Advanced Metal-Ion Catalyzed Reduction for High-Purity Beta-Halo-Phenylethanol Manufacturing
The chemical industry constantly seeks more efficient pathways to synthesize critical intermediates, particularly those containing sensitive functional groups like halogens adjacent to reactive centers. Patent CN1247504C introduces a groundbreaking methodology for the preparation of beta-halogen-alpha-phenyl ethyl alcohol compounds, a class of molecules indispensable in the synthesis of various pharmaceuticals and agrochemicals. This innovation addresses a long-standing challenge in organic synthesis: the selective reduction of alpha-halo-ketones without compromising the integrity of the halogen substituent. By leveraging the coordination chemistry of specific metal ions such as Calcium (Ca2+), Magnesium (Mg2+), or Lanthanides (Ln3+), this process achieves unprecedented selectivity and yield under mild conditions. For R&D directors and procurement specialists alike, this technology represents a significant leap forward, transforming a potentially hazardous and low-yielding transformation into a robust, scalable, and commercially viable operation that ensures a reliable supply of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the reduction of ketones to alcohols has relied on several established techniques, each carrying distinct disadvantages when applied to alpha-halo substrates. Catalytic hydrogenation, while effective for simple ketones, often necessitates expensive pressure-resistant equipment and precious metal catalysts like platinum or rhodium, driving up capital expenditure and operational costs significantly. Furthermore, this method frequently suffers from over-reduction, leading to the formation of unwanted ethylbenzene derivatives where the halogen is completely removed. Alternatively, the use of strong hydride reagents like lithium aluminum hydride requires strictly anhydrous conditions, complicating the workflow and increasing safety risks due to the reagent's pyrophoric nature. Even standard sodium borohydride reductions, though safer, often fail to maintain selectivity; without specific controls, the reaction generates substantial heat and gas, leading to dangerous overflow risks and a plethora of by-products such as phenyl oxiranes and dehalogenated alcohols, which severely complicate downstream purification and reduce overall process efficiency.
The Novel Approach
The methodology disclosed in the patent fundamentally alters the reaction landscape by introducing stoichiometric amounts of metal ions into the reaction medium prior to reduction. This novel approach utilizes common, inexpensive metal salts like calcium chloride or manganese chloride in conjunction with sodium borohydride in alcoholic solvents. The presence of these metal ions facilitates the formation of a transient but stable complex with the alpha-halo-acetophenone substrate. This complexation effectively shields the alpha-halogen atom from nucleophilic attack or reduction, directing the hydride species exclusively toward the carbonyl group. Consequently, the reaction can proceed smoothly at room temperature or with mild heating, eliminating the need for cryogenic conditions often required to suppress side reactions in conventional protocols. This shift not only simplifies the operational procedure by allowing for liquid addition of reagents rather than hazardous solid dosing but also ensures that the final product retains its critical halogen functionality with exceptional fidelity.
Mechanistic Insights into Metal-Ion Assisted Chemoselective Reduction
The core of this technological advancement lies in the coordination chemistry between the metal cation and the organic substrate. When metal ions such as Ca2+ or Ln3+ are introduced into the alcoholic solution containing the alpha-halo-ketone, they coordinate with the lone pair electrons on the carbonyl oxygen. More critically, due to the proximity and electronic nature of the alpha-halogen, a chelate-like structure is formed. This interaction significantly reduces the electrophilicity of the alpha-carbon, rendering it inert to the nucleophilic hydride ions generated from the borohydride source. Without this protective complexation, the hydride would readily attack the alpha-carbon, displacing the halogen via an SN2 mechanism or inducing elimination reactions that form epoxides. By locking the substrate in this specific conformation, the metal ion acts as a temporary protecting group that is intrinsic to the reaction mechanism rather than an external additive requiring separate protection and deprotection steps.
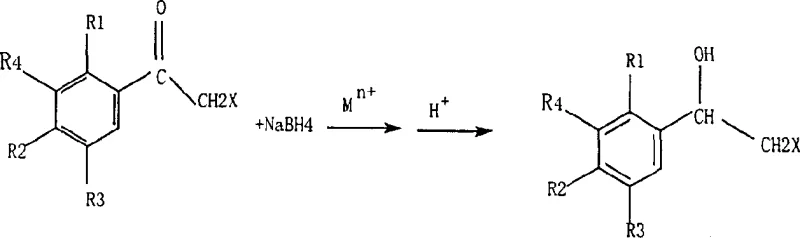
Furthermore, the choice of metal ion offers tunability to the process. The patent highlights a broad spectrum of effective cations, including alkaline earth metals like Strontium and transition metals like Zinc and Copper, as well as the entire series of trivalent lanthanides. Each metal ion possesses a unique ionic radius and Lewis acidity, which influences the stability of the intermediate complex and the kinetics of the hydride transfer. For instance, the use of Calcium ions has been shown to provide excellent results for chlorinated substrates, while Lanthanum ions might offer superior performance for more sterically hindered or fluorinated analogs. This versatility allows process chemists to optimize the reaction conditions for specific substrates within the beta-halogen-alpha-phenyl ethyl alcohol family, ensuring that even complex molecules with multiple sensitive functional groups can be reduced with high chemoselectivity. The mechanism effectively decouples the reduction potential of the carbonyl group from the lability of the carbon-halogen bond, a feat that is difficult to achieve with traditional reducing agents alone.
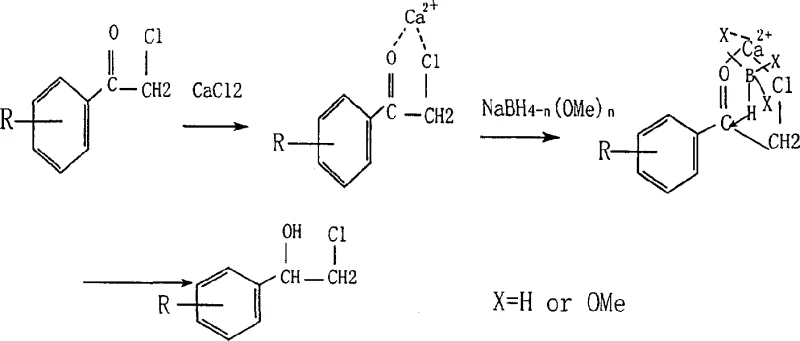
How to Synthesize Beta-Halogen-Alpha-Phenyl Ethyl Alcohol Efficiently
Implementing this synthesis route involves a straightforward sequence of mixing, reduction, and workup that is highly amenable to industrial scale-up. The process begins by dissolving the metal salt and the alpha-halo-ketone in a suitable alcohol solvent, allowing time for the crucial complexation to occur before any reducing agent is introduced. Once the complex is formed, a stabilized aqueous solution of sodium borohydride is added dropwise, controlling the exotherm and gas evolution effectively. Following the reduction, a simple acid quench decomposes the boron complexes and liberates the free alcohol product.
- Complexation: Mix metal chloride or nitrate (e.g., CaCl2) with alpha-halo-acetophenone in alcohol solvent at room temperature.
- Reduction: Dropwise add aqueous sodium borohydride solution to the mixture under stirring and cooling.
- Workup: Quench with dilute acid, distill solvent, extract organic layer, dry, and purify to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The elimination of high-pressure hydrogenation equipment translates to significantly reduced capital investment and lower maintenance costs for manufacturing facilities. Moreover, the ability to conduct the reaction at ambient pressure and near-room temperature drastically lowers energy consumption compared to processes requiring reflux or cryogenic cooling. The use of inexpensive, commodity-grade metal salts like calcium chloride instead of precious metal catalysts further drives down the raw material costs, making the production of these intermediates much more economically attractive. Additionally, the enhanced selectivity means that less starting material is wasted on by-products, improving the overall atom economy and reducing the burden on waste treatment systems, which aligns with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts and high-pressure reactors with common metal salts and standard glass-lined or stainless steel vessels results in substantial cost savings. The process avoids the need for specialized anhydrous solvents or rigorous drying procedures associated with lithium aluminum hydride, further simplifying the supply chain for raw materials. By minimizing the formation of by-products, the downstream purification load is lightened, reducing the consumption of solvents and adsorbents required for chromatography or recrystallization, which collectively contributes to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available reagents such as sodium borohydride and calcium chloride ensures that the supply chain is robust and less susceptible to geopolitical disruptions often associated with rare earth metals or specialized catalysts. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment malfunction, leading to more consistent production schedules and reliable delivery times for customers. This stability is crucial for maintaining continuous operations in the pharmaceutical and agrochemical sectors, where interruptions in the supply of key intermediates can have cascading effects on the production of final active ingredients.
- Scalability and Environmental Compliance: The protocol's inherent safety features, such as the use of liquid reagent addition to control exotherms, make it highly scalable from laboratory benchtop to multi-ton commercial production without significant re-engineering. The reduction in hazardous waste generation, specifically the avoidance of heavy metal catalyst residues and the minimization of halogenated by-products, simplifies compliance with environmental discharge standards. This eco-friendly profile not only mitigates regulatory risks but also enhances the corporate sustainability image, which is increasingly valued by global partners and end-consumers in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-ion assisted reduction technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, aiming to clarify the operational benefits and scope of application for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing manufacturing lines or new product development pipelines.
Q: Why are metal ions like Calcium or Lanthanum added during the reduction of alpha-halo ketones?
A: Metal ions form a stable complex with the carbonyl oxygen and the alpha-halogen, preventing the halogen from being substituted or reduced, thereby drastically improving chemoselectivity.
Q: Does this process require high-pressure equipment like catalytic hydrogenation?
A: No, this method operates at atmospheric pressure and moderate temperatures (room temperature to reflux), eliminating the need for expensive pressure-resistant reactors.
Q: What is the typical purity achievable with this metal-ion assisted method?
A: The process yields products with extremely high purity, often exceeding 97% to nearly 100% as determined by gas chromatography, minimizing downstream purification costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Halogen-Alpha-Phenyl Ethyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development and manufacturing programs. Our team of expert chemists has extensively evaluated the metal-ion catalyzed reduction pathway described in patent CN1247504C and possesses the technical capability to execute this synthesis with precision and efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-halogen-alpha-phenyl ethyl alcohol compounds meets the highest industry standards for impurity profiles and chemical identity.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. By leveraging our expertise in this specific reduction technology, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your project timelines and budgetary goals effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
