Advanced Synthesis of Trans-4-(Trans-4'-Alkyl Cyclohexyl) Cyclohexanal for High-Performance Liquid Crystal Applications
The development of high-performance liquid crystal materials relies heavily on the availability of precise intermediates with exceptional stereochemical purity. Patent CN101671242B introduces a groundbreaking method for synthesizing trans-4-(trans-4'-alkyl cyclohexyl) cyclohexanal, a critical building block for medium-to-high grade mixed liquid crystal formulations. This technology addresses the longstanding challenges of viscosity and response speed in liquid crystal displays by providing a route that ensures high trans-isomer content while minimizing environmental impact. The innovation lies in a two-step sequence that bypasses the hazardous and costly steps associated with conventional methodologies, offering a robust solution for the commercial scale-up of complex electronic chemical intermediates. By leveraging specific borohydride reductions followed by TEMPO-catalyzed oxidations, this process guarantees the structural integrity of the cyclohexane skeleton, which is paramount for the thermal stability and nematic phase range of the final display material.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclohexane-based aldehydes has been plagued by inefficient and environmentally detrimental pathways that hinder cost reduction in display material manufacturing. Traditional routes often involve the conversion of carboxylic acids to acyl chlorides, a step that generates significant amounts of corrosive hydrogen chloride gas and sulfur dioxide byproducts, posing severe health risks to operators and requiring expensive scrubbing systems. Furthermore, subsequent reduction steps frequently rely on noble metal catalysts such as palladium-carbon, which not only drive up raw material costs but also necessitate rigorous and costly heavy metal removal processes to meet pharmaceutical and electronic grade purity standards. Another common approach utilizes triethyl silane, a highly flammable liquid with a low flash point, creating substantial safety hazards during large-scale production and complicating logistics for a reliable liquid crystal intermediate supplier. These legacy methods often struggle to maintain stereochemical purity, requiring multiple recrystallization steps that drastically reduce overall yield and increase waste generation.

The Novel Approach
In stark contrast to the hazardous legacy pathways, the novel method described in the patent utilizes a streamlined and environmentally benign strategy that significantly enhances process safety and efficiency. The process initiates with the direct reduction of trans-4-(trans-4'-alkyl cyclohexyl) cyclohexyl carboxylic acid using borohydride reagents, such as zinc borohydride or potassium borohydride, which are safer and more cost-effective than silane-based alternatives. This is followed by a highly selective oxidation step employing substituted TEMPO radicals, sodium bromide, and sodium hypochlorite, which operates under mild conditions to yield the target aldehyde without compromising the cyclohexane configuration. This approach eliminates the need for acyl chloride intermediates and noble metal catalysts, thereby removing major bottlenecks related to waste treatment and catalyst recovery. The result is a synthesis that is not only cheaper to execute but also inherently safer, making it an ideal candidate for reducing lead time for high-purity electronic chemical intermediates in a regulated manufacturing environment.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Stereochemical Control
The core of this technological advancement lies in the sophisticated use of substituted TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) derivatives as organocatalysts for the oxidation of the intermediate alcohol to the aldehyde. Unlike harsh oxidizing agents that might epimerize the sensitive cyclohexane ring, the TEMPO-mediated system operates via a radical mechanism that is highly selective for primary alcohols while leaving the stereocenters untouched. The presence of sodium bromide acts as a co-catalyst to facilitate the regeneration of the active oxoammonium species from sodium hypochlorite, ensuring a continuous and efficient catalytic cycle. This mechanism allows the reaction to proceed at low temperatures, typically below 5°C, which further suppresses any potential side reactions or thermal degradation of the product. The ability to control the oxidation state precisely without affecting the geometric configuration of the molecule is crucial for maintaining the liquid crystalline properties of the final material.
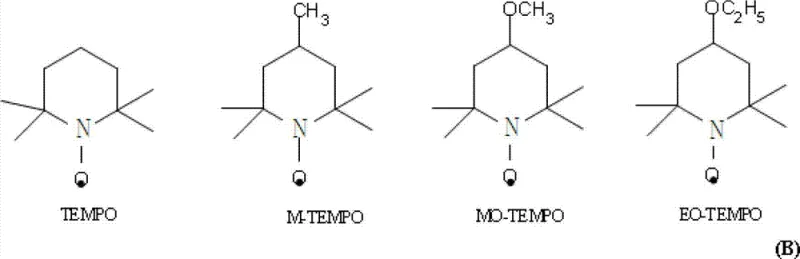
Furthermore, the preservation of the trans-configuration throughout the synthesis is a critical quality attribute that this mechanism guarantees. The starting carboxylic acid is stable and readily available with high trans-purity, and the subsequent reduction and oxidation steps are designed to be configurationally neutral. This means that the cis-isomer content in the final aldehyde product can be consistently maintained below 0.1%, a specification that is difficult to achieve with routes involving high-temperature isomerization or harsh acidic conditions. High trans-purity is essential because cis-isomers can disrupt the alignment of liquid crystal molecules, leading to poor contrast ratios and slower response times in display panels. By ensuring that the synthetic pathway does not involve configuration transformation, the process provides a high-purity liquid crystal intermediate that meets the stringent requirements of advanced display technologies without the need for extensive and yield-losing purification steps.
How to Synthesize Trans-4-(Trans-4'-Alkyl Cyclohexyl) Cyclohexanal Efficiently
The implementation of this synthesis requires careful control of reaction parameters to maximize yield and safety, particularly during the exothermic oxidation phase. The process begins with the preparation of the alcohol intermediate via borohydride reduction in a solvent like tetrahydrofuran, followed by a workup that ensures the removal of boron residues. The subsequent oxidation step demands precise temperature control, typically maintaining the reaction mixture below 5°C during the addition of the oxidant to manage heat release effectively. Detailed standard operating procedures regarding reagent addition rates, stirring speeds, and quenching protocols are essential for reproducibility. For a comprehensive guide on the specific molar ratios, solvent volumes, and isolation techniques required to replicate this patent's success, please refer to the standardized synthesis steps provided below.
- Reduce trans-4-(trans-4'-alkyl cyclohexyl) cyclohexyl carboxylic acid using a borohydride reagent such as zinc borohydride or potassium borohydride to form the corresponding alcohol intermediate.
- Oxidize the resulting alcohol intermediate using a catalytic system comprising substituted TEMPO, sodium bromide, and sodium hypochlorite under controlled low-temperature conditions.
- Isolate the final trans-4-(trans-4'-alkyl cyclohexyl) cyclohexanal product through standard extraction and purification techniques, ensuring cis-isomer content remains below 0.1%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly address the pain points of procurement and supply chain management in the fine chemical sector. By shifting away from noble metal catalysts and hazardous silane reagents, the process fundamentally alters the cost structure of production, leading to substantial savings in both raw material procurement and waste disposal. The use of common, stable reagents like borohydrides and bleach simplifies the supply chain, reducing the risk of disruptions caused by the scarcity of specialized catalysts. Additionally, the mild reaction conditions lower the energy consumption and equipment maintenance costs associated with high-pressure or high-temperature reactors. These factors combine to create a more resilient and cost-effective supply chain for reliable liquid crystal intermediate supplier networks, ensuring consistent availability of critical materials for downstream display manufacturers.
- Cost Reduction in Manufacturing: The elimination of palladium-carbon catalysts and acyl chloride reagents removes significant cost centers from the production budget. Noble metals are subject to volatile market pricing and require expensive recovery processes, while acyl chlorides generate corrosive waste that demands costly neutralization and disposal. By replacing these with inexpensive borohydrides and TEMPO systems, the overall cost of goods sold is drastically reduced. Furthermore, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the loss of valuable starting materials and lowers the cost of purification. This economic efficiency allows for more competitive pricing strategies without compromising on the quality or purity of the final electronic chemical product.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable raw materials significantly de-risks the supply chain. Reagents such as sodium hypochlorite and potassium borohydride are commodity chemicals with robust global supply networks, unlike specialized silanes or noble metals which can face supply constraints. This availability ensures that production schedules can be maintained without interruption, providing a reliable flow of materials to customers. Moreover, the stability of the intermediates allows for safer storage and transportation, reducing the logistical complexities and insurance costs associated with hazardous materials. This reliability is crucial for maintaining the continuity of supply for high-volume display manufacturing lines that cannot afford downtime due to material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily managed in large-scale reactors. The absence of highly flammable solvents like triethyl silane and the use of aqueous oxidants simplify the safety protocols required for scale-up, facilitating a smoother transition from pilot plant to commercial production. Environmentally, the reduction in hazardous waste generation aligns with increasingly strict global regulations on chemical manufacturing. The lower pollution load reduces the burden on wastewater treatment facilities and minimizes the environmental footprint of the production site. This compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a key factor in supplier selection for major multinational electronics corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthetic technology. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for R&D and procurement teams evaluating the feasibility of adopting this route for their specific product lines. The information below highlights the key differentiators in terms of purity, safety, and cost efficiency that define the value proposition of this technology.
Q: How does the TEMPO oxidation method improve stereochemical purity compared to traditional routes?
A: The TEMPO-mediated oxidation operates under mild conditions that do not induce configuration transformation of the cyclohexane ring. This ensures that the trans-stereochemistry of the starting acid is preserved throughout the synthesis, resulting in a final product with cis-isomer content lower than 0.1%, which is critical for liquid crystal performance.
Q: What are the primary cost drivers eliminated by this synthetic route?
A: This route eliminates the need for expensive noble metal catalysts like palladium-carbon and avoids the use of hazardous acyl chloride intermediates. By utilizing common borohydrides and organocatalytic TEMPO systems, the process significantly reduces raw material costs and removes the expense associated with heavy metal scavenging and waste treatment.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It uses stable, commercially available raw materials and avoids highly dangerous reagents like triethyl silane. The reaction conditions are mild and controllable, making it safe and efficient for commercial scale-up of complex electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-(Trans-4'-Alkyl Cyclohexyl) Cyclohexanal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of advanced liquid crystal displays. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative TEMPO-mediated synthesis described in CN101671242B can be effectively translated into industrial reality. We are committed to delivering products that meet stringent purity specifications, with cis-isomer content controlled to the lowest possible levels to guarantee optimal display performance. Our rigorous QC labs employ state-of-the-art analytical techniques to verify every batch, providing our partners with the confidence that their supply chain is built on a foundation of technical excellence and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this cost-effective and environmentally friendly technology into your production portfolio. Together, we can drive innovation in the electronic materials sector while achieving significant operational efficiencies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
