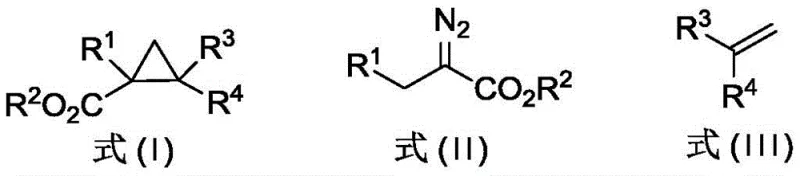
Advanced Photocatalytic Synthesis of Polysubstituted Cyclopropanes for Commercial API Manufacturing
The landscape of organic synthesis for complex cyclic structures is undergoing a significant transformation, driven by the urgent need for greener, more efficient methodologies in the production of high-value pharmaceutical intermediates. Patent CN113292434A, published in August 2021, introduces a groundbreaking approach to the cyclopropanation of non-aryl diazo compounds and olefins utilizing visible light photocatalysis. This technology represents a paradigm shift away from harsh thermal conditions and expensive noble metal catalysts, offering a robust pathway to polysubstituted cyclopropanes. For R&D directors and process chemists, the ability to bypass the notorious 1,2-hydrogen transfer side reactions that plague traditional diazo chemistry is a critical advancement. By leveraging specific Iridium (Ir) photosensitizers under blue LED irradiation, this method achieves high yields and exceptional selectivity, directly addressing the purity challenges inherent in API intermediate manufacturing. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative synthetic routes is essential for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropane rings has relied heavily on transition metal catalysis involving Rhodium (Rh), Ruthenium (Ru), or Copper (Cu) complexes. While effective for aromatic diazo compounds, these traditional methods face severe limitations when applied to non-aromatic substrates, particularly alpha-diazo compounds bearing substituents at the beta-position. Under these conventional conditions, the electron-deficient carbenes generated are highly prone to undergoing 1,2-hydrogen transfer reactions. This competing pathway leads to the formation of unsaturated carbonyl compounds as major by-products, drastically reducing the yield of the desired cyclopropane and complicating downstream purification. Furthermore, the use of stoichiometric or high-loading noble metals increases the cost of goods sold (COGS) and introduces significant environmental burdens related to heavy metal waste disposal. For procurement managers, the volatility in the supply and price of Rh and Ru poses a continuous risk to production stability and cost reduction in API manufacturing.
The Novel Approach
The methodology disclosed in the patent offers a sophisticated solution by replacing thermal activation with visible light photocatalysis. By employing an Iridium photosensitizer and irradiating the reaction mixture with blue LED light (wavelength 390-456 nm), the system generates reactive intermediates through a fundamentally different mechanistic pathway. This photo-induced process effectively suppresses the 1,2-hydrogen transfer side reaction, allowing for the exclusive formation of the cyclopropane ring even with challenging non-aromatic diazo substrates. The reaction proceeds under remarkably mild conditions, typically between 25-60°C, eliminating the need for energy-intensive heating or cryogenic cooling. This not only simplifies the operational protocol but also enhances the safety profile of the process, making it highly attractive for the commercial scale-up of complex pharmaceutical intermediates. The result is a cleaner reaction profile with fewer impurities, directly translating to reduced purification costs and higher overall process efficiency.
Mechanistic Insights into Ir-Photocatalyzed Cyclopropanation
The core of this technological breakthrough lies in the unique photophysical properties of the Iridium complexes utilized as catalysts. Upon absorption of blue light photons, the Ir photosensitizer transitions to an excited state capable of interacting with the diazo compound to facilitate nitrogen extrusion and carbene formation without the instability associated with thermal metal-carbene species. The specific ligand environment of the catalysts, ranging from Formula (IV) to Formula (IX), plays a pivotal role in tuning the redox potential and steric bulk, thereby controlling the stereoselectivity of the cycloaddition. For instance, catalysts bearing bulky tert-butyl groups or electron-withdrawing trifluoromethyl substituents can significantly influence the diastereomeric ratio (d.r.) of the product. This level of control is crucial for R&D teams aiming to synthesize specific stereoisomers required for biological activity in drug candidates.
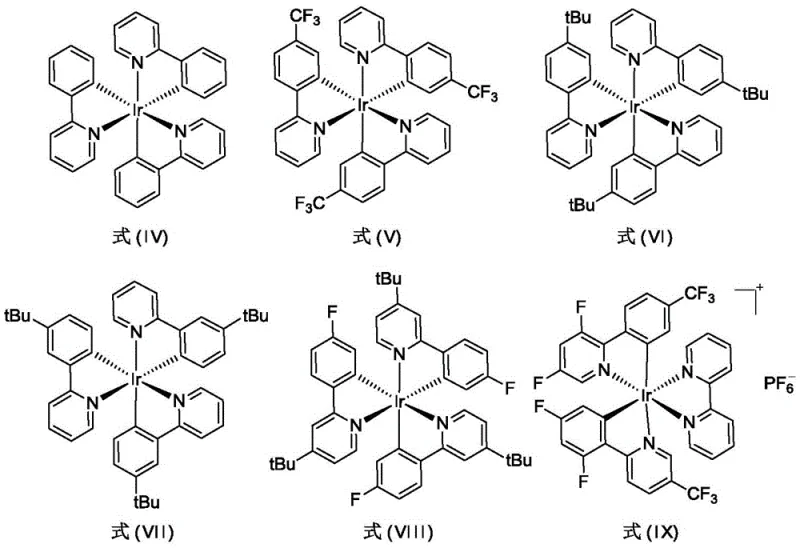
Furthermore, the mechanism inherently minimizes the generation of hazardous by-products. In traditional Lewis acid or transition metal catalysis, the carbene intermediate is often too reactive, leading to polymerization or rearrangement. The photocatalytic cycle moderates this reactivity, ensuring that the carbene reacts preferentially with the olefin dipolarophile. This selectivity is evident in the high yields reported across various substrates, including those with heterocyclic and ester functionalities. From an impurity control perspective, this means the crude reaction mixture contains significantly fewer structurally related impurities, simplifying the chromatographic separation steps. For quality assurance teams, this translates to a more consistent impurity profile and easier validation of the cleaning processes in multi-purpose manufacturing facilities.
How to Synthesize Polysubstituted Cyclopropanes Efficiently
The practical implementation of this photocatalytic method is straightforward and aligns well with existing infrastructure in fine chemical plants. The general procedure involves charging a reaction vessel with the alpha-diazoate compound and a substantial excess of the olefin (typically 500 mol% relative to the diazo compound) to drive the equilibrium forward. The Iridium catalyst is added in low molar quantities (0.01 to 2 mol%), and the mixture is stirred under an inert nitrogen atmosphere. Irradiation with standard blue LED lamps provides the necessary energy input, maintaining the reaction temperature between 25°C and 60°C for a duration of 12 to 36 hours. Following the reaction, the solvent is removed, and the product is isolated via standard column chromatography. Detailed standardized synthetic steps for specific derivatives are provided in the guide below.
- Charge a reaction flask with the alpha-diazoate compound (100 mmol), the olefin substrate (500 mmol), and the Iridium photosensitizer catalyst (1 mmol) in dichloromethane (500 mL).
- Maintain the reaction mixture under a nitrogen atmosphere and irradiate with a blue LED lamp (wavelength 390-456 nm) while stirring at 25-60°C for 12 to 36 hours.
- Upon completion, remove the solvent via rotary evaporation and purify the crude residue using column chromatography with petroleum ether and ethyl acetate to isolate the target cyclopropane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers tangible strategic benefits beyond mere chemical elegance. The shift from thermal to photochemical activation fundamentally alters the cost structure and risk profile of producing cyclopropane-containing intermediates. By eliminating the reliance on scarce and expensive noble metals like Rhodium, the process significantly reduces raw material costs and mitigates supply chain vulnerabilities associated with geopolitical instability in metal mining regions. Additionally, the mild reaction conditions reduce energy consumption and equipment stress, leading to lower operational expenditures (OPEX) over the lifecycle of the production campaign.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive transition metal catalysts with more abundant Iridium photosensitizers used at very low loadings. Traditional methods often require high catalyst loading to overcome deactivation, whereas this photocatalytic system maintains high turnover numbers. Furthermore, the suppression of the 1,2-hydrogen transfer by-product means that less starting material is wasted on side reactions, improving the overall atom economy. The simplified workup procedure, which avoids complex quenching steps required for reactive metal species, further reduces labor and waste disposal costs, contributing to substantial overall savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity to downstream API manufacturers. The use of commercially available olefins and diazo precursors, combined with a catalyst system that is stable under ambient storage conditions, minimizes the risk of production delays due to reagent degradation. Moreover, the scalability of LED photoreactors has improved dramatically in recent years, allowing for seamless translation from laboratory gram-scale to multi-kilogram production without the need for specialized high-pressure or high-temperature equipment, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles. The elimination of heavy metal waste streams simplifies effluent treatment and reduces the regulatory burden associated with metal residue limits in final drug substances. The energy efficiency of LED light sources compared to thermal heating or UV lamps lowers the carbon footprint of the synthesis. These factors make the process highly scalable and compliant with increasingly stringent environmental regulations, ensuring long-term viability for commercial production facilities aiming for sustainable operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic cyclopropanation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their pipeline.
Q: How does this photocatalytic method improve upon traditional transition metal catalysis?
A: Traditional methods using Rh, Ru, or Cu often suffer from 1,2-hydrogen transfer side reactions, especially with non-aromatic diazo compounds. This Ir-photocatalytic method suppresses those pathways, allowing for the direct synthesis of polysubstituted cyclopropanes with higher selectivity and yield.
Q: What are the specific reaction conditions required for scale-up?
A: The process operates under mild conditions, specifically between 25-60°C, using standard blue LED light sources (456 nm preferred). The reaction times range from 12 to 36 hours, making it compatible with standard batch reactors used in fine chemical manufacturing.
Q: Is this method suitable for generating quaternary carbon centers?
A: Yes, the methodology is particularly effective for constructing polysubstituted cyclopropane compounds containing quaternary carbon structures, which are often challenging to access via conventional thermal or Lewis acid-catalyzed routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Cyclopropanes Supplier
The technological potential of visible-light mediated cyclopropanation is immense, offering a pathway to complex scaffolds that were previously difficult or uneconomical to access. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations in driving the next generation of pharmaceutical discoveries. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with advanced photoreactor capabilities and rigorous QC labs to ensure that every batch meets stringent purity specifications required for clinical and commercial applications. We are committed to bridging the gap between academic innovation and industrial reality.
We invite potential partners to engage with our technical team to explore how this specific photocatalytic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this greener methodology for your specific targets. We encourage you to contact our technical procurement team today to discuss specific COA data, route feasibility assessments, and how we can support your project timelines with reliable, high-quality supply.
