Scalable Production of 5-Chloromethyl Salicylaldehyde via Quaternary Ammonium Catalysis for Global Markets
The chemical industry continuously seeks robust methodologies for synthesizing critical intermediates that serve as the backbone for advanced functional materials and pharmaceutical agents. Patent CN102180779A introduces a transformative preparation method for 5-chloromethyl salicylaldehyde (CMSA), a pivotal precursor in the synthesis of water-soluble, oil-soluble, and chiral Salen-type complexes used extensively in oxidation and olefin polymerization catalysis. This technical disclosure addresses long-standing inefficiencies in the chloromethylation of salicylaldehyde, specifically targeting the notorious agglomeration issues that plague traditional batch processes. By integrating a specific class of quaternary ammonium salts as phase-transfer catalysts and anti-agglomeration agents, the invention achieves a dramatic enhancement in both operational feasibility and final product yield. For R&D directors and procurement strategists evaluating reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in process reliability, offering a pathway to high-purity materials with simplified downstream processing requirements that directly impact the bottom line.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-chloromethyl salicylaldehyde has been hindered by severe physical processing challenges that limit its commercial viability and economic efficiency. Traditional protocols involving the reaction of salicylaldehyde with formaldehyde or paraformaldehyde in concentrated hydrochloric acid typically result in the formation of a viscous, sticky solid product. This undesirable physical state arises because the insoluble 5-chloromethyl salicylaldehyde precipitate tends to fuse with dispersed liquid droplets of unreacted salicylaldehyde, forming large, heterogeneous spherical agglomerates of varying diameters. These sticky masses encapsulate significant amounts of raw material, preventing full participation in the chloromethylation reaction and capping yields at approximately 70%. Furthermore, the resulting product cake is notoriously difficult to filter, wash, and dry, leading to extended batch cycles, high labor costs, and inconsistent purity profiles that complicate subsequent synthetic steps in ligand manufacturing.
The Novel Approach
The innovative methodology described in the patent overcomes these physical barriers through the strategic introduction of quaternary ammonium salts into the reaction matrix. These catalysts serve a dual function: acting as phase-transfer agents to enhance the solubility of organic reactants in the aqueous acid phase, and critically, adsorbing onto the surface of the nascent product crystals to prevent inter-particle fusion. This surface modification ensures that the 5-chloromethyl salicylaldehyde precipitates as a dry, fluffy powder with small particle size rather than a sticky mass. 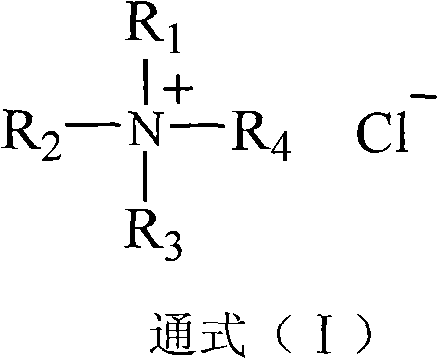 As illustrated by the general structure in the patent, where R groups are C1-C8 alkyl chains, these cationic surfactants effectively isolate the product particles from the liquid salicylaldehyde phase. This breakthrough not only boosts raw material conversion rates significantly but also transforms the workup procedure into a straightforward filtration and drying operation, drastically reducing processing time and facilitating cost reduction in fine chemical manufacturing.
As illustrated by the general structure in the patent, where R groups are C1-C8 alkyl chains, these cationic surfactants effectively isolate the product particles from the liquid salicylaldehyde phase. This breakthrough not only boosts raw material conversion rates significantly but also transforms the workup procedure into a straightforward filtration and drying operation, drastically reducing processing time and facilitating cost reduction in fine chemical manufacturing.
Mechanistic Insights into Quaternary Ammonium Catalyzed Chloromethylation
The core chemical transformation relies on the electrophilic aromatic substitution of salicylaldehyde, facilitated by the in situ generation of a chloromethyl carbocation species. In the acidic medium, formaldehyde reacts with hydrochloric acid to form the reactive hydroxymethyl cation intermediate, which subsequently attacks the electron-rich aromatic ring of salicylaldehyde at the position para to the hydroxyl group. 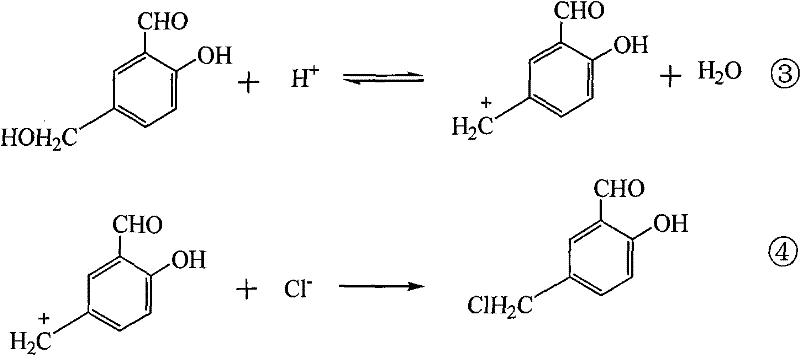 The mechanism involves the protonation of the hydroxymethyl group followed by the loss of water to generate the electrophilic methylene bridge, which is then trapped by a chloride ion to form the stable chloromethyl functionality. The presence of the quaternary ammonium salt accelerates this sequence by increasing the effective concentration of the organic substrate in the aqueous phase where the electrophile is generated, thereby increasing the collision frequency between reactants and driving the equilibrium toward the desired product with high regioselectivity.
The mechanism involves the protonation of the hydroxymethyl group followed by the loss of water to generate the electrophilic methylene bridge, which is then trapped by a chloride ion to form the stable chloromethyl functionality. The presence of the quaternary ammonium salt accelerates this sequence by increasing the effective concentration of the organic substrate in the aqueous phase where the electrophile is generated, thereby increasing the collision frequency between reactants and driving the equilibrium toward the desired product with high regioselectivity.
Beyond the electronic acceleration, the physical chemistry of crystallization plays a vital role in the success of this process. As the 5-chloromethyl salicylaldehyde product forms, it exceeds its solubility limit in the hydrochloric acid solution and begins to nucleate. Without the additive, these nuclei would rapidly grow and coalesce with the immiscible salicylaldehyde oil phase. However, the amphiphilic nature of the quaternary ammonium cation allows it to orient at the solid-liquid interface, creating a steric and electrostatic barrier that inhibits crystal growth into large agglomerates. This control over particle morphology ensures that the product remains free-flowing and easily separable from the mother liquor. For technical teams focused on high-purity pharmaceutical intermediates, this mechanism guarantees a product with minimal occlusion of impurities or starting materials, simplifying the purification train and ensuring consistent quality batches suitable for sensitive catalytic applications.
How to Synthesize 5-Chloromethyl Salicylaldehyde Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize the benefits of the quaternary ammonium catalyst. The process begins with the sequential charging of salicylaldehyde, a formaldehyde source (either aqueous solution or paraformaldehyde), and the selected quaternary ammonium salt into a reactor equipped with efficient agitation. Concentrated hydrochloric acid is added while maintaining the temperature between 5°C and 35°C to manage the exotherm and control the rate of electrophile generation. The mixture is then stirred continuously for an extended period, ranging from 40 to 200 hours depending on the specific formaldehyde source and catalyst loading, to ensure complete conversion. Detailed standardized operating procedures regarding specific stoichiometric ratios, temperature ramps, and workup protocols are essential for reproducibility.
- Charge salicylaldehyde, formaldehyde source, and quaternary ammonium catalyst into the reactor with concentrated hydrochloric acid under controlled low temperature.
- Maintain continuous stirring for an extended period (40-200 hours) to ensure complete conversion while preventing product agglomeration.
- Filter the dry, fluffy precipitate, wash with water and sodium bicarbonate solution, and vacuum dry to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this quaternary ammonium catalyzed process offers profound advantages over legacy manufacturing routes. The primary value driver is the substantial increase in yield, moving from a historical baseline of roughly 70% to over 93-96% as demonstrated in the patent examples. This efficiency gain directly translates to a lower cost of goods sold (COGS) by maximizing the throughput of raw materials such as salicylaldehyde and formaldehyde, which are commodity chemicals subject to market price fluctuations. Additionally, the elimination of sticky byproduct handling reduces equipment downtime for cleaning and maintenance, thereby increasing overall asset utilization rates and allowing for tighter production scheduling to meet urgent client demands without compromising quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the dual action of yield enhancement and waste minimization. By preventing the encapsulation of raw materials within sticky product masses, the process ensures that nearly all charged salicylaldehyde is converted into valuable product, significantly lowering the raw material cost per kilogram of output. Furthermore, the ability to recycle the reaction mother liquor, as shown in the patent examples, reduces the consumption of hydrochloric acid and minimizes the volume of wastewater requiring treatment. The absence of transition metal catalysts also eliminates the need for expensive and complex heavy metal scavenging steps, further streamlining the cost structure and making this a highly competitive option for cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Operational reliability is significantly bolstered by the physical characteristics of the product obtained through this method. The formation of a dry, fluffy powder instead of a viscous paste ensures that filtration and drying units operate at peak efficiency, preventing bottlenecks that often delay batch release in traditional processes. This predictability in cycle times allows supply chain managers to provide more accurate lead time estimates to downstream customers. Moreover, the robustness of the reaction conditions, which tolerate both aqueous formaldehyde and paraformaldehyde feeds, provides flexibility in raw material sourcing, mitigating risks associated with supply disruptions of specific formaldehyde grades and ensuring continuous production capability.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the simplicity of the unit operations involved, which rely on standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. The environmental footprint is favorably impacted by the reduction in three wastes (wastewater, waste gas, and solid waste) due to higher atom economy and mother liquor recycling. The process avoids the use of hazardous chloromethyl methyl ether (CMME), a known carcinogen often used in alternative chloromethylation routes, thereby enhancing workplace safety and simplifying regulatory compliance for commercial scale-up of complex fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-chloromethyl salicylaldehyde using this advanced catalytic system. These insights are derived directly from the experimental data and mechanistic explanations provided in the patent literature, aiming to clarify the operational benefits for potential partners. Understanding these nuances is critical for evaluating the feasibility of integrating this intermediate into your specific supply chain or R&D pipeline.
Q: How does the quaternary ammonium salt improve the physical properties of the product?
A: The quaternary ammonium salt adsorbs onto the surface of the precipitating 5-chloromethyl salicylaldehyde crystals. This adsorption layer prevents the product particles from fusing with unreacted salicylaldehyde droplets, thereby eliminating the formation of sticky, large spherical agglomerates that are difficult to filter and dry.
Q: What is the typical yield improvement compared to conventional methods?
A: Conventional methods often suffer from yields around 70% due to reactant encapsulation within sticky product masses. The patented quaternary ammonium catalyzed process significantly enhances raw material utilization, achieving yields consistently above 93% and up to 96% in optimized examples, representing a substantial increase in process efficiency.
Q: Can the reaction mother liquor be recycled to reduce waste?
A: Yes, the process allows for the recycling of the reaction mother liquor after filtration of the crude product. Examples in the patent demonstrate that reusing the mother liquor with fresh feedstocks maintains high reaction efficiency and yield, contributing to reduced waste generation and lower overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloromethyl Salicylaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation catalysts and pharmaceutical agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of innovative processes like the quaternary ammonium catalyzed chloromethylation are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-chloromethyl salicylaldehyde meets the exacting standards required for Salen ligand synthesis, providing our clients with a foundation of reliability and consistency for their own manufacturing operations.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this superior manufacturing method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and cost-effective chemical strategy available in the market.
