Scalable Production of High-Purity Epoxysuccinic Acid via Green Molecular Sieve Catalysis
Introduction to Advanced Epoxysuccinic Acid Manufacturing
The chemical industry is currently witnessing a paradigm shift towards greener synthesis routes, particularly for high-volume intermediates like epoxysuccinic acid (ESA). Patent CN112521348B introduces a groundbreaking methodology that replaces traditional heavy metal catalysts with efficient molecular sieves, specifically targeting the epoxidation of maleic anhydride. This innovation addresses long-standing challenges regarding heavy metal contamination and catalyst recovery that have plagued the production of ESA, a critical component in non-phosphorus water treatment agents and pharmaceutical chelating formulations. By utilizing a titanium silicalite molecular sieve under controlled negative pressure, the process achieves exceptional selectivity while maintaining mild reaction conditions around 60°C. This technical advancement not only aligns with stringent global environmental regulations but also offers a robust pathway for manufacturers seeking to optimize their supply chains for high-purity fine chemicals. The transition from soluble tungsten salts to heterogeneous solid catalysts represents a significant leap in process safety and product quality assurance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of epoxysuccinic acid has relied heavily on homogeneous catalytic systems utilizing sodium tungstate or ammonium molybdate. As illustrated in the reaction mechanism below, these traditional pathways involve the formation of peroxo-tungsten or peroxo-molybdenum complexes that facilitate oxygen transfer but introduce severe downstream processing burdens. 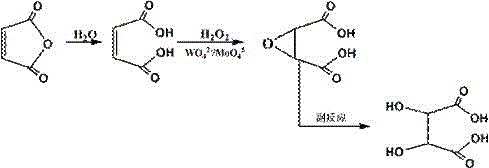 The primary drawback of these soluble salt catalysts is their inability to be easily separated from the reaction mixture, often necessitating complex ion-exchange resin purification steps to meet heavy metal specifications. Furthermore, the exothermic nature of the epoxidation reaction, if not meticulously managed, leads to the hydrolysis of the desired epoxide into tartaric acid, drastically reducing yield and complicating purification. The accumulation of tungsten or molybdenum residues in the final product poses significant toxicity risks, limiting the application of ESA in sensitive sectors such as food additives or high-grade pharmaceutical intermediates where trace metal limits are strictly enforced by regulatory bodies.
The primary drawback of these soluble salt catalysts is their inability to be easily separated from the reaction mixture, often necessitating complex ion-exchange resin purification steps to meet heavy metal specifications. Furthermore, the exothermic nature of the epoxidation reaction, if not meticulously managed, leads to the hydrolysis of the desired epoxide into tartaric acid, drastically reducing yield and complicating purification. The accumulation of tungsten or molybdenum residues in the final product poses significant toxicity risks, limiting the application of ESA in sensitive sectors such as food additives or high-grade pharmaceutical intermediates where trace metal limits are strictly enforced by regulatory bodies.
The Novel Approach
The novel approach detailed in the patent data fundamentally reengineers the catalytic landscape by employing a heterogeneous titanium silicalite molecular sieve with a particle size distribution of 200-500 nm. This solid catalyst operates via a distinct mechanism where the tetrahedral titanium active sites within the zeolite framework activate hydrogen peroxide for selective oxygen transfer without leaching into the solution. A critical innovation in this process is the implementation of negative pressure conditions, ranging from -0.045 to -0.065 MPa, which actively removes water vapor and excess heat from the reaction vessel. This dynamic control prevents local hot spots that typically trigger the unwanted hydrolysis of the epoxide ring. Additionally, the strategy of split-adding the catalyst—introducing a portion at the start and the remainder after peroxide addition—optimizes the surface area availability and maintains consistent catalytic activity throughout the reaction cycle. This results in a streamlined workflow where the catalyst is simply filtered off post-reaction, eliminating the need for energy-intensive ion exchange or complex extraction procedures.
Mechanistic Insights into Titanium Silicalite-Catalyzed Epoxidation
The efficacy of the titanium silicalite catalyst lies in its unique crystalline structure, which mimics the active sites found in enzymatic oxidation processes. The tetrahedrally coordinated titanium atoms embedded within the silicate lattice act as Lewis acid centers that coordinate with hydrogen peroxide to form a reactive hydroperoxo-titanium species. This intermediate is highly electrophilic and selectively attacks the electron-rich double bond of the maleic anhydride derivative, facilitating the formation of the three-membered epoxide ring with high stereochemical fidelity. Unlike homogeneous systems where the catalyst is freely diffusing and prone to deactivation via dimerization, the rigid framework of the molecular sieve isolates the active sites, preventing mutual deactivation and ensuring prolonged catalyst life. The pore structure of the sieve, tailored to the molecular dimensions of the reactants, further enhances selectivity by sterically hindering the approach of larger molecules that might lead to polymerization or other side reactions, thus preserving the integrity of the epoxysuccinic acid backbone.
Impurity control is rigorously managed through the precise regulation of pH and temperature, which are interdependent variables in this exothermic system. The patent specifies maintaining a pH range of 4.4 to 5.0 through the controlled滴加 (dropwise addition) of sodium hydroxide, which is critical because acidic conditions favor the hydrolysis of the epoxide to tartaric acid, while overly alkaline conditions can lead to ring-opening polymerization. The negative pressure environment plays a dual role here: it lowers the boiling point of water, allowing for evaporative cooling that counteracts the heat of reaction, and it shifts the equilibrium by removing water, a byproduct of the condensation steps involved in the catalytic cycle. This synergistic control ensures that the concentration of the active oxidant remains optimal while minimizing the residence time of the product in potentially degrading conditions, resulting in tartaric acid levels as low as 0.62% in optimized examples, a significant improvement over conventional batch processes.
How to Synthesize Epoxysuccinic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production, emphasizing the importance of sequential addition and thermal management. The process begins with the formation of a homogeneous maleic acid solution by hydrating maleic anhydride at moderate temperatures, followed by the strategic introduction of the solid catalyst.
- Mix maleic anhydride and water at 55-60°C to form a homogeneous liquid phase, then add a portion of the molecular sieve catalyst.
- Maintain the system at 60±5°C under negative pressure while simultaneously dropwise adding hydrogen peroxide and sodium hydroxide solutions.
- After peroxide addition, add the remaining catalyst, adjust pH to 4.4-5.0, maintain temperature for 1-2 hours, then cool and filter to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this molecular sieve-based technology offers profound economic and operational benefits that extend beyond simple yield improvements. The elimination of expensive and volatile tungsten or molybdenum salts directly translates to a significant reduction in raw material costs, while the ability to recover and reuse the solid catalyst multiple times further amortizes the catalyst expense over numerous production batches. This shift from a consumable homogeneous catalyst to a durable heterogeneous system drastically simplifies the supply chain logistics, as there is no longer a dependency on the continuous sourcing of high-purity heavy metal salts which are subject to market fluctuations and regulatory scrutiny. Furthermore, the simplified workup procedure, which replaces complex ion-exchange columns with standard filtration units, reduces the capital expenditure required for plant equipment and lowers the operational overhead associated with resin regeneration and waste disposal.
- Cost Reduction in Manufacturing: The transition to a recyclable solid catalyst system fundamentally alters the cost structure of epoxysuccinic acid production by removing the need for costly heavy metal scavengers and extensive purification steps. Since the titanium silicalite catalyst can be filtered, dried, and reintroduced into the reactor, the effective cost per kilogram of catalyst consumed is drastically lowered compared to single-use tungsten salts. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes the loss of valuable maleic anhydride feedstock to byproducts like tartaric acid, thereby maximizing the overall atom economy of the process.
- Enhanced Supply Chain Reliability: Relying on solid molecular sieve catalysts mitigates the supply risks associated with specialty metal salts, which can be subject to geopolitical trade restrictions and mining volatility. The robustness of the molecular sieve allows for longer storage stability and easier handling, reducing the risk of supply disruptions due to catalyst degradation or hazardous material transport regulations. Moreover, the simplified purification train means that production cycles are shorter and more predictable, enabling manufacturers to respond more agilely to fluctuating market demands for water treatment chemicals and pharmaceutical intermediates without the bottleneck of lengthy resin regeneration protocols.
- Scalability and Environmental Compliance: The inherent safety features of the negative pressure operation make this process highly scalable, as the risk of thermal runaway is effectively managed by the evaporative cooling effect, a critical factor when moving from pilot plants to multi-ton reactors. From an environmental perspective, the absence of heavy metal effluents simplifies wastewater treatment compliance, allowing facilities to meet increasingly strict discharge standards without investing in advanced tertiary treatment technologies. The reduction in liquid waste volume, achieved by avoiding large volumes of wash water required for resin cleaning, further aligns the manufacturing process with green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic system, derived directly from the experimental data and advantageous effects described in the patent documentation.
Q: How does the molecular sieve catalyst improve product purity compared to traditional methods?
A: Unlike traditional tungsten or molybdenum salts which are homogeneous and difficult to separate, the solid molecular sieve catalyst can be removed via simple filtration. This fundamentally eliminates heavy metal residues in the final epoxysuccinic acid solution, ensuring higher purity standards required for sensitive applications like pharmaceutical intermediates.
Q: What is the role of negative pressure in this epoxidation process?
A: Operating under negative pressure (-0.045 to -0.065 MPa) serves two critical functions: it continuously removes water generated during the reaction to maintain optimal concentration, and it facilitates the removal of exothermic heat. This thermal management prevents the hydrolysis of the epoxide ring into tartaric acid, thereby significantly improving selectivity and yield.
Q: Is the titanium silicalite catalyst reusable for industrial scale-up?
A: Yes, the patent data confirms that the titanium silicalite molecular sieve catalyst can be recovered by filtration, dried, and reused for subsequent batches without significant loss of activity. This recyclability drastically reduces catalyst consumption costs and minimizes solid waste generation compared to single-use homogeneous catalytic systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epoxysuccinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the molecular sieve-catalyzed epoxidation route for producing high-quality epoxysuccinic acid. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs are equipped to verify stringent purity specifications, guaranteeing that every batch meets the low heavy metal and low tartaric acid profiles essential for downstream applications in water treatment and fine chemical synthesis. We are committed to leveraging this green technology to deliver a superior product that balances performance with environmental responsibility.
We invite global partners to collaborate with us to optimize their supply chains for epoxysuccinic acid and related derivatives. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how this advanced catalytic method can enhance your product portfolio while reducing your total cost of ownership.
