leveraging the differential solubility of the salt versus neutral impurities. This strategic intervention ensures that the final free base, obtained after controlled liberation and recrystallization, exhibits an off-white appearance with purity levels exceeding 99.5%, thereby setting a new benchmark for intermediate quality in the industry.
Mechanistic Insights into Adsorption and Salt-Formation Purification
The efficacy of this purification strategy lies in its multi-modal approach to impurity rejection, combining physical adsorption with chemical differentiation. During the oil removal phase, the use of silica gel in n-hexane at elevated temperatures (50-60°C) capitalizes on the polar interactions between the silica surface and polar impurities, while the target pyrimidine derivative remains soluble in the non-polar medium. This step is critical for removing the dark-colored tarry substances often generated during harsh chlorination conditions. Furthermore, the subsequent salt formation step utilizes the basic nature of the aminopyrimidine nitrogen. By introducing concentrated hydrochloric acid in methanol, the target molecule precipitates as a hydrochloride salt, leaving behind non-basic organic impurities in the solution. This acid-base manipulation acts as a highly selective filter, ensuring that only the desired chemical entity proceeds to the final liberation and crystallization stages.
Impurity control is further refined during the liberation and final recrystallization steps, where precise temperature management plays a pivotal role. The dissociation of the hydrochloride salt is conducted at low temperatures (0-10°C) using alkaline solutions, which promotes the formation of uniform crystals while minimizing the occlusion of mother liquor contaminants. The final recrystallization in n-hexane serves as a polishing step, removing any trace residual solvents or minor byproducts that may have survived previous stages. This rigorous control over thermodynamic parameters ensures that the crystal lattice formed is highly ordered, resulting in a product with exceptional chemical stability and consistent particle size distribution, which are essential attributes for reliable downstream processing in API manufacturing.
How to Synthesize 4,6-Dichloro-2-(thiopropyl)-5-aminopyrimidine Efficiently
The implementation of this purification protocol requires careful attention to solvent ratios and temperature gradients to maximize recovery and purity. The process is designed to be integrated directly after the chlorination reaction workup, treating the crude concentrate without the need for intermediate isolation of unstable species. Operators must strictly adhere to the specified mass ratios of adsorbents and solvents to ensure effective impurity capture without excessive product loss. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate these high-yield results in a pilot or production environment.
- Perform chlorination and workup to obtain crude product 1, removing excess phosphorus oxychloride.
- Execute oil removal by dissolving crude product in n-hexane and adsorbing impurities with silica gel.
- Form hydrochloride salt in methanol, followed by liberation to free base and final recrystallization in n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced purification technology translates into tangible strategic benefits beyond mere technical specifications. By significantly enhancing the purity and physical appearance of the intermediate, manufacturers can reduce the risk of batch rejections during API synthesis, thereby stabilizing the overall production schedule. The elimination of complex and costly chromatographic purification steps in favor of crystallization and filtration operations drastically simplifies the manufacturing workflow. This simplification not only reduces the consumption of high-cost solvents and consumables but also shortens the overall cycle time per batch, allowing for increased throughput and better responsiveness to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing inefficient purification attempts with a high-yield crystallization sequence. By avoiding the need for repeated recrystallizations or expensive column chromatography to remove colored impurities, the overall consumption of solvents and energy is significantly lowered. The high recovery rate of the target compound at each stage ensures that raw material costs are amortized over a greater quantity of saleable product, directly improving the margin profile for the intermediate. Furthermore, the use of commodity chemicals like n-hexane and hydrochloric acid keeps operational expenditures predictable and manageable compared to specialized reagents.
- Enhanced Supply Chain Reliability: The robustness of this purification method ensures consistent output quality, which is critical for maintaining uninterrupted API production lines. The ability to consistently produce off-white, high-purity material reduces the variability often associated with batch-to-batch processing of complex heterocycles. This consistency allows supply chain planners to forecast inventory levels with greater accuracy and reduces the need for safety stock buffers typically held to mitigate quality risks. Additionally, the scalability of the filtration and crystallization steps means that production capacity can be expanded linearly without encountering the bottlenecks often seen with more sensitive purification technologies.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process favors widely accepted solvents and avoids the generation of heavy metal waste streams often associated with catalytic reduction purification methods. The reliance on physical separation techniques like filtration and centrifugation simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles facilitates easier regulatory compliance and permits faster approval cycles for new manufacturing sites. The straightforward nature of the unit operations also lowers the barrier for technology transfer to different geographic locations, ensuring a resilient and diversified global supply network.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their existing supply chains or development pipelines.
Q: What are the primary impurities removed by this purification method?
A: The method effectively removes colored oily impurities and residual chlorinating agents through silica gel adsorption and salt formation, transforming brown oil into off-white crystals.
Q: How does this process improve yield compared to conventional recrystallization?
A: By utilizing a sequential salt formation and dissociation strategy prior to final recrystallization, the process minimizes product loss in mother liquors, achieving yields exceeding 90%.
Q: Is this purification method suitable for large-scale manufacturing?
A: Yes, the process avoids complex chromatography and uses common solvents like n-hexane and methanol, making it highly scalable for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Dichloro-2-(thiopropyl)-5-aminopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final API is inextricably linked to the purity of its precursors. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that technologies like the one described in CN111763175A are implemented with precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4,6-dichloro-2-(thiopropyl)-5-aminopyrimidine meets the exacting standards required for cardiovascular drug manufacturing. We are committed to delivering consistent, high-quality intermediates that support your regulatory filings and commercial success.
We invite you to engage with our technical procurement team to discuss how our advanced purification capabilities can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our streamlined processes can reduce your overall cost of goods. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and reliability.
