Advanced Photocatalytic Synthesis of 1,1-Disubstituted Cyclopropanes for Commercial Scale-Up
Advanced Photocatalytic Synthesis of 1,1-Disubstituted Cyclopropanes for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to construct complex molecular architectures, particularly strained ring systems like cyclopropanes which are prevalent in bioactive molecules. Patent CN110041361B introduces a groundbreaking methodology for the synthesis of 1,1-disubstituted cyclopropanes through a novel photocatalytic allylation and cyclopropanation tandem reaction strategy. This technology leverages visible light catalysis to drive the transformation under exceptionally mild conditions, utilizing an iridium-based photocatalyst to mediate the radical generation and subsequent cyclization events. By shifting away from traditional thermal activation methods, this process not only enhances energy efficiency but also significantly improves the functional group tolerance, allowing for the synthesis of diverse intermediates that were previously difficult to access. For R&D directors and process chemists, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity, offering a robust platform for developing next-generation active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for constructing cyclopropane rings, such as the Simmons-Smith reaction or transition metal-catalyzed carbene transfers, often suffer from significant operational and economic drawbacks that hinder their widespread adoption in large-scale manufacturing. These conventional routes frequently require the use of stoichiometric amounts of hazardous reagents, such as diethylzinc or diazo compounds, which pose severe safety risks regarding thermal instability and toxicity during handling and storage. Furthermore, the reaction conditions are typically harsh, necessitating cryogenic temperatures or elevated heating that increases energy consumption and complicates process control in a production environment. The presence of heavy metal residues from catalysts like copper or rhodium also mandates rigorous and costly downstream purification steps to meet the stringent regulatory limits for elemental impurities in drug substances. Consequently, procurement managers often face inflated costs and extended lead times due to the specialized equipment and safety protocols required to manage these high-risk chemical transformations effectively.
The Novel Approach
In stark contrast, the technology disclosed in CN110041361B employs a visible-light driven photocatalytic system that operates at room temperature, thereby eliminating the need for extreme thermal inputs and reducing the overall energy footprint of the synthesis. This innovative approach utilizes stable halomethyl bis(catechol) silicates as radical precursors, which are safer and easier to handle than traditional diazo or organometallic reagents, significantly enhancing workplace safety and supply chain reliability. The tandem nature of the reaction allows for the direct conversion of allyl halides into cyclopropanes in a single pot, streamlining the synthetic route and minimizing the number of isolation steps required. By avoiding the use of toxic heavy metals in stoichiometric quantities, this method inherently reduces the burden on waste treatment facilities and lowers the cost associated with metal scavenging processes. This paradigm shift offers a compelling value proposition for supply chain heads looking to de-risk their manufacturing operations while maintaining high standards of product quality and consistency.
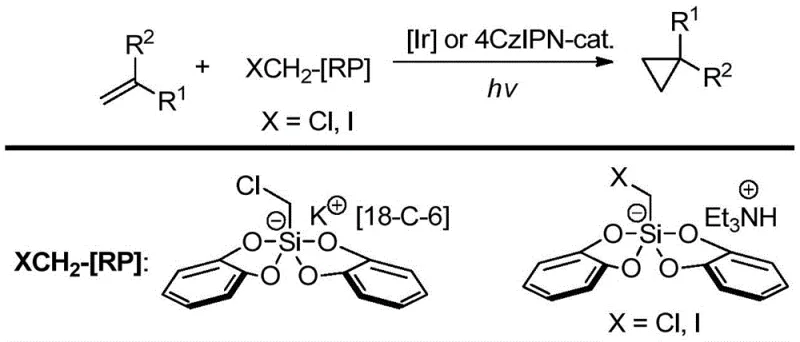
Mechanistic Insights into Photocatalytic Allylation/Cyclopropanation Tandem Reaction
The core of this technological advancement lies in the sophisticated interplay between the iridium photocatalyst and the silicate radical precursors under visible light irradiation, which facilitates a precise radical addition-cyclization sequence. Upon excitation by blue LEDs, the iridium complex enters a reactive state capable of engaging in single-electron transfer processes with the halomethyl silicate species, generating the crucial carbon-centered radicals needed for bond formation. These radicals then undergo a highly selective addition to the electron-deficient olefinic bond of the allyl halide substrate, initiating the cascade that ultimately leads to the formation of the strained three-membered ring. The mechanistic pathway is designed to minimize competing side reactions, such as polymerization or non-specific radical coupling, ensuring that the majority of the starting material is channeled towards the desired cyclopropane product. For technical teams, understanding this mechanism is vital as it highlights the importance of light intensity and catalyst loading in maintaining the efficiency of the radical chain propagation throughout the reaction duration.
Furthermore, the choice of dimethyl sulfoxide (DMSO) as the solvent plays a critical role in stabilizing the charged intermediates and facilitating the solubility of both the organic substrates and the ionic silicate reagents. The reaction environment is maintained under an inert nitrogen atmosphere to prevent the quenching of radical species by oxygen, which could otherwise lead to oxidation byproducts and reduced yields. The compatibility of this system with various functional groups, including phosphonates and esters, demonstrates the robustness of the catalytic cycle against potential deactivation pathways. This level of control over the reaction trajectory allows for the synthesis of complex intermediates with high stereochemical integrity, which is essential for ensuring the biological activity of the final pharmaceutical compound. The detailed elucidation of this mechanism provides a solid foundation for process optimization and scale-up activities in a commercial setting.
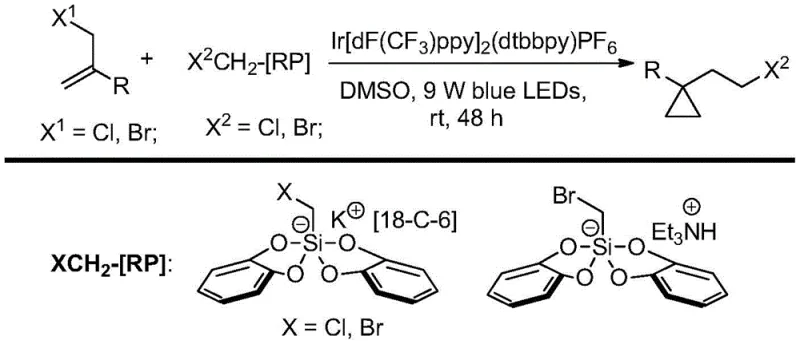
How to Synthesize 1,1-Disubstituted Cyclopropane Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction mixture and the maintenance of optimal illumination conditions to ensure consistent results across different batches. The process begins with the precise weighing of the allyl halide substrate and the iridium photocatalyst, followed by the addition of the halomethyl bis(catechol) silicate reagent in a reaction vessel equipped with efficient stirring capabilities. It is imperative to degas the dimethyl sulfoxide solvent prior to injection to remove dissolved oxygen, which acts as a radical scavenger and can inhibit the catalytic cycle if present in significant quantities. Once the reagents are combined under nitrogen protection, the reaction vessel is exposed to 9W blue LED light sources, and the mixture is stirred continuously for a period ranging from 45 to 50 hours to allow for complete conversion. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined in the guide below to assist technical teams in replicating this high-efficiency protocol.
- Charge a reaction vessel with allyl halide, iridium photocatalyst, and halomethyl bis(catechol) silicate under nitrogen protection.
- Inject degassed dimethyl sulfoxide (DMSO) and stir the mixture under 9W blue LED illumination for 45 to 50 hours at room temperature.
- Quench the reaction with ethyl acetate and saturated brine, extract the organic phase, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this photocatalytic technology offers substantial strategic benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. By transitioning to a method that relies on abundant and stable starting materials rather than scarce or hazardous reagents, companies can mitigate the risks associated with supply chain disruptions and volatile raw material pricing. The elimination of expensive transition metal catalysts and the reduction in purification complexity translate into significant cost savings in terms of both material consumption and labor hours required for production. Additionally, the mild reaction conditions reduce the wear and tear on manufacturing equipment, extending the lifespan of reactors and lowering capital expenditure requirements for facility maintenance. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding rapidly to market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The process drastically simplifies the production workflow by removing the need for costly metal scavenging resins and extensive washing procedures typically required to remove heavy metal residues from the final product. This reduction in downstream processing steps leads to a decrease in solvent usage and waste generation, aligning with green chemistry principles while simultaneously lowering disposal costs. Furthermore, the use of commercially available iridium catalysts in low loading amounts ensures that the catalyst cost per kilogram of product remains minimal compared to traditional stoichiometric methods. The overall economic efficiency is further enhanced by the high atom economy of the tandem reaction, which maximizes the incorporation of starting materials into the final structure, reducing material loss and improving yield metrics.
- Enhanced Supply Chain Reliability: Utilizing stable silicate reagents and allyl halides ensures a consistent supply of high-quality raw materials that are less susceptible to degradation during storage and transportation compared to sensitive organometallic reagents. This stability allows for larger inventory buffers and reduces the frequency of urgent replenishment orders, thereby smoothing out procurement cycles and improving planning accuracy. The robustness of the reaction against minor variations in conditions also means that production schedules are less likely to be disrupted by batch failures or quality deviations. For supply chain heads, this reliability translates into greater confidence in meeting delivery commitments to downstream pharmaceutical clients and maintaining a steady flow of intermediates.
- Scalability and Environmental Compliance: The scalability of this photocatalytic process is supported by the availability of industrial-grade LED lighting systems that can be easily integrated into existing reactor setups without major infrastructure overhauls. The absence of high-pressure or high-temperature requirements simplifies the safety validation process for new production lines, accelerating the timeline from pilot scale to commercial manufacturing. Moreover, the reduced generation of hazardous waste and the avoidance of toxic reagents facilitate compliance with increasingly stringent environmental regulations, minimizing the risk of regulatory penalties. This environmentally friendly profile enhances the corporate sustainability image and meets the growing demand from global partners for responsibly sourced chemical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method, providing clarity on its practical application and benefits. These insights are derived directly from the patent data and are intended to assist decision-makers in evaluating the feasibility of adopting this technology for their specific production needs. Understanding these key aspects will help stakeholders appreciate the transformative potential of visible-light catalysis in modern chemical manufacturing and its alignment with industry trends towards sustainability and efficiency.
Q: What are the primary advantages of this photocatalytic method over traditional cyclopropanation?
A: This method utilizes visible light catalysis to drive the reaction under mild conditions, eliminating the need for harsh thermal conditions or stoichiometric toxic metal reagents often required in conventional Simmons-Smith or metal-carbenoid approaches.
Q: How does the use of silicate reagents impact impurity profiles?
A: The use of halomethyl bis(catechol) silicates as radical precursors ensures high functional group compatibility and minimizes side reactions, resulting in a cleaner crude reaction mixture that simplifies downstream purification processes.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the reaction operates at room temperature with simple LED lighting setups and avoids hazardous high-pressure or high-temperature conditions, making it inherently safer and more adaptable for commercial scale-up in GMP environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Disubstituted Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into reliable commercial reality, and we possess the technical expertise to scale this photocatalytic pathway for your specific needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial application is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,1-disubstituted cyclopropane meets the highest international standards for pharmaceutical intermediates. Our commitment to quality and consistency makes us the ideal partner for companies seeking to secure a stable supply of high-value chemical building blocks for their drug development pipelines.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your manufacturing costs and enhance your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver superior value and support your long-term strategic goals in the competitive global market.
