Advanced Synthesis of Trans-4-Alkyl Cyclohexyl Formic Acid for Commercial Scale-Up
Introduction to Next-Generation Liquid Crystal Intermediate Synthesis
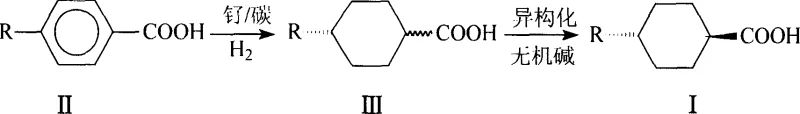
The synthesis of high-purity trans-4-alkyl cyclohexyl formic acid represents a critical advancement in the manufacturing of liquid crystal materials, as detailed in patent CN1239460C. This specific chemical architecture is fundamental for producing cyclohexane-based liquid crystals, which are prized in the display industry for their superior transformation temperatures and lower viscosity compared to traditional biphenyl systems. The technological breakthrough described in this patent addresses long-standing inefficiencies in the production of these vital intermediates, offering a streamlined pathway that integrates hydrogenation and isomerization into a cohesive, industrially viable workflow. By leveraging supported ruthenium catalysts and a novel solvent-free isomerization technique, the process achieves exceptional stereochemical control, driving the trans-isomer content to levels exceeding 96% without the need for exhaustive recrystallization cycles. This development is not merely a laboratory curiosity but a robust solution designed for the rigorous demands of commercial chemical manufacturing, promising significant enhancements in both yield and operational simplicity for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of trans-4-alkyl cyclohexyl formic acid has been plagued by thermodynamic and operational hurdles that severely impacted cost-efficiency and scalability. Traditional isomerization methods often relied on harsh acidic conditions, such as heating in hydrochloric acid solutions at temperatures approaching 180°C, which necessitated the use of expensive, corrosion-resistant reactor equipment and posed significant safety risks. Alternative approaches involved multi-step sequences including esterification, base-catalyzed isomerization with sodium hydride, and subsequent hydrolysis, each step introducing potential yield losses and increasing the complexity of waste management. Furthermore, prior art techniques frequently struggled to shift the equilibrium sufficiently towards the desired trans-isomer, typically resulting in cis/trans ratios hovering around 25/75 to 15/85, thereby mandating multiple, yield-depleting recrystallization steps to achieve pharmaceutical or electronic grade purity. These legacy processes were not only capital intensive due to the need for high-pressure vessels and specialized materials but also environmentally burdensome due to the generation of large volumes of acidic or saline wastewater.
The Novel Approach
In stark contrast, the methodology outlined in CN1239460C introduces a paradigm shift by utilizing a tandem strategy that seamlessly couples catalytic hydrogenation with solid-phase isomerization. The process initiates with the hydrogenation of 4-alkyl-benzoic acid in an aqueous alkaline medium using a commercially available ruthenium on carbon catalyst, generating a mixture of cis and trans isomers. Crucially, instead of isolating the product through complex extraction or neutralization immediately, the reaction mixture is simply filtered to remove the catalyst and then concentrated via distillation to remove water. The resulting solid residue, containing the crude product and the inorganic base, is directly subjected to thermal isomerization under a nitrogen atmosphere. This ingenious elimination of intermediate separation steps not only drastically reduces solvent consumption but also leverages the residual inorganic base to catalyze the isomerization effectively in the solid state. The outcome is a highly favorable cis/trans ratio ranging from 4/96 to 1/99, achieved with an overall yield surpassing 90%, demonstrating a level of efficiency that renders previous methods obsolete for large-scale applications.
Mechanistic Insights into Ru/C Catalyzed Hydrogenation and Solid-Phase Isomerization
The core of this synthetic success lies in the precise interplay between heterogeneous catalysis and thermodynamic control during the isomerization phase. The initial hydrogenation step utilizes ruthenium dispersed on a carbon support, a catalyst choice that provides high surface area and stability, facilitating the reduction of the aromatic ring under moderate temperatures of 80°C to 130°C and pressures of 2 to 7 MPa. The presence of inorganic hydroxide, such as potassium or sodium hydroxide, serves a dual purpose: it solubilizes the carboxylic acid substrate in the aqueous phase, ensuring efficient contact with the catalyst, and it remains in the system to act as a base catalyst for the subsequent isomerization. Mechanistically, the isomerization is driven by the thermodynamic stability of the trans-isomer, where both the alkyl and carboxyl substituents occupy equatorial positions on the cyclohexane chair conformation, minimizing steric strain compared to the axial-equatorial arrangement of the cis-isomer. By heating the solid mixture to temperatures between 120°C and 250°C, sufficient thermal energy is provided to overcome the rotational barrier, allowing the system to equilibrate towards the more stable trans-configuration without the dilution effects of a solvent, which would otherwise slow down the reaction kinetics and require higher energy inputs for solvent removal later.
From an impurity control perspective, this solvent-free solid-state approach offers distinct advantages over solution-phase methods. The absence of organic solvents during the high-temperature isomerization step eliminates the risk of solvent-derived side reactions or the formation of azeotropes that could complicate downstream purification. Furthermore, because the inorganic base is not separated prior to isomerization, the process avoids the introduction of additional acidic quenching steps that could lead to salt formation and product entrapment. The high selectivity observed, with trans-isomer content reaching up to 99%, suggests that the solid-state environment may restrict certain conformational pathways that lead to degradation, thereby preserving the integrity of the alkyl chain. This mechanistic robustness ensures that the final product profile is clean, requiring minimal downstream processing—often just a single recrystallization or even none at all—to meet the stringent purity specifications required for liquid crystal applications, thus maintaining a tight impurity spectrum that is critical for device performance.
How to Synthesize Trans-4-Alkyl Cyclohexyl Formic Acid Efficiently
The practical implementation of this synthesis route is designed for straightforward adoption in existing multipurpose chemical plants, requiring only standard hydrogenation reactors and heating units capable of reaching 250°C. The process begins by charging 4-alkyl-benzoic acid and an aqueous solution of potassium or sodium hydroxide into a pressure vessel, followed by the addition of the Ru/C catalyst. After hydrogenation is complete, indicated by the cessation of hydrogen uptake, the catalyst is filtered off, and the filtrate is concentrated to dryness. The resulting solid is then heated under nitrogen flow to effect isomerization, cooled, dissolved in water, acidified, and extracted to yield the pure product. For a detailed, step-by-step breakdown of the exact molar ratios, temperature ramps, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Hydrogenate 4-alkyl-benzoic acid in aqueous alkali using Ru/C catalyst at 80-130°C and 2-7MPa pressure.
- Filter the catalyst and distill the reaction mixture to remove water, obtaining a solid residue containing the product and base.
- Heat the solid residue to 120-250°C under nitrogen for solid-phase isomerization to achieve high trans-isomer purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The most significant advantage is the drastic simplification of the unit operations; by eliminating the need for intermediate isolation, solvent exchanges, and corrosive acidic treatments, the process reduces the total cycle time per batch and minimizes the consumption of auxiliary materials. This streamlining translates into substantial cost reductions in liquid crystal intermediate manufacturing, as fewer processing steps mean lower labor costs, reduced energy consumption for heating and cooling, and decreased waste disposal fees associated with solvent and salt byproducts. Moreover, the use of robust, commercially available catalysts and common inorganic bases ensures that raw material sourcing is stable and不受 geopolitical fluctuations that might affect exotic reagents, thereby enhancing the resilience of the supply chain against market volatility.
- Cost Reduction in Manufacturing: The elimination of organic solvents during the critical isomerization step represents a major area of cost savings, as solvent purchase, recovery, and disposal are often among the highest variable costs in fine chemical production. Additionally, the high yield of over 90% means that less raw material is wasted, maximizing the output per kilogram of input and improving the overall atom economy of the process. The ability to reuse the inorganic base effectively within the reaction sequence further lowers the cost of goods sold, making the final product highly competitive in price-sensitive markets like consumer electronics.
- Enhanced Supply Chain Reliability: The operational simplicity of this method reduces the likelihood of batch failures caused by complex process controls or sensitive reagent handling. Since the reaction tolerates a broad range of temperatures and pressures without compromising safety or yield, production scheduling becomes more predictable, allowing for tighter delivery commitments to downstream customers. The reliance on standard equipment rather than specialized corrosion-resistant alloys for acidic conditions also means that more contract manufacturing organizations (CMOs) can potentially produce this intermediate, diversifying the supplier base and reducing single-source risk.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial tonnage is facilitated by the absence of difficult-to-handle exotherms associated with strong acid additions or reactive metal hydrides. The solvent-free nature of the isomerization step significantly reduces the facility's volatile organic compound (VOC) emissions, simplifying environmental permitting and compliance with increasingly strict green chemistry regulations. This environmental friendliness not only future-proofs the supply chain against regulatory tightening but also aligns with the sustainability goals of major multinational corporations seeking greener supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of trans-4-alkyl cyclohexyl formic acid using this patented technology. These answers are derived directly from the experimental data and process descriptions found in the source intellectual property, providing a transparent view of the method's capabilities and limitations for potential partners.
Q: What is the key advantage of the solid-phase isomerization method?
A: The solid-phase isomerization eliminates the need for organic solvents and complex separation steps between hydrogenation and isomerization, significantly reducing operational costs and environmental waste while achieving a trans/cis ratio of up to 99:1.
Q: Why is Ruthenium on Carbon (Ru/C) preferred for this hydrogenation?
A: Ru/C offers high catalytic activity for aromatic ring hydrogenation under relatively mild conditions compared to other noble metals, and being a heterogeneous catalyst, it allows for easy filtration and recovery, which is crucial for industrial scalability.
Q: How does this process improve supply chain reliability for liquid crystal materials?
A: By utilizing readily available raw materials like 4-alkyl-benzoic acid and common inorganic bases, and avoiding corrosive acidic conditions or high-pressure specialized equipment for isomerization, the process ensures consistent production uptime and easier regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-Alkyl Cyclohexyl Formic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the performance of advanced liquid crystal displays and fine chemical applications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our infrastructure is designed to handle complex catalytic hydrogenations and high-temperature solid-state reactions safely, guaranteeing a consistent supply of this critical intermediate to support your manufacturing timelines.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can become a strategic asset for your organization.
