Scalable Synthesis of Octaaminophenyl Silsesquioxane Using Novel Iron-Aluminum Catalytic Systems
Scalable Synthesis of Octaaminophenyl Silsesquioxane Using Novel Iron-Aluminum Catalytic Systems
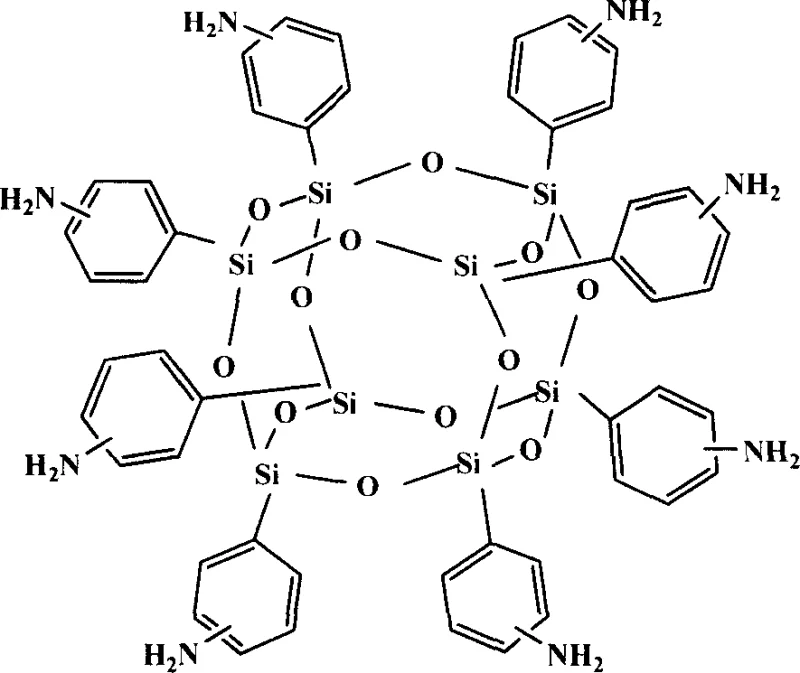
The landscape of advanced material synthesis is undergoing a significant transformation driven by the need for cost-effective and scalable production methods for high-performance nanostructure units. A pivotal development in this domain is documented in patent CN1916003A, which discloses a robust method for preparing octaaminophenyl clathrate silsesquioxane (OAPS). This compound serves as a critical building block for organic-inorganic hybrid polymers, finding extensive applications in high-temperature resistant resins, flame retardants, and precursors for organic light-emitting diodes (OLEDs). The disclosed technology represents a paradigm shift from traditional noble metal-catalyzed reductions to a more economical and environmentally benign transition metal system. By leveraging a synergistic iron-aluminum catalytic couple in conjunction with hydrazine hydrate, this process achieves high yields under mild conditions, effectively addressing the economic and operational bottlenecks associated with previous methodologies. As a reliable octaaminophenyl silsesquioxane supplier, understanding these mechanistic nuances is essential for evaluating the commercial viability of supply chains dependent on these specialized intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of octaaminophenyl clathrate silsesquioxane has relied heavily on reduction protocols employing palladium on carbon (Pd/C) catalysts with formic acid as the hydrogen source. While chemically effective, this conventional approach presents substantial drawbacks for large-scale industrial adoption. The primary concern is the exorbitant cost associated with palladium, a precious metal whose market volatility directly impacts the cost reduction in advanced materials manufacturing. Furthermore, the use of formic acid introduces significant safety and handling challenges; at elevated temperatures required for efficient reduction, formic acid is prone to volatilization, complicating reaction control and posing inhalation risks. Additionally, palladium catalysts are highly susceptible to poisoning by trace impurities in the starting materials, necessitating ultra-high purity reagents and stringent atmospheric controls that drive up operational expenditures. The difficulty in completely removing residual palladium from the final product also poses a risk for downstream applications, particularly in electronics where metal contamination can degrade device performance, thereby limiting the utility of the resulting polymers in sensitive sectors.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent utilizes a non-noble metal catalytic system comprising iron and aluminum compounds, such as ferric chloride and aluminum trichloride, supported by activated carbon. This strategic substitution fundamentally alters the economic equation of the synthesis. By replacing palladium with abundant base metals, the process eliminates the dependency on precious metal markets, offering a pathway for substantial cost savings without compromising catalytic efficiency. The use of hydrazine hydrate as the reducing agent, instead of formic acid, provides a more stable and controllable hydrogen source that operates effectively at moderate temperatures ranging from 50-80°C. This milder thermal profile not only enhances safety by reducing the risk of solvent boil-off and reagent decomposition but also improves the selectivity of the reduction, minimizing side reactions that could lead to complex impurity profiles. The robustness of the iron-aluminum catalyst against poisoning further simplifies the quality control requirements for raw materials, making the process inherently more suitable for the commercial scale-up of complex polymer additives where consistency is paramount.
Mechanistic Insights into Fe/Al-Catalyzed Transfer Hydrogenation
The core of this innovative synthesis lies in the mechanism of transfer hydrogenation facilitated by the iron-aluminum catalyst system. In this reaction, hydrazine hydrate acts as the hydrogen donor, decomposing on the surface of the activated carbon and metal catalyst to generate active hydrogen species in situ. The iron species, likely functioning through redox cycling between Fe(II) and Fe(III) states, activate the nitro groups on the octanitrophenyl precursor, rendering them susceptible to nucleophilic attack by the generated hydrogen. The presence of aluminum compounds appears to play a Lewis acidic role, potentially coordinating with the oxygen atoms of the nitro groups or the silsesquioxane cage, thereby polarizing the N-O bonds and lowering the activation energy for reduction. This synergistic interaction between the transition metal and the Lewis acid creates a highly active catalytic environment that drives the conversion of nitro groups to amino groups with high fidelity. The activated carbon support not only disperses the metal salts to maximize surface area but may also participate in electron transfer processes, stabilizing intermediate radical species and preventing the formation of azo or azoxy byproducts that are common in incomplete reductions.
From an impurity control perspective, this mechanism offers distinct advantages over palladium-catalyzed routes. The absence of noble metals means there is no risk of leaching toxic palladium residues into the final product, a critical factor for high-purity silsesquioxane derivatives intended for biomedical or electronic applications. Furthermore, the mild reaction conditions minimize the thermal degradation of the silsesquioxane cage structure, which can be sensitive to harsh acidic or basic environments. The selectivity of the hydrazine reduction ensures that the amino groups are formed cleanly without affecting the integrity of the Si-O-Si backbone. Post-reaction workup involves simple filtration to remove the solid catalyst and activated carbon, followed by standard washing and drying procedures. This straightforward purification protocol contrasts sharply with the complex scavenging resins or chromatography often required to meet strict metal limits in palladium-catalyzed processes, thereby streamlining the production workflow and reducing the overall processing time and waste generation associated with the manufacturing of these specialized intermediates.
How to Synthesize Octaaminophenyl Silsesquioxane Efficiently
The practical execution of this synthesis requires careful attention to reagent stoichiometry and atmospheric conditions to maximize yield and purity. The process begins with the dissolution of the octanitrophenyl precursor in a suitable solvent, with tetrahydrofuran (THF) being the preferred medium due to its excellent solubility characteristics and compatibility with the catalyst system. The addition of activated carbon, which may be pre-treated with nitric acid to remove trace heavy metals, is crucial for supporting the catalytic activity. The iron and aluminum catalysts are introduced in specific molar ratios relative to the nitro groups to ensure complete conversion without excessive metal loading. The reaction is conducted under an inert nitrogen atmosphere to prevent the oxidation of the newly formed amine groups, which are sensitive to air. Hydrazine hydrate is added dropwise to control the exotherm and gas evolution, followed by a reflux period that allows the reduction to proceed to completion. Detailed standardized synthetic steps see the guide below.
- Dissolve octanitrophenyl clathrate silsesquioxane in tetrahydrofuran and add activated carbon along with iron and aluminum chloride catalysts.
- Heat the mixture to 50-80°C under nitrogen atmosphere and slowly add hydrazine hydrate dropwise while maintaining reflux conditions.
- Filter the reaction mixture to remove solids, wash the filtrate, and isolate the final product via vacuum distillation or precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed route offers compelling strategic advantages that extend beyond simple unit cost metrics. The elimination of palladium from the supply chain mitigates exposure to the volatile pricing of precious metals, providing greater predictability in long-term budgeting and cost reduction in advanced materials manufacturing. Furthermore, the reliance on commodity chemicals like iron chloride, aluminum chloride, and hydrazine hydrate ensures a robust and diversified supply base, reducing the risk of single-source bottlenecks that can plague specialty catalyst markets. The simplified purification process, which avoids the need for expensive metal scavengers or complex chromatographic separations, translates directly into reduced processing time and lower utility consumption. This efficiency gain allows for faster turnaround times between batches, enhancing the overall agility of the production facility to respond to fluctuating market demands for high-performance polymer additives.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the substitution of expensive palladium catalysts with inexpensive iron and aluminum salts. This change eliminates the capital tied up in precious metal inventory and removes the recurring cost of catalyst recovery or disposal. Additionally, the avoidance of formic acid reduces the need for specialized corrosion-resistant equipment and ventilation systems required for handling volatile acids, leading to lower capital expenditure (CAPEX) for plant infrastructure. The high selectivity of the reaction minimizes the formation of byproducts, which in turn reduces the loss of valuable starting material and lowers the cost of waste treatment. These cumulative factors contribute to a significantly leaner cost structure, allowing for more competitive pricing of the final octaaminophenyl silsesquioxane product in the global marketplace.
- Enhanced Supply Chain Reliability: The raw materials required for this process are widely available commodities with established global supply networks, unlike specialized palladium catalysts which may have limited suppliers and longer lead times. This abundance ensures continuity of supply even during periods of geopolitical instability or logistics disruptions. The robustness of the catalyst system against impurities means that the process can tolerate slight variations in raw material quality without significant impact on yield, reducing the pressure on quality assurance teams to source ultra-high-grade reagents. This flexibility simplifies vendor management and allows for the qualification of multiple secondary suppliers, thereby de-risking the supply chain. Consequently, manufacturers can maintain consistent production schedules and reducing lead time for high-purity functionalized POSS deliveries to their downstream customers.
- Scalability and Environmental Compliance: The mild operating conditions of 50-80°C and the use of standard solvents like THF make this process highly amenable to scale-up from laboratory to multi-ton production scales. The absence of hazardous formic acid vapors and the reduced generation of heavy metal waste align with increasingly stringent environmental regulations and corporate sustainability goals. The simplified workup procedure generates less hazardous waste, lowering disposal costs and minimizing the environmental footprint of the manufacturing operation. The high reproducibility of the reaction ensures that scaling up does not result in the unpredictable yield drops often seen when transitioning sensitive noble metal catalysis to larger reactors. This reliability is critical for securing long-term contracts with major polymer producers who require guaranteed volumes of high-purity silsesquioxane derivatives for their own commercial scale-up of complex polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of octaaminophenyl silsesquioxane using this novel catalytic method. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering a transparent view into the process capabilities. Understanding these details is vital for R&D teams evaluating the material for new formulations and for procurement specialists assessing supplier qualifications. The answers reflect the consensus on best practices for handling and synthesizing this class of nanostructured materials.
Q: Why is the iron-aluminum catalyst preferred over palladium for this synthesis?
A: The iron-aluminum catalyst system significantly reduces raw material costs by eliminating expensive noble metals and simplifies the purification process by avoiding heavy metal residue removal steps.
Q: What are the optimal reaction conditions for high yield?
A: Optimal conditions involve using tetrahydrofuran as a solvent, maintaining a temperature between 50-80°C, and utilizing a nitrogen atmosphere to prevent amine oxidation during the hydrazine reduction.
Q: How does this method improve scalability for industrial production?
A: The method uses mild temperatures and stable reagents like hydrazine hydrate, avoiding the volatility issues of formic acid and the strict poisoning controls required for palladium catalysts, thus ensuring consistent batch-to-batch reproducibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octaaminophenyl Silsesquioxane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis route for octaaminophenyl silsesquioxane in driving innovation across the advanced materials sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in industrial practice. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify the absence of heavy metal contaminants and ensure consistent batch-to-batch performance. Our facility is equipped to handle the specific solvent and reagent requirements of this process, guaranteeing a secure and efficient supply chain for our global partners who rely on these critical intermediates for high-value applications.
We invite industry leaders to collaborate with us to optimize their material sourcing strategies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments that demonstrate how our implementation of this patented technology can enhance your product competitiveness. Let us help you navigate the complexities of advanced material synthesis and secure a reliable supply of high-quality octaaminophenyl silsesquioxane for your next generation of high-performance polymers.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
