Advanced Regioselective Synthesis of Pyrazolopyrrolidine Intermediates for Type 2 Diabetes Treatment
The pharmaceutical industry's relentless pursuit of effective treatments for Type 2 diabetes has placed significant emphasis on the development of dipeptidyl peptidase-IV (DPP-IV) inhibitors. Patent CN103702562A discloses a groundbreaking process for the preparation of chiral pyrazolopyrrolidines, which serve as critical intermediates in the synthesis of these potent therapeutic agents. This technology represents a substantial leap forward in process chemistry, addressing long-standing challenges related to regioselectivity and purification efficiency. By introducing a novel sulfonylation and isomerization strategy, the inventors have established a robust pathway that significantly enhances the yield of the desired positional isomer. For R&D directors and process chemists, this patent offers a compelling solution to optimize the manufacturing of complex heterocyclic scaffolds essential for modern antidiabetic medications.
For procurement managers and supply chain heads, the implications of this technology extend beyond mere chemical elegance; it translates directly into operational reliability and cost efficiency. The ability to produce high-purity intermediates without resorting to labor-intensive purification methods like column chromatography is a game-changer for commercial scale-up. This report delves deep into the technical nuances of this regioselective process, analyzing its mechanistic underpinnings and evaluating its potential for reducing lead times in high-purity pharmaceutical intermediates manufacturing. As a reliable pharmaceutical intermediate supplier, understanding these advanced synthetic routes is paramount for ensuring a continuous and cost-effective supply of vital drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
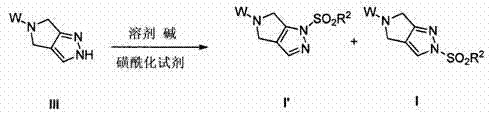
Prior to the innovations described in CN103702562A, the synthesis of pyrazolopyrrolidines faced significant hurdles, particularly regarding regiocontrol. As documented in earlier literature such as PCT International Patent Application WO 2010/056708, conventional synthetic routes often resulted in a problematic 1:1 mixture of structural isomers, specifically formula Ia and formula Ib. This lack of selectivity posed a severe bottleneck for manufacturing, as isolating the biologically active isomer required additional, resource-intensive purification steps. Typically, this involved column chromatography, a technique that is notoriously difficult to scale up for industrial production due to high solvent consumption, low throughput, and significant waste generation. For a commercial operation, relying on chromatographic separation drastically increases the cost of goods sold (COGS) and extends the production cycle time, thereby impacting the overall supply chain agility.
The Novel Approach
The present invention overcomes these limitations through a sophisticated regioisomer-selective process that leverages controlled sulfonylation followed by strategic isomerization. Instead of accepting a statistical mixture of products, this method actively drives the reaction towards the desired pyrazolopyrrolidine regioisomer, designated as formula I. The process can be executed either as a single-step "one-pot" procedure or a two-step sequence, offering flexibility depending on the specific substrate and production requirements. By utilizing specific sulfonylating agents and bases, the reaction conditions are tuned to favor the formation of the target isomer, often achieving conversion rates exceeding 90% and selectivity ratios that render chromatographic purification unnecessary. This shift from separation-based purification to reaction-based selectivity is a hallmark of green chemistry and process intensification.
This fundamental change in strategy eliminates the need for column chromatography, replacing it with more scalable unit operations such as crystallization and filtration. The ability to generate a greater percentage of the desired product directly from the reaction mixture means that downstream processing is simplified, leading to substantial reductions in solvent usage and waste disposal costs. For manufacturers aiming for cost reduction in API manufacturing, this approach provides a clear pathway to improve margins while maintaining stringent quality standards. The versatility of the method allows it to be adapted for various substituents on the pyrazole ring, making it a platform technology for a wide range of DPP-IV inhibitor analogs.
Mechanistic Insights into Regioselective Sulfonylation and Isomerization
The core of this technological advancement lies in the precise control of the sulfonylation reaction and the subsequent isomerization equilibrium. The process begins with the sulfonylation of a pyrazolopyrrolidine precursor, such as a compound of formula III. When reacted with a sulfonylating agent like methanesulfonyl chloride (MsCl) in the presence of a base, the initial attack can occur at different nitrogen atoms within the heterocyclic system, potentially leading to a mixture of kinetic and thermodynamic products. The choice of base and solvent plays a critical role in dictating the outcome. For instance, using strong non-nucleophilic bases like sodium hexamethyldisilazide (NaHMDS) in polar aprotic solvents such as DMF or DMAc at low temperatures (e.g., -20°C) can influence the initial distribution of isomers. However, the true power of the process is revealed in the isomerization step, where the kinetic product is converted into the more stable thermodynamic product.
In the two-step embodiment, the initial sulfonylation yields a mixture of isomers, often denoted as I' and I. The subsequent addition of a second base, such as potassium tert-butoxide (KOtBu) or additional NaHMDS, facilitates the migration of the sulfonyl group to the preferred nitrogen position. This isomerization is driven by thermodynamic stability and can be optimized by adjusting temperature and reaction time. Experimental data from the patent indicates that maintaining the reaction at controlled temperatures, such as -10°C to 0°C, allows for high conversion efficiencies. For example, specific embodiments demonstrate that treating the mixture with KOtBu in THF can shift the isomer ratio to favor the desired product I by a factor of over 1600:1. This level of selectivity is achieved through careful manipulation of the reaction environment, ensuring that the equilibrium strongly favors the target structure.
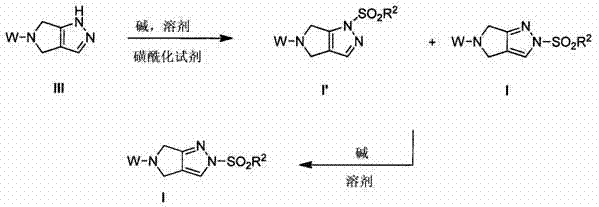
Furthermore, the mechanism allows for the formation of stable salts, which aids in purification. Once the desired free base is obtained, it can be converted into a salt form, such as the benzenesulfonate salt, using acids like benzenesulfonic acid (BSA). Salt formation not only improves the physical properties of the intermediate, such as solubility and crystallinity, but also serves as an effective purification step to remove organic and inorganic impurities. The recrystallization of these salts from solvent systems like isopropyl acetate or THF/heptane mixtures ensures that the final intermediate meets the rigorous purity specifications required for pharmaceutical applications. This mechanistic understanding enables process chemists to fine-tune reaction parameters to maximize yield and minimize impurity profiles, ensuring a robust and reproducible manufacturing process.
How to Synthesize Chiral Pyrazolopyrrolidine Efficiently
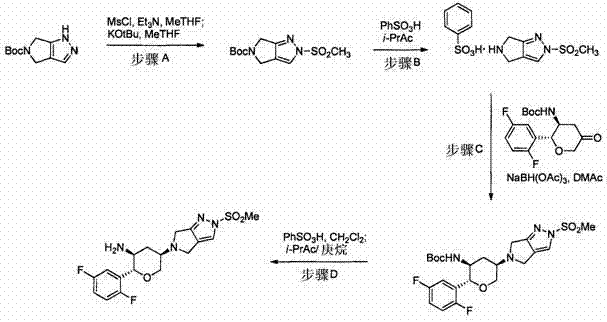
The synthesis of these valuable intermediates follows a logical progression from the protected pyrazole precursor to the final deprotected amine ready for coupling. The process is designed to be operationally simple, utilizing common laboratory and plant equipment. Key to the success of this synthesis is the strict control of stoichiometry and temperature during the sulfonylation phase to prevent over-reaction or decomposition. Following the isomerization, the workup procedures involve standard aqueous quenches and extractions, which are easily scalable. The final steps involve reductive amination with a chiral ketone component, linking the pyrazole scaffold to the rest of the DPP-IV inhibitor molecule. This modularity allows for the efficient production of various analogs by simply changing the ketone partner. Detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Perform regioselective sulfonylation of the pyrazolopyrrolidine precursor using methanesulfonyl chloride and a base such as triethylamine or NaHMDS in solvents like MeTHF or DMF.
- Execute an isomerization step using a second base like KOtBu or NaHMDS to convert the kinetic isomer into the desired thermodynamic product with high selectivity.
- Form a stable salt (e.g., benzenesulfonate), followed by reductive amination with a chiral ketone and final deprotection to yield the active pharmaceutical intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this regioselective synthesis method offers transformative benefits that go beyond simple yield improvements. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the need for column chromatography, which is a major bottleneck in fine chemical manufacturing, companies can significantly reduce their reliance on expensive silica gel and vast quantities of organic solvents. This reduction in material consumption directly correlates to lower operational expenditures and a smaller environmental footprint. Moreover, the removal of chromatographic steps shortens the overall batch cycle time, allowing for faster turnover and increased production capacity without the need for additional capital investment in equipment. This efficiency is crucial for meeting the demanding delivery schedules of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the avoidance of chromatographic purification. Chromatography is not only slow but also consumes large volumes of solvents that require recovery or disposal, adding significant cost to the final product. By shifting to a crystallization-based purification strategy enabled by high regioselectivity, manufacturers can achieve substantial cost savings. The use of commodity reagents like methanesulfonyl chloride and triethylamine further ensures that raw material costs remain low and predictable. Additionally, the high conversion rates reported in the patent examples mean that less starting material is wasted, improving the overall atom economy of the process. These factors combine to create a highly cost-competitive manufacturing route suitable for high-volume production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to variability. This new method enhances reliability by utilizing robust reaction conditions that are less sensitive to minor fluctuations. The ability to perform the reaction in common solvents like THF, MeTHF, and DMAc ensures that raw material sourcing is straightforward and resilient to market shortages. Furthermore, the formation of stable crystalline salts allows for easier storage and transportation of intermediates, reducing the risk of degradation during logistics. This stability is vital for maintaining a consistent supply of high-quality intermediates to downstream API manufacturers, ensuring that drug production schedules are met without interruption.
- Scalability and Environmental Compliance: Scalability is a critical consideration for any process intended for commercial use. The patent provides evidence of successful kilogram-scale runs, demonstrating that the chemistry translates well from the lab to the plant. The two-step process, in particular, allows for better heat management and control over exothermic reactions, which is essential for safety at large scales. From an environmental perspective, the reduction in solvent waste and the elimination of silica waste from chromatography align with green chemistry principles. This makes the process more attractive from a regulatory standpoint, facilitating smoother approvals and compliance with increasingly stringent environmental regulations. The ease of waste treatment and solvent recovery further enhances the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this regioselective synthesis technology. These insights are derived directly from the experimental data and embodiments described in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines. The answers provide clarity on reaction conditions, scalability, and the specific advantages over legacy methods.
Q: How does this process improve upon previous methods for pyrazolopyrrolidine synthesis?
A: Unlike prior art methods that produced a 1:1 mixture of regioisomers requiring difficult column chromatography, this novel process utilizes a specific sulfonylation and isomerization sequence to achieve high regioselectivity for the desired isomer directly.
Q: What are the key reaction conditions for the isomerization step?
A: The isomerization typically involves treating the sulfonylated mixture with a strong base such as potassium tert-butoxide (KOtBu) or NaHMDS in polar aprotic solvents like DMAc or THF at controlled temperatures ranging from -10°C to room temperature.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the patent describes kilogram-scale examples (e.g., 30 kg scale in Step A) utilizing standard industrial reagents and avoiding complex purification techniques like chromatography, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolopyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the technology disclosed in CN103702562A and possesses the capability to implement these advanced regioselective processes at a commercial scale. We understand that transitioning from a novel patent to a robust manufacturing line requires deep technical expertise and state-of-the-art facilities. Our organization boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We are committed to delivering intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation.
We invite you to collaborate with us to leverage this cutting-edge technology for your DPP-IV inhibitor programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to a supply chain that prioritizes efficiency, quality, and reliability. Please contact us today to request specific COA data and route feasibility assessments for your projects. Let us help you optimize your manufacturing strategy and secure a competitive advantage in the global market for diabetes therapeutics.
