Advanced Synthesis of 4-Piperazinyl Benzothiophene Hydrochloride for Commercial Scale-Up
Advanced Synthesis of 4-Piperazinyl Benzothiophene Hydrochloride for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and cost-effective pathways for synthesizing critical intermediates, particularly for high-value antipsychotic agents like brexpiprazole. Patent CN115894434A introduces a groundbreaking preparation method for 4-piperazinyl benzothiophene hydrochloride, a pivotal intermediate in the production of this therapeutic agent. This innovative protocol diverges from traditional reliance on expensive noble metal catalysts and scarce starting materials, instead leveraging a six-step sequence initiated by low-cost thiophene. By integrating Friedel-Crafts acylation, Huang Minlon reduction, and a novel iodine-catalyzed aromatization, this technology offers a scalable solution that addresses both economic and environmental constraints in modern fine chemical manufacturing. The strategic shift away from palladium-dependent coupling reactions represents a significant technical evolution, promising enhanced purity profiles and streamlined operational workflows for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-piperazinyl benzothiophene derivatives has been hindered by significant technological bottlenecks associated with transition metal catalysis and precursor availability. Conventional routes, such as those disclosed in earlier patents, frequently employ Buchwald-Hartwig coupling reactions utilizing 4-bromobenzothiophene and piperazine in the presence of palladium catalysts. While chemically feasible, these methods impose severe economic burdens due to the high cost of palladium and the requisite phosphine ligands, alongside the complex downstream processing needed to remove trace heavy metals to meet pharmaceutical standards. Furthermore, these coupling reactions are notoriously prone to forming double-coupling impurities, which complicates purification and reduces overall yield. Alternative approaches starting from 4-aminobenzothiophene face different challenges, primarily the exorbitant cost and limited commercial availability of the starting material itself, rendering them unsuitable for large-scale industrial application where consistent supply and cost efficiency are paramount.
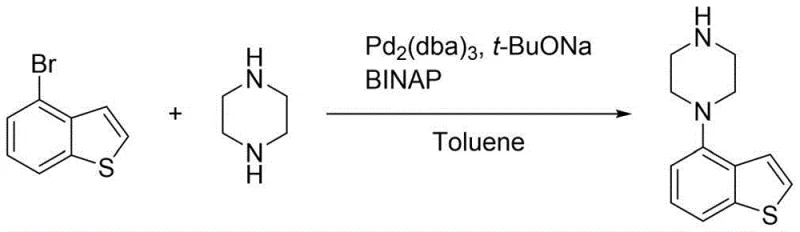
The Novel Approach
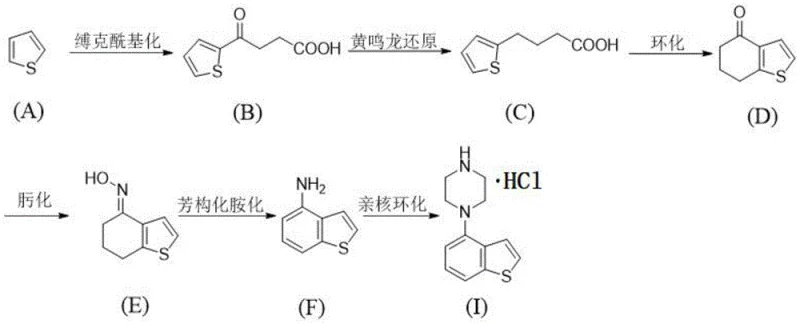
In stark contrast to these legacy methods, the novel synthetic strategy outlined in the patent utilizes thiophene, an abundant and inexpensive coal chemical byproduct, as the foundational building block. This route constructs the target molecule through a logical progression of acylation, reduction, cyclization, oximation, and finally, iodine-catalyzed aromatization. By replacing the palladium-mediated coupling with an iodine-source catalyzed aromatization amination, the process effectively circumvents the issues of heavy metal contamination and double-coupling side products. The use of readily available reagents such as succinic anhydride, hydrazine hydrate, and trifluoroacetic anhydride ensures that the supply chain remains resilient and cost-effective. This comprehensive redesign of the synthetic pathway not only lowers the barrier to entry for production but also aligns with green chemistry principles by valorizing waste thiophene, thereby transforming a low-value industrial byproduct into a high-value pharmaceutical intermediate with exceptional efficiency.
Mechanistic Insights into Iodine-Catalyzed Aromatization and Cyclization
The core technical breakthrough of this methodology lies in the strategic application of iodine-source catalysis during the aromatization amination step, which facilitates the conversion of the dihydrobenzothiophene ketoxime intermediate into the aromatic amine derivative. Unlike oxidative systems that might require harsh conditions or expensive oxidants, the iodine catalyst operates under relatively mild thermal conditions in solvents like ethylene glycol dimethyl ether. This mechanism likely involves the activation of the oxime functionality followed by an elimination-aromatization sequence that restores the aromaticity of the benzene ring fused to the thiophene moiety. The choice of iodine sources, ranging from elemental iodine to sodium iodide, provides flexibility in optimizing reaction kinetics and cost, allowing manufacturers to select the most economically viable variant without compromising reaction fidelity. This step is critical as it establishes the 4-amino substitution pattern required for the subsequent piperazine attachment, doing so with high regioselectivity and minimal formation of isomeric impurities that often plague electrophilic aromatic substitutions on heterocyclic systems.
Furthermore, the preceding cyclization step utilizing trifluoroacetic anhydride and trifluoroacetic acid demonstrates a highly efficient intramolecular Friedel-Crafts acylation mechanism. This transformation converts the linear 4-(thien-2-yl)butyric acid derivative into the fused 6,7-dihydrobenzo[b]thiophen-4(5H)-one system with remarkable precision. The strong acidic environment promotes the formation of a highly reactive acylium ion species, which subsequently attacks the electron-rich thiophene ring to close the six-membered carbocycle. This specific cyclization is advantageous because it proceeds under mild temperatures compared to traditional polyphosphoric acid mediated cyclizations, reducing energy consumption and minimizing thermal degradation of the sensitive thiophene ring. The combination of this robust cyclization with the subsequent oximation and aromatization steps creates a seamless flow of chemical transformations that maintain high atom economy and structural integrity throughout the synthesis, ensuring that the final product meets the stringent purity specifications required for active pharmaceutical ingredient manufacturing.
How to Synthesize 4-Piperazinyl Benzothiophene Hydrochloride Efficiently
The execution of this synthetic route requires precise control over reaction parameters to maximize yield and minimize impurity formation at each stage. The process begins with the acylation of thiophene, followed by a high-temperature reduction and a carefully managed cyclization. Detailed operational protocols regarding stoichiometry, temperature ramps, and workup procedures are essential for reproducibility. For a comprehensive breakdown of the standardized operating procedures, including specific solvent volumes, reaction times, and purification techniques for each intermediate, please refer to the technical guide below.
- Perform Friedel-Crafts acylation of thiophene with succinic anhydride using AlCl3 catalyst to form compound (B).
- Execute Huang Minlon reduction on compound (B) using hydrazine hydrate and KOH to yield compound (C).
- Cyclize compound (C) using trifluoroacetic anhydride and acid to construct the six-membered ring compound (D).
- React compound (D) with hydroxylamine hydrochloride to form the oxime intermediate compound (E).
- Conduct iodine-catalyzed aromatization amination on compound (E) to generate 4-aminobenzothiophene derivative (F).
- Complete the synthesis via nucleophilic substitution of compound (F) with bis(2-chloroethyl)amine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple chemical curiosity. The primary advantage stems from the fundamental shift in raw material sourcing; by anchoring the synthesis on thiophene, a commodity chemical with massive global production capacity, the supply chain becomes decoupled from the volatility of specialized halogenated benzothiophene markets. This ensures a consistent and reliable flow of materials, mitigating the risk of production stoppages due to precursor shortages. Additionally, the elimination of palladium catalysts removes a significant cost center, not only in terms of the catalyst purchase price but also in the downstream costs associated with metal scavenging and waste disposal. The simplified purification requirements further contribute to operational efficiency, reducing the time and resources needed to bring the product to market while maintaining the high quality standards demanded by regulatory bodies.
- Cost Reduction in Manufacturing: The economic impact of replacing noble metal catalysts with inexpensive iodine sources cannot be overstated in the context of large-scale production. Palladium catalysts represent a substantial portion of the bill of materials in traditional routes, and their removal necessitates expensive purification resins or activated carbon treatments to meet residual metal limits. By adopting this iodine-catalyzed pathway, manufacturers can achieve drastic reductions in direct material costs while simultaneously lowering the operational expenditure related to waste management and catalyst recovery. The use of common solvents and reagents like potassium carbonate and hydrazine hydrate further stabilizes the cost structure, making the final intermediate significantly more price-competitive in the global marketplace without sacrificing quality or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and geographic distribution of raw materials. Thiophene is produced in vast quantities as a byproduct of coal tar processing and petroleum refining, ensuring a diverse and robust supplier base that is not subject to the monopolies often seen with specialized pharmaceutical building blocks. This abundance translates directly into shorter lead times and greater flexibility in procurement planning. Furthermore, the synthetic route avoids the use of highly regulated or controlled substances that might trigger additional compliance hurdles, streamlining the logistics of international transport and storage. This reliability allows pharmaceutical companies to maintain leaner inventory levels while confidently meeting production schedules for the final drug product.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is designed for seamless translation from laboratory bench to multi-ton industrial reactors. The reaction conditions, such as the use of trifluoroacetic acid for cyclization and standard aqueous workups, are compatible with existing infrastructure in most fine chemical plants, requiring minimal capital investment for retrofitting. The avoidance of heavy metals aligns perfectly with increasingly stringent environmental regulations regarding effluent discharge and worker safety. By generating less hazardous waste and consuming fewer precious resources, this method supports corporate sustainability goals and reduces the environmental footprint of the manufacturing process, positioning the supplier as a responsible partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the new process. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this manufacturing route.
Q: Why is the iodine-catalyzed aromatization step superior to traditional palladium coupling?
A: Traditional methods often rely on expensive palladium catalysts for Buchwald-Hartwig coupling, which introduces heavy metal contamination risks and requires complex purification steps. The novel iodine-catalyzed approach eliminates noble metal usage, significantly reducing raw material costs and simplifying post-reaction processing while maintaining high purity standards suitable for pharmaceutical applications.
Q: What are the cost advantages of using thiophene as a starting material?
A: Thiophene is a low-cost, readily available raw material often derived from coal chemical processes, making it significantly more economical than specialized starting materials like 4-aminobenzothiophene or 4-bromobenzothiophene. Utilizing this abundant feedstock allows for substantial cost reduction in manufacturing and ensures a stable, continuous supply chain independent of niche precursor availability.
Q: How does this process address impurity control compared to prior art?
A: Conventional palladium-catalyzed routes are prone to generating double-coupling impurities that are difficult to remove. The new synthetic pathway avoids these specific side reactions by utilizing a distinct mechanistic route involving cyclization and aromatization, resulting in a cleaner reaction profile and higher overall yield without the need for extensive chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Piperazinyl Benzothiophene Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is smooth and efficient. We leverage our rigorous QC labs and stringent purity specifications to guarantee that every batch of 4-piperazinyl benzothiophene hydrochloride meets the exacting standards required for API synthesis. Our capability to implement the iodine-catalyzed route described in CN115894434A allows us to offer a product that is not only chemically superior but also economically optimized for our partners.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential financial improvements this route offers compared to your current sourcing strategy. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and security in your pharmaceutical manufacturing operations.
