Advanced Synthesis of L-Aspartic Acid Beta Benzyl Ester for Commercial Peptide Manufacturing
Advanced Synthesis of L-Aspartic Acid Beta Benzyl Ester for Commercial Peptide Manufacturing
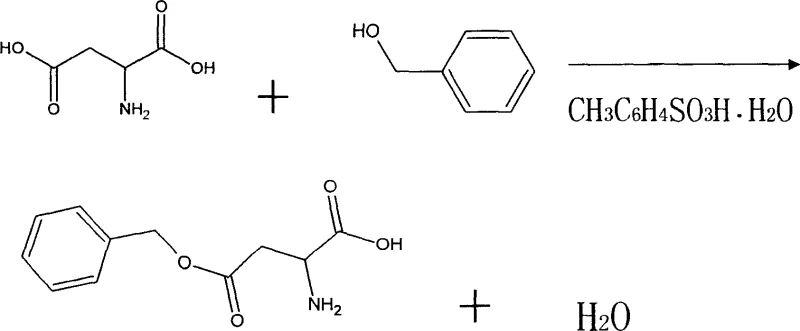
The landscape of peptide synthesis intermediates is undergoing a significant transformation driven by the need for safer, more scalable, and environmentally compliant manufacturing processes. A pivotal development in this sector is detailed in Chinese Patent CN1303057C, which outlines a robust synthetic method for L-aspartic acid-beta-benzyl ester, a critical building block for numerous bioactive polypeptides. This patent introduces a catalytic system that replaces traditional, hazardous strong acids with p-toluenesulfonic acid hydrate, fundamentally altering the risk profile and operational efficiency of the production line. For R&D directors and procurement specialists alike, understanding the nuances of this transition from sulfuric acid catalysis to aromatic sulfonic acid catalysis is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent global quality standards. The methodology described not only addresses the chemical challenges of esterifying amino acids but also solves significant downstream processing bottlenecks associated with solvent toxicity and waste management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-aspartic acid-beta-benzyl ester relied heavily on the use of concentrated sulfuric acid as the primary catalyst, a practice documented in earlier Japanese patents such as Showa 37-13976 and Showa 57-26658. While sulfuric acid is undeniably a potent dehydrating agent with strong acidic properties, its application in amino acid esterification presents severe drawbacks that compromise both product quality and operational safety. The intense oxidizing nature of sulfuric acid often leads to unavoidable side reactions, including sulfonation of the aromatic ring, carbonization of the organic backbone, and uncontrolled polymerization, all of which degrade the yield and purity of the final intermediate. Furthermore, the workup procedures associated with these legacy methods frequently necessitated the use of diethyl ether for recrystallization, a solvent notorious for its high volatility, extreme flammability, and significant health hazards, creating a dangerous environment for plant operators and complicating regulatory compliance regarding volatile organic compound (VOC) emissions.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach disclosed in the patent data leverages p-toluenesulfonic acid hydrate as a superior solid acid catalyst, offering a pathway that is both chemically selective and operationally benign. This method operates effectively within a moderate temperature range of 25°C to 100°C, significantly reducing the thermal stress on the sensitive amino acid structure compared to the harsher conditions often required by mineral acids. By shifting the catalytic paradigm, the process eliminates the oxidative degradation pathways inherent to sulfuric acid, thereby preserving the integrity of the chiral center and ensuring high optical purity essential for downstream peptide coupling. Additionally, the replacement of ether with a combination of absolute ethanol and water for precipitation and recrystallization represents a major leap forward in green chemistry, drastically lowering the fire risk and toxicity profile of the manufacturing facility while simplifying solvent recovery systems.
Mechanistic Insights into p-Toluenesulfonic Acid Catalyzed Esterification
The core chemical transformation in this synthesis is a Fischer esterification, where the carboxylic acid group of L-aspartic acid reacts with benzyl alcohol to form the ester linkage, releasing water as a byproduct. The choice of p-toluenesulfonic acid (p-TsOH) as the catalyst is mechanistically profound because it acts as a soluble organic acid that provides protons to activate the carbonyl oxygen of the carboxylic acid without introducing nucleophilic counter-ions that could interfere with the reaction. Unlike inorganic acids, the bulky organic structure of the tosylate anion stabilizes the transition state effectively while remaining inert towards the amino group and the benzyl ring, thus preventing the sulfonation side reactions that plague sulfuric acid catalysis. The hydration state of the catalyst (monohydrate) also plays a subtle role in managing the water activity within the reaction matrix, allowing for a controlled equilibrium shift towards ester formation without requiring aggressive azeotropic distillation setups that can be energy-intensive.
Impurity control in this system is achieved through a precise stoichiometric balance and a gentle neutralization strategy. The patent specifies a molar ratio of L-aspartic acid to benzyl alcohol ranging from 1.0:1.3 to 1.0:13.0, with a preferred range of 1.0:5.5 to 1.0:6.5, ensuring that the alcohol is in sufficient excess to drive the equilibrium forward without creating an unmanageable volume of waste solvent. Following the reaction, the addition of absolute ethanol serves to reduce the solubility of the product, inducing precipitation, while the subsequent neutralization with saturated sodium carbonate solution adjusts the pH to exactly 7. This specific pH control is critical; it ensures that the free amino group remains unprotonated enough to allow precipitation but prevents the hydrolysis of the newly formed ester bond, which can occur under strongly alkaline conditions. This delicate balance results in a crude precipitate that is already of high purity, requiring only a simple water recrystallization to achieve pharmaceutical grade specifications.
How to Synthesize L-Aspartic Acid-beta-Benzyl Ester Efficiently
Implementing this synthesis route on a commercial scale requires strict adherence to the optimized parameters defined in the patent to ensure consistent batch-to-batch reproducibility and maximum yield. The process begins with the careful charging of the reactor with benzyl alcohol, L-aspartic acid, and the p-toluenesulfonic acid hydrate catalyst, followed by a controlled heating phase that maintains the reaction mixture between 25°C and 100°C for a duration of 1 to 3 hours. Detailed standardized operating procedures regarding mixing speeds, addition rates of the neutralizing agent, and filtration temperatures are critical to replicating the reported yields of approximately 40% to 47% observed in the experimental embodiments. For a comprehensive breakdown of the exact equipment setup and safety protocols required for this specific transformation, please refer to the technical guide below.
- Charge the reactor with benzyl alcohol, L-aspartic acid, and p-toluenesulfonic acid hydrate catalyst, maintaining a molar ratio optimized for yield.
- Heat the mixture to between 25°C and 100°C and stir for 1 to 3 hours to facilitate the esterification reaction while minimizing side products.
- Quench the reaction with absolute ethanol, neutralize to pH 7 using saturated sodium carbonate, and purify the precipitate via water recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this p-toluenesulfonic acid catalyzed route offers tangible benefits that extend far beyond simple chemical yield, impacting the total cost of ownership and supply continuity. The elimination of sulfuric acid removes the need for specialized corrosion-resistant reactors and extensive neutralization waste streams, directly translating to reduced capital expenditure on maintenance and lower costs associated with hazardous waste disposal. Furthermore, the substitution of diethyl ether with ethanol and water significantly mitigates the insurance premiums and safety infrastructure costs associated with handling highly flammable solvents, creating a more resilient and cost-effective manufacturing environment that is less susceptible to regulatory shutdowns due to safety violations.
- Cost Reduction in Manufacturing: The shift to a solid acid catalyst like p-toluenesulfonic acid hydrate simplifies the dosing and handling processes, as it avoids the complexities of pumping and storing corrosive liquid mineral acids. This operational simplification reduces labor hours and minimizes the risk of accidental spills that could lead to costly production stoppages or environmental fines. Additionally, the ability to use water as the primary recrystallization solvent drastically cuts down on raw material costs compared to purchasing and recovering large volumes of organic solvents like ether, leading to substantial overall savings in the variable cost structure of the intermediate.
- Enhanced Supply Chain Reliability: By utilizing widely available and stable reagents such as benzyl alcohol and p-toluenesulfonic acid, the supply chain becomes less vulnerable to the fluctuations and shortages often seen with specialized or heavily regulated hazardous chemicals. The robustness of the reaction conditions, which tolerate a broad temperature range and do not require inert atmospheres or ultra-dry conditions, ensures that production can continue reliably even under varying utility conditions, guaranteeing consistent delivery schedules for downstream peptide manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids exothermic runaways common with sulfuric acid dilution and uses solvents that are easily treated in standard wastewater facilities. The absence of halogenated solvents and the reduction of toxic VOC emissions align perfectly with modern environmental, social, and governance (ESG) goals, making the facility more attractive to international partners who require their suppliers to adhere to strict sustainability mandates. This compliance advantage future-proofs the production line against tightening environmental regulations, ensuring long-term operational viability without the need for costly retrofits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of L-aspartic acid-beta-benzyl ester, derived directly from the comparative analysis of the patented technology and industry standards. These insights are intended to clarify the specific advantages of the p-toluenesulfonic acid route for stakeholders evaluating potential suppliers or considering technology transfers. Understanding these distinctions is vital for making informed decisions about raw material sourcing and process validation.
Q: Why is p-toluenesulfonic acid preferred over sulfuric acid for this esterification?
A: Unlike sulfuric acid, p-toluenesulfonic acid hydrate lacks strong oxidizing properties, thereby preventing undesirable side reactions such as sulfonation, carbonization, or polymerization of the amino acid substrate.
Q: What are the safety advantages of the recrystallization solvent used in this method?
A: This process utilizes water and ethanol for purification, eliminating the need for diethyl ether, which is highly volatile, toxic, and poses significant explosion hazards in large-scale manufacturing environments.
Q: How does this synthesis route impact the purity of the final polypeptide intermediate?
A: By avoiding harsh oxidative conditions and utilizing a mild neutralization step with sodium carbonate, the method ensures high optical purity and minimizes racemization or degradation of the chiral center.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Aspartic Acid-beta-Benzyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final therapeutic product is inextricably linked to the purity and consistency of your starting materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN1303057C is executed with precision at every scale. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of L-aspartic acid-beta-benzyl ester meets the exacting standards required for GMP peptide synthesis.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can enhance your supply chain efficiency and product quality. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the competitive pharmaceutical market.
