Breakthrough Synthesis of Stable Isotope Labeled Tauryl Amine Hydrochloride for Commercial Scale-Up
Breakthrough Synthesis of Stable Isotope Labeled Tauryl Amine Hydrochloride for Commercial Scale-Up
The pharmaceutical industry continuously demands higher precision in metabolic studies, driving the need for advanced stable isotope labeled compounds. Patent CN114539106A discloses a robust synthetic method for stable isotope labeled tauryl amine hydrochloride, a critical metabolite of the antibacterial agent taurolidine. This technology addresses the significant challenges associated with maintaining isotopic abundance during complex organic synthesis, particularly avoiding the loss of deuterium atoms which plagues traditional exchange methods. By utilizing lithium aluminum deuteride or sodium boron deuteride in conjunction with 15N-marked ammonia water, the process achieves an isotopic abundance greater than 98% and chemical purity exceeding 99.4%. For R&D directors and procurement specialists seeking a reliable stable isotope labeled tauryl amine hydrochloride supplier, this methodology represents a paradigm shift in producing high-fidelity reference standards for clinical pharmacokinetic research.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of non-labeled tauryl amine has relied on routes involving cysteine hydrochloride oxidation or taurine protection strategies, yet adapting these for stable isotope labeling presents severe technical hurdles. Previous attempts to synthesize deuterated products using inexpensive precursors like 1,2-dibromoethane-d4 failed because two deuterons on the sulfone-bound methylene group were significantly lost during the sulfonyl chloride synthesis step. Furthermore, deuterium exchange methods proved ineffective, unable to yield the target product with the high deuterium abundance required for rigorous scientific analysis. These limitations not only increase the difficulty of synthesizing stable isotopically labeled products but also inflate costs due to low yields and the need for extensive purification to remove unlabeled impurities. Consequently, the lack of a reported synthesis method for isotopically labeled bezoar amide in prior literature created a supply gap for researchers studying the metabolic mechanism of taurolidine drugs.
The Novel Approach
The patented innovation overcomes these historical barriers by introducing a five-step reaction sequence that efficiently converts isotope-marked raw materials into the target product without significant isotopic loss. Instead of attempting to label the molecule post-synthesis, this novel approach integrates the isotopic labels at the very beginning of the synthetic pathway using specialized reducing agents and nitrogen sources. This strategic design ensures that the deuterium and nitrogen-15 labels are structurally locked into the carbon and nitrogen frameworks before the harsh oxidation steps occur. The result is a process that is reasonable in design and strong in operability, capable of realizing industrialized production of isotope labeled tauryl amine. For stakeholders focused on cost reduction in pharmaceutical intermediates manufacturing, this route eliminates the waste associated with failed labeling attempts and provides a consistent source of high-purity material.
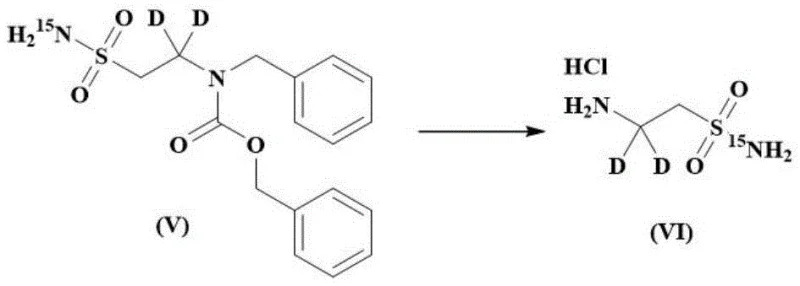
Mechanistic Insights into Deuterium and Nitrogen-15 Incorporation
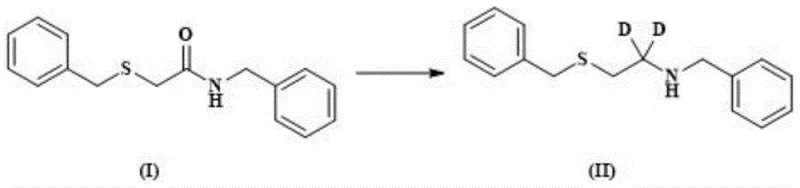
The core of this synthesis lies in the precise control of reaction conditions to preserve isotopic integrity throughout the transformation. The first critical stage involves the reduction of the starting material using a deuterated reducing agent, such as a sodium deuteride boron plus Lewis acid system, preferably boron trifluoride diethyl etherate. This step is conducted at controlled temperatures between 0-80°C, preferably 50°C, to ensure complete conversion while minimizing side reactions that could lead to proton-deuterium exchange with the solvent. Following reduction, the intermediate undergoes amino protection and subsequent oxidation using agents like 1,3-dichloro-5,5-dimethylhydantoin. This oxidation step converts the sulfide moiety into the necessary sulfonyl chloride functionality, a transformation that previously caused deuterium loss in older methods but is now managed effectively through the stability of the preceding intermediates.
Impurity control is paramount in the production of reference standards, and this mechanism inherently limits the formation of unlabeled byproducts. By avoiding the use of non-labeled reagents in the key bond-forming steps, the process ensures that the final impurity profile is dominated by chemically related substances rather than isotopic variants. The final deprotection step utilizes catalytic palladium carbon under hydrogen pressure to remove protecting groups, followed by recrystallization to achieve the stringent purity specifications required for analytical applications. This meticulous attention to mechanistic detail allows for the commercial scale-up of complex pharmaceutical intermediates with confidence in the batch-to-batch consistency of isotopic abundance. For supply chain heads, understanding this mechanism confirms that the process is not merely a laboratory curiosity but a robust manufacturing protocol capable of reducing lead time for high-purity API intermediates.
How to Synthesize Stable Isotope Labeled Tauryl Amine Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent choice, temperature control, and reagent stoichiometry to maximize yield and isotopic retention. The process begins with the dissolution of raw material in dried THF, followed by the careful addition of the deuterated reducing agent under ice bath conditions to manage exothermicity. Subsequent steps involve sequential protection, oxidation, ammonolysis with 15N-labeled ammonia, and final hydrogenolysis, each requiring specific monitoring via TLC to ensure reaction completion before proceeding. The detailed standardized synthesis steps see the guide below, which outlines the precise operational protocols necessary to replicate the high yields and purity reported in the patent data.
- Reduction of raw material I using lithium aluminum deuteride or sodium boron deuteride to introduce deuterium labels.
- Protection of the amino group followed by oxidation to form the sulfonyl chloride intermediate.
- Ammonolysis with 15N-labeled ammonia water and final deprotection to yield the target hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial advantages by resolving traditional supply chain and cost pain points associated with specialty isotope-labeled chemicals. The ability to efficiently convert labeled raw materials into the target product means that expensive isotopic reagents are not wasted on low-yield processes, leading to significant cost savings in the overall manufacturing budget. Furthermore, the high operability and reasonable process design facilitate a smoother transition from laboratory scale to industrial production, ensuring that supply continuity is maintained even for niche pharmaceutical intermediates. By eliminating the need for complex and often unsuccessful deuterium exchange procedures, manufacturers can drastically simplify their production workflows and reduce the environmental burden associated with multiple purification cycles.
- Cost Reduction in Manufacturing: The elimination of inefficient labeling steps and the high conversion efficiency of the starting materials directly contribute to a more economical production model. By avoiding the loss of expensive deuterated reagents during the sulfonyl chloride synthesis, the process ensures that the cost of goods sold is optimized without compromising on the quality of the final isotope-labeled standard. This qualitative improvement in process efficiency translates to better pricing stability for downstream purchasers who rely on these materials for critical drug development studies.
- Enhanced Supply Chain Reliability: The use of readily available reagents and standard reaction conditions enhances the reliability of the supply chain by reducing dependency on exotic or hard-to-source catalysts. The robustness of the five-step route means that production schedules are less likely to be disrupted by technical failures or the need for re-processing batches that fail isotopic specifications. This reliability is crucial for procurement managers who need to secure long-term contracts for high-purity OLED material or pharmaceutical intermediates without fear of unexpected shortages.
- Scalability and Environmental Compliance: The process is designed with industrial production in mind, featuring steps that are easily scalable from 100 kgs to 100 MT annual commercial production volumes. Additionally, the streamlined nature of the synthesis reduces the volume of chemical waste generated per unit of product, aligning with increasingly stringent environmental compliance regulations. This scalability ensures that as demand for taurolidine metabolite standards grows, the supply can be expanded rapidly to meet market needs without requiring fundamental changes to the manufacturing infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this stable isotope labeled compound. These answers are derived directly from the technical disclosures and beneficial effects outlined in the patent documentation to provide clarity on the process capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this material into their existing research and development pipelines.
Q: Why is this isotope labeling method superior to conventional exchange methods?
A: Conventional deuterium exchange methods often result in significant loss of isotopic abundance at the sulfone-bound methylene group. This patented five-step route ensures stable labeling with >98% abundance by incorporating isotopes during the initial reduction and ammonolysis steps rather than relying on post-synthesis exchange.
Q: What is the chemical purity achievable with this synthesis route?
A: The optimized process conditions described in the patent allow for the production of isotope labeled tauryl amine hydrochloride with a chemical purity exceeding 99.4%. This high level of purity is critical for its application as a standard substance in clinical pharmacokinetic research.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly states that the process design is reasonable and possesses strong operability for industrialized production. The use of standard reagents and manageable reaction conditions facilitates the commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stable Isotope Labeled Tauryl Amine Hydrochloride Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to support your drug development initiatives with unparalleled quality and consistency. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our commitment to stringent purity specifications and the operation of rigorous QC labs guarantees that every batch of stable isotope labeled tauryl amine hydrochloride meets the exacting standards required for clinical pharmacokinetic research and regulatory submissions.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall R&D expenditures. We encourage potential partners to reach out for specific COA data and route feasibility assessments to confirm that our capabilities align perfectly with your strategic sourcing goals for high-value pharmaceutical intermediates.
