Advanced Supramolecular Separation of Biphenylcarboxylic Acid Isomers for Commercial Scale-Up
The pharmaceutical and fine chemical industries constantly face the challenge of separating structural isomers, particularly when dealing with aromatic carboxylic acids like biphenylcarboxylic acid. Patent CN101891609A introduces a groundbreaking inclusion separation method that leverages the principles of supramolecular chemistry to effectively isolate 3-biphenylcarboxylic acid and 4-biphenylcarboxylic acid. This technology addresses a critical gap in the market, as traditional separation techniques often struggle with the similar physicochemical properties of these meta- and para-isomers. By utilizing β-cyclodextrin as a host molecule, the process achieves a purity of over 99% for the valuable 4-isomer, which is essential for applications in liquid crystal materials and advanced organic synthesis. This report analyzes the technical feasibility and commercial potential of this novel approach for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of biphenylcarboxylic acid isomers has been a cumbersome and energy-intensive process, often relying on fractional crystallization or complex chromatographic techniques. These conventional methods are frequently plagued by low selectivity due to the minimal differences in boiling points and solubility between the 3- and 4-isomers. For procurement managers, this translates into high operational costs, significant solvent consumption, and extended processing times that delay time-to-market for downstream products. Furthermore, traditional distillation methods require high vacuum conditions and precise temperature control, increasing the risk of thermal degradation and reducing overall yield. The inability to efficiently separate these isomers from waste streams, such as those generated during toluene oxidation for benzoic acid production, represents a significant loss of potential value and creates environmental disposal challenges.
The Novel Approach
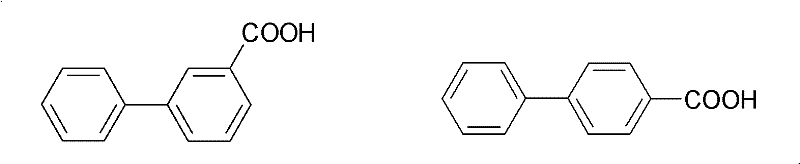
The patented inclusion separation method offers a paradigm shift by utilizing molecular recognition rather than relying solely on physical property differences. This approach employs β-cyclodextrin, a cyclic oligosaccharide with a unique barrel-shaped structure containing a hydrophobic internal cavity. The process selectively captures the 4-biphenylcarboxylic acid isomer because its linear molecular geometry allows it to enter and stabilize within the cyclodextrin cavity, forming a solid inclusion complex. In contrast, the 3-biphenylcarboxylic acid isomer, with its bent structure, is sterically hindered from entering the cavity and remains in the mother liquor. This mechanism drastically simplifies the separation workflow, eliminating the need for repeated recrystallizations or expensive chromatography columns, thereby offering a robust solution for cost reduction in fine chemical manufacturing.
Mechanistic Insights into β-Cyclodextrin Inclusion Separation
The core of this technology lies in the host-guest chemistry between β-cyclodextrin and the biphenylcarboxylic acid isomers. β-Cyclodextrin consists of seven glucopyranose units linked by α-1,4-glycosidic bonds, forming a toroidal shape with a hydrophilic exterior and a hydrophobic interior. The separation efficiency is dictated by the geometric complementarity between the guest molecule and the host cavity. The 4-biphenylcarboxylic acid molecule possesses a linear conformation that aligns perfectly with the cylindrical channel of the β-cyclodextrin, facilitating strong van der Waals interactions and hydrogen bonding. Conversely, the carboxyl group at the meta-position in 3-biphenylcarboxylic acid creates a kink in the molecular structure, preventing effective inclusion. This steric exclusion is the fundamental driver of the high selectivity observed in the process.
Following the formation of the inclusion complex, the separation is finalized through a controlled resolution step. The solid inclusion complex is isolated via filtration and washed to remove any adhering mother liquor containing the unwanted 3-isomer. To recover the pure product, the complex is dissolved in ethanol and heated to reflux at temperatures between 88°C and 90°C. At this elevated temperature, the stability of the host-guest interaction decreases, causing the release of the 4-biphenylcarboxylic acid into the solution while the β-cyclodextrin remains soluble or can be separated. This thermal dissociation mechanism ensures that the final product is free from the host molecule, yielding high-purity crystals upon subsequent cooling and recrystallization. This precise control over the association and dissociation phases is critical for maintaining the stringent purity specifications required by R&D directors in the electronic and pharmaceutical sectors.
How to Synthesize 4-Biphenylcarboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this separation technology on a pilot or commercial scale. The process begins with the preparation of a saturated aqueous solution of β-cyclodextrin, maintained at a constant temperature between 60°C and 70°C to ensure maximum solubility. An ethanolic solution of the crude biphenylcarboxylic acid mixture is then added dropwise, maintaining a specific molar ratio of substrate to host between 1:1.5 and 1:2.5 to drive the equilibrium towards complex formation. Detailed standardized synthesis steps follow below.
- Prepare a saturated aqueous solution of β-cyclodextrin at 60-70°C and mix with an ethanolic solution of the biphenylcarboxylic acid isomer mixture at a molar ratio of 1: 1.5 to 1:2.5.
- Stir the mixture for 6-8 hours, then allow it to cool and stand until the inclusion complex precipitates, followed by washing with distilled water and ethanol.
- Dissolve the inclusion complex in ethanol, heat to reflux at 88-90°C for 2-3 hours to release the 4-biphenylcarboxylic acid, then distill and recrystallize.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this inclusion separation technology presents a compelling value proposition centered on waste valorization and operational efficiency. By transforming low-value still waste from benzoic acid production into high-purity intermediates, manufacturers can significantly reduce raw material acquisition costs. The process eliminates the dependency on expensive, high-grade starting materials, as it effectively upgrades waste streams into premium products. Furthermore, the simplicity of the operation—primarily involving mixing, filtration, and standard distillation—reduces the need for specialized equipment and highly skilled labor, leading to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The elimination of energy-intensive fractional distillation and multiple recrystallization steps directly lowers utility consumption and processing time. Since the separation relies on selective precipitation rather than volatility differences, the process operates at atmospheric pressure and moderate temperatures, reducing the capital expenditure associated with high-vacuum systems. Additionally, the recovery of β-cyclodextrin from the mother liquor allows for potential recycling of the host molecule, further driving down the variable cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: Sourcing high-purity 4-biphenylcarboxylic acid from traditional suppliers can be volatile due to the complexity of synthesis. This technology enables in-house production or reliable contracting from suppliers who utilize waste-derived feedstocks, ensuring a more stable and continuous supply. The robustness of the inclusion method against variations in feedstock composition means that supply interruptions due to raw material quality issues are minimized, providing greater security for long-term production planning.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton production without significant re-engineering. From an environmental perspective, converting hazardous chemical waste into valuable commodities aligns with green chemistry principles and circular economy goals. This reduces the environmental footprint of the facility and mitigates regulatory risks associated with waste disposal, making the supply chain more resilient to tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this separation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy for decision-makers evaluating this route.
Q: What is the primary advantage of using β-cyclodextrin for separating biphenylcarboxylic acid isomers?
A: The primary advantage lies in molecular recognition; the linear structure of 4-biphenylcarboxylic acid fits precisely into the hydrophobic cavity of β-cyclodextrin, allowing for highly selective separation from the meta-isomer which does not fit well.
Q: What purity levels can be achieved with this inclusion separation method?
A: According to patent data, this method can achieve a purity of greater than 99% for 4-biphenylcarboxylic acid, making it suitable for high-end applications like liquid crystal materials.
Q: Is this process suitable for large-scale industrial waste recovery?
A: Yes, the process is specifically designed to recover valuable isomers from benzoic acid still waste liquid, offering a circular economy solution that transforms low-value waste into high-purity chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Biphenylcarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-purity intermediates like 4-biphenylcarboxylic acid in the development of next-generation liquid crystal materials and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global R&D teams, delivering consistency that your projects depend on.
We invite you to collaborate with us to leverage this advanced separation technology for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our supply capabilities can enhance your competitive advantage in the market.
