Advanced Manufacturing of 4-Trifluoromethylsulfanyl Aniline for High-Performance Veterinary Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to synthesize critical intermediates, particularly for the burgeoning sector of green veterinary medicines. Patent CN101265224A introduces a groundbreaking methodology for the preparation of 4-trifluoromethylsulfanyl aniline, a pivotal building block for advanced anticoccidial drugs such as toltrazuril. This compound is essential for developing veterinary therapeutics that offer rapid metabolism and minimal environmental impact, addressing the growing global demand for sustainable livestock management solutions. The disclosed process represents a significant technological leap by optimizing the fluorination step, traditionally a bottleneck in organofluorine chemistry, thereby enhancing both economic viability and operational safety.
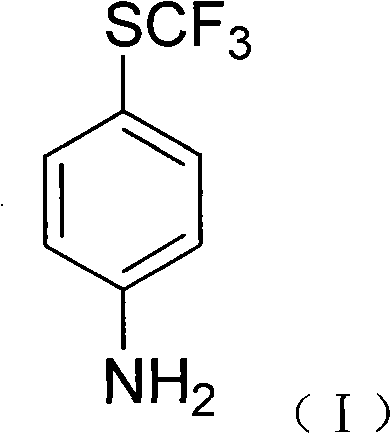
Historically, the synthesis of fluorinated aniline derivatives has been plagued by reliance on scarce reagents and hazardous conditions. Conventional methodologies, as cited in prior art such as US3632629 and EP 546391, predominantly utilized antimony trifluoride (SbF3) as the fluorinating agent. This approach suffered from severe limitations, including the high cost and limited commercial availability of antimony trifluoride, which created supply chain vulnerabilities for manufacturers. Furthermore, these legacy processes often necessitated harsh reaction environments, such as prolonged exposure to chlorine gas under intense illumination, leading to complex post-treatment procedures, lower overall yields, and significant challenges in maintaining product purity standards required for pharmaceutical applications.
In stark contrast, the novel approach detailed in the patent data revolutionizes this synthetic landscape by substituting antimony trifluoride with anhydrous hydrogen fluoride (HF). This strategic shift not only drastically reduces raw material costs but also simplifies the reaction engineering requirements, allowing for the use of more common industrial equipment. The new route proceeds through a logical sequence of nucleophilic substitution, chlorination, fluorination, and finally catalytic hydrogenation, each step optimized for mild conditions and high conversion rates. By eliminating the need for expensive heavy metal fluorinating agents and avoiding extreme photochemical conditions, this method offers a robust, scalable, and environmentally friendlier alternative for producing high-value veterinary intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The traditional synthesis of 4-trifluoromethylsulfanyl aniline was heavily dependent on the Swarts reaction variant using antimony trifluoride, a reagent known for its prohibitive cost and supply instability. Manufacturers faced significant hurdles in sourcing high-purity SbF3, often leading to production delays and inflated operational expenditures that eroded profit margins. Additionally, the reaction conditions associated with these older patents were notoriously difficult to control, requiring specialized reactors capable of withstanding long-term illumination and aggressive chlorination environments. These harsh parameters frequently resulted in the formation of complex impurity profiles, necessitating rigorous and costly purification steps to meet the stringent quality specifications demanded by the veterinary pharmaceutical sector.
The Novel Approach
The innovative strategy presented in CN101265224A overcomes these historical barriers by implementing a streamlined four-step synthesis that prioritizes reagent accessibility and process safety. By utilizing anhydrous hydrogen fluoride, a widely available industrial chemical, the process achieves high-efficiency fluorination without the economic burden of antimony salts. The reaction conditions are remarkably mild, with temperatures ranging between 30°C and 100°C across the various stages, allowing for precise thermal control and minimizing the risk of thermal runaway or side reactions. This methodological refinement ensures that the final product, 4-trifluoromethylsulfanyl aniline, is obtained with superior purity and yield, directly translating to enhanced process reliability for commercial manufacturers.
Mechanistic Insights into the Multi-Step Synthesis Route
The synthesis begins with a nucleophilic aromatic substitution where a 4-halogenated nitrobenzene reacts with a methyl mercaptan salt, such as sodium methyl mercaptide, in a solvent like acetonitrile. This initial step effectively installs the sulfur moiety onto the aromatic ring, forming 4-methylthio nitrobenzene with high regioselectivity. The reaction is typically conducted under reflux conditions for 12 to 24 hours, ensuring complete consumption of the starting halide. Following isolation, the intermediate undergoes a radical chlorination process where chlorine gas is introduced in the presence of a catalyst like benzoyl peroxide or diisopropyl azodicarboxylate. This transformation converts the methylthio group into a trichloromethylthio group, setting the stage for the subsequent fluorination.
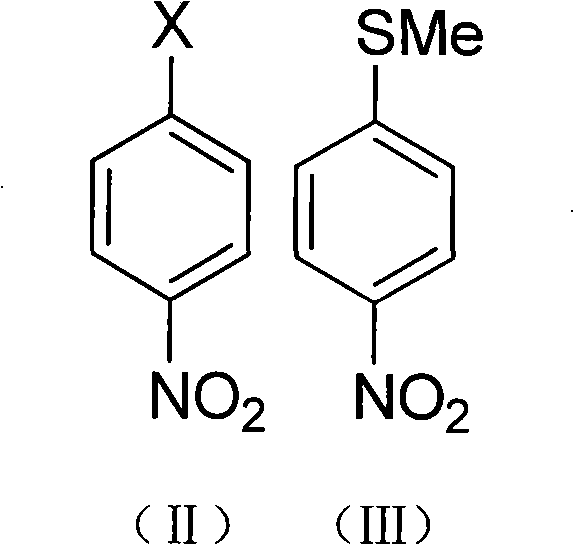
The core innovation lies in the fluorination step, where 4-trichloromethylthio nitrobenzene reacts with anhydrous hydrogen fluoride in a chlorinated solvent such as 1,2-dichloroethane. Under heated conditions, typically around 80°C, the chlorine atoms on the sulfur-bound carbon are exchanged for fluorine atoms, yielding 4-trifluoromethylthio nitrobenzene. This halogen exchange is highly efficient and avoids the generation of heavy metal waste streams associated with antimony reagents. The final step involves the catalytic hydrogenation of the nitro group using Raney nickel or palladium on carbon under hydrogen pressure (0.5-8MPa). This reduction cleanly converts the nitro functionality to the primary amine, delivering the target 4-trifluoromethylsulfanyl aniline with excellent purity.
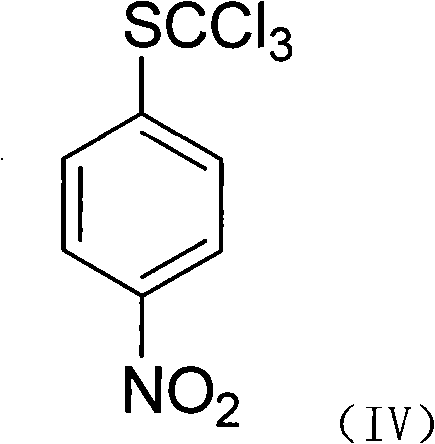
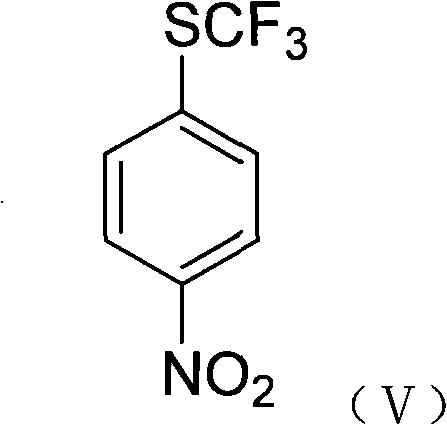
How to Synthesize 4-Trifluoromethylsulfanyl Aniline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of stoichiometric control and temperature management. To achieve optimal results, practitioners should adhere to the specified molar ratios, particularly using a slight excess of the methyl mercaptan salt (1.2 equivalents) to drive the initial substitution to completion. The chlorination step requires careful monitoring of chlorine feed rates to prevent overflow and ensure safety, while the fluorination step benefits from the use of autoclaves capable of handling anhydrous HF safely. Detailed standardized operating procedures for each unit operation are critical for maintaining batch-to-batch consistency.
- Perform nucleophilic substitution of 4-halogenated nitrobenzene with methyl mercaptan salt to obtain 4-methylthio nitrobenzene.
- Conduct chlorination of 4-methylthio nitrobenzene using chlorine gas to form 4-trichloromethylthio nitrobenzene.
- React 4-trichloromethylthio nitrobenzene with anhydrous hydrogen fluoride to achieve fluorination, yielding 4-trifluoromethylthio nitrobenzene.
- Complete the synthesis via catalytic hydrogenation of the nitro group to produce the final 4-trifluoromethylsulfanyl aniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic advantages centered around cost stability and operational resilience. The substitution of expensive, niche reagents like antimony trifluoride with commodity chemicals like anhydrous hydrogen fluoride fundamentally alters the cost structure of the manufacturing process. This shift mitigates the risk of price volatility associated with rare earth or heavy metal reagents, ensuring more predictable budgeting and long-term cost competitiveness in the global veterinary intermediate market.
- Cost Reduction in Manufacturing: The elimination of antimony trifluoride removes a significant cost driver from the bill of materials, as HF is produced on a massive industrial scale and is considerably cheaper. Furthermore, the simplified post-treatment processes reduce the consumption of solvents and energy required for purification, leading to substantial overall savings in production costs. The avoidance of complex photochemical reactors also lowers capital expenditure requirements for new production lines, making the technology accessible for a wider range of manufacturers.
- Enhanced Supply Chain Reliability: By relying on raw materials that are widely available in the chemical market, such as 4-halogenated nitrobenzenes and chlorine gas, manufacturers can secure a more robust supply chain less prone to disruptions. The use of standard catalysts like Raney nickel and palladium carbon, which are easily sourced from multiple global suppliers, further diversifies the supply base and reduces dependency on single-source vendors. This redundancy is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal waste streams simplify the environmental compliance landscape, reducing the costs and complexities associated with waste disposal and treatment. The process is inherently safer and easier to scale from kilogram to multi-ton quantities without requiring exotic engineering solutions, facilitating rapid commercialization. This scalability ensures that suppliers can quickly ramp up production to meet surging demand for green veterinary drugs without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-trifluoromethylsulfanyl aniline, based on the specific innovations disclosed in the patent literature. Understanding these details helps stakeholders evaluate the feasibility and benefits of integrating this technology into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this specific chemical pathway.
Q: What is the primary advantage of using anhydrous hydrogen fluoride in this synthesis?
A: The use of anhydrous hydrogen fluoride replaces expensive and less available antimony trifluoride, significantly reducing raw material costs and simplifying equipment requirements while maintaining high fluorination efficiency.
Q: How does this method improve upon conventional synthesis routes for veterinary intermediates?
A: Unlike conventional methods that require harsh illumination conditions and complex post-treatment, this novel approach utilizes mild reaction conditions (30-100°C) and standard operations, resulting in higher yields and easier scalability.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process relies on readily available market raw materials and avoids complex apparatus, making it highly suitable for industrial scale-up with consistent quality control for veterinary drug manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Trifluoromethylsulfanyl Aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation veterinary therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global pharmaceutical partners. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
