Scalable Production of m-Aminophenylacetylene via Seyferth-Gilbert Homologation
Introduction to Next-Generation m-Aminophenylacetylene Synthesis
The pharmaceutical and advanced materials industries rely heavily on high-purity building blocks like m-aminophenylacetylene, a critical intermediate for synthesizing polyimide resins used in aerospace applications and tyrosine kinase inhibitors such as Erlotinib. Traditional manufacturing routes have long been plagued by reliance on expensive halogenated starting materials and toxic transition metal catalysts, creating bottlenecks in both cost and supply continuity. A breakthrough detailed in patent CN103724211A introduces a streamlined, three-step methodology that fundamentally shifts the economic landscape of producing this valuable alkyne. By leveraging the Seyferth-Gilbert homologation using the Bestmann-Ohira reagent, this novel approach bypasses the need for precious metal catalysis entirely.
This technical insight report analyzes the strategic implications of adopting this patented route for large-scale commercial production. The process begins with the in-situ generation of a diazophosphonate species, which reacts efficiently with commodity-grade m-nitrobenzaldehyde to install the terminal alkyne functionality. Unlike legacy methods that struggle with low yields and difficult purification, this pathway offers a robust alternative suitable for multi-ton manufacturing. The structural integrity of the final product, essential for downstream polymerization or drug synthesis, is maintained through mild reaction conditions that minimize thermal degradation.
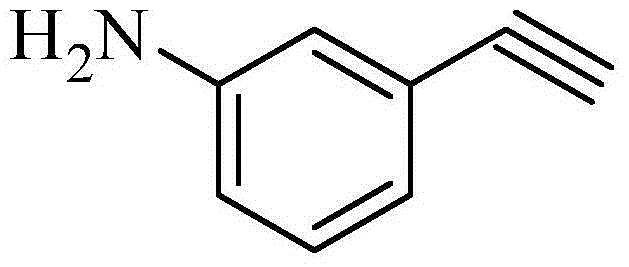
For R&D directors and procurement strategists, understanding the mechanistic advantages of this route is paramount. The shift from halogenated aromatics to nitro-aldehydes represents a significant decoupling from volatile halogen markets. Furthermore, the elimination of palladium residues simplifies the regulatory burden for API intermediates, ensuring that the final material meets stringent heavy metal specifications without additional scavenging steps. This report will dissect the reaction engineering, compare it against conventional limitations, and outline the tangible supply chain benefits for global chemical buyers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of m-aminophenylacetylene has relied heavily on transition-metal catalyzed cross-coupling reactions, specifically the Sonogashira coupling. As noted in prior art such as US Patent No. 20060224016, this traditional route typically employs 3-haloanilines or iodo-nitrobenzenes reacting with protected alkynes in the presence of palladium and copper catalysts. While chemically effective on a small scale, this method presents severe drawbacks for industrial application. The primary concern is the exorbitant cost of the catalyst system; palladium is a precious metal with fluctuating market prices, and its inclusion necessitates rigorous downstream purification to meet ppm-level residue limits required by pharmaceutical regulators.
Furthermore, alternative routes involving Vilsmeier-Haack formylation followed by elimination, or multi-step debromination sequences, suffer from poor atom economy and hazardous operating conditions. For instance, methods utilizing phosphorus oxychloride generate significant acidic waste streams, while debromination steps often require harsh basic conditions that lead to polymerization byproducts and reduced yields, often reported below 60%. These inefficiencies translate directly into higher manufacturing costs and increased environmental compliance burdens, making them less attractive for modern green chemistry initiatives.
The Novel Approach
The methodology disclosed in CN103724211A circumvents these historical bottlenecks by utilizing a diazo-phosphonate mediated homologation strategy. Instead of coupling a halide with an alkyne, this approach builds the alkyne directly onto an aldehyde scaffold using the Bestmann-Ohira reagent. This reagent, (1-diazo-2-oxopropyl) dimethyl phosphonate, acts as a synthetic equivalent of the acetylide anion but operates under much milder, metal-free conditions. The reaction proceeds through a unique mechanism where the diazo compound attacks the carbonyl carbon, followed by a rearrangement that extrudes nitrogen gas and a phosphate group to reveal the triple bond.
This novel pathway offers distinct operational advantages, including the ability to use crude reagents without intermediate purification, which drastically reduces solvent consumption and processing time. The starting material, m-nitrobenzaldehyde, is a widely available commodity chemical, ensuring stable pricing and supply security compared to specialized halogenated intermediates. Additionally, the final reduction step can be performed using economical iron powder rather than expensive catalytic hydrogenation setups, further lowering the barrier to entry for large-scale production facilities.
Mechanistic Insights into Seyferth-Gilbert Homologation
The core of this innovative synthesis lies in the generation and reactivity of the Bestmann-Ohira reagent. The process initiates with the deprotonation of acetonyl dimethyl phosphonate by a strong base, such as sodium hydride, to form a reactive carbanion. This nucleophile then attacks p-toluenesulfonyl azide, displacing the tosylate group to yield the unstable diazo intermediate. In the subsequent key step, this diazo species undergoes base-catalyzed deprotonation to form a diazo-stabilized anion, which performs a nucleophilic addition to the carbonyl group of m-nitrobenzaldehyde.
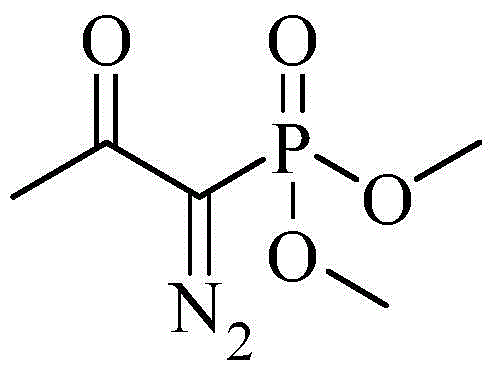
Following the initial addition, the tetrahedral intermediate collapses via an intramolecular attack of the alkoxide on the phosphorus atom, forming a transient four-membered oxaphosphetane ring. The fragmentation of this ring releases dimethyl phosphate and generates a vinyl diazonium species. The driving force of the reaction is the rapid loss of nitrogen gas from this vinyl diazonium intermediate, which triggers a 1,2-shift to produce the terminal alkyne, m-nitrophenylacetylene. This mechanism is highly advantageous because it avoids the formation of metal-acetylide complexes that can be explosive or difficult to handle on a large scale.
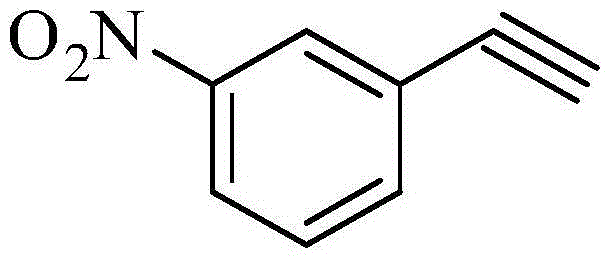
From an impurity control perspective, this mechanism is remarkably clean. The byproducts are primarily nitrogen gas and water-soluble phosphates, which are easily separated from the organic product during aqueous workup. Unlike palladium-catalyzed routes that often generate homocoupled diyne byproducts (Glaser coupling), the metal-free nature of this homologation virtually eliminates such dimerization impurities. This results in a crude product profile that is significantly easier to purify, often requiring only crystallization or simple distillation rather than complex chromatography, thereby enhancing the overall throughput of the manufacturing line.
How to Synthesize m-Aminophenylacetylene Efficiently
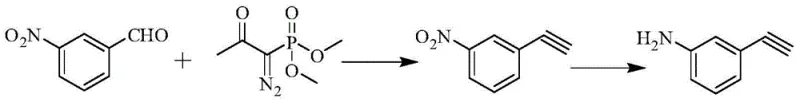
Implementing this synthesis requires careful control of reaction stoichiometry and temperature, particularly during the generation of the diazo reagent. The patent outlines a robust protocol where the Bestmann-Ohira reagent is generated in situ or prepared separately and then reacted with m-nitrobenzaldehyde in a polar protic solvent like methanol or ethanol. The use of mild bases such as potassium carbonate in the second step ensures that the sensitive diazo group reacts selectively with the aldehyde without decomposing. The final reduction of the nitro group to the amine is achieved using iron powder in an acidic alcoholic medium, a classic and scalable reduction method.
- Preparation of (1-diazo-2-oxopropyl) dimethyl phosphonate (Bestmann-Ohira reagent) from acetonyl dimethyl phosphonate and p-toluenesulfonyl azide under strong alkaline conditions.
- Reaction of the crude Bestmann-Ohira reagent with m-nitrobenzaldehyde in the presence of a basic catalyst (e.g., potassium carbonate) to form m-nitrophenylacetylene.
- Reduction of the nitro group in m-nitrophenylacetylene using iron powder or catalytic hydrogenation to yield the final m-aminophenylacetylene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented route offers transformative benefits in terms of cost structure and risk mitigation. The most immediate impact is the drastic reduction in raw material costs. By substituting expensive iodo- or bromo-nitrobenzenes with m-nitrobenzaldehyde, manufacturers can leverage the economies of scale associated with commodity aldehydes. This switch not only lowers the direct material bill but also insulates the production process from the volatility of the halogen market, which is often subject to supply disruptions and price spikes due to environmental regulations on halogenated solvents.
- Cost Reduction in Manufacturing: The elimination of palladium and copper catalysts removes a significant line item from the production budget. Precious metal catalysts are not only expensive to purchase but also incur hidden costs related to recovery, recycling, and disposal of metal-laden waste. Furthermore, the ability to use crude intermediates without purification reduces solvent usage and energy consumption associated with distillation steps. The process operates at atmospheric pressure and moderate temperatures, avoiding the need for specialized high-pressure reactors or cryogenic cooling systems, which translates to lower capital expenditure and operational overhead.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified by relying on m-nitrobenzaldehyde, a chemical produced in massive quantities for the dye and fragrance industries. This ensures a stable, multi-vendor supply base, reducing the risk of single-source dependency often seen with specialized halogenated intermediates. Additionally, the simplified workflow, which avoids complex multi-step protection and deprotection sequences, shortens the overall manufacturing cycle time. This agility allows suppliers to respond more rapidly to fluctuations in demand from downstream pharmaceutical or polymer clients.
- Scalability and Environmental Compliance: The environmental profile of this process is superior to traditional methods. The absence of heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with discharging metal-contaminated effluents. The byproducts, primarily nitrogen gas and phosphate salts, are benign and easily managed. This aligns with increasingly strict global environmental standards, facilitating smoother permitting processes for plant expansion. The robustness of the iron powder reduction step also ensures that the process can be scaled from pilot kilograms to multi-ton commercial batches without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield expectations, safety profiles, and purification requirements for potential licensees or contract manufacturers.
Q: Why is the Seyferth-Gilbert route preferred over Sonogashira coupling for this intermediate?
A: The Seyferth-Gilbert route avoids the use of expensive palladium and copper catalysts required in Sonogashira coupling. It utilizes readily available m-nitrobenzaldehyde instead of costly halo-anilines or iodo-nitrobenzenes, significantly reducing raw material costs and eliminating the need for complex heavy metal removal processes.
Q: What are the purity advantages of using iron powder reduction in the final step?
A: Iron powder reduction is a classical, robust method that avoids the potential safety hazards associated with high-pressure hydrogenation while still achieving high conversion rates. The resulting iron salts are easily removed by filtration, simplifying the workup compared to methods requiring high-vacuum distillation.
Q: Can the Bestmann-Ohira reagent be used directly without purification?
A: Yes, the patent demonstrates that the crude (1-diazo-2-oxopropyl) dimethyl phosphonate obtained from the first step can be used directly in the homologation reaction with m-nitrobenzaldehyde. This telescoping capability reduces solvent usage and processing time, enhancing overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Aminophenylacetylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our technical team has extensively evaluated the route described in CN103724211A and confirmed its viability for commercial scale-up. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of m-aminophenylacetylene meets the exacting standards required for API synthesis and high-performance polymer applications.
We invite you to collaborate with us to optimize your supply chain for this critical building block. By leveraging our expertise in process chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization.
