Scalable Synthesis of Novel Tetracyclic Diene Liquid Crystals for High-Performance Display Applications
Scalable Synthesis of Novel Tetracyclic Diene Liquid Crystals for High-Performance Display Applications
The rapid evolution of the flat-panel display industry, driven by the ubiquitous adoption of LCD TVs, notebooks, and mobile devices, has placed stringent demands on the performance metrics of liquid crystal materials. Patent CN102134183B introduces a breakthrough class of novel tetracyclic diene liquid crystal compounds designed to address critical limitations in existing formulations, such as operating voltage, viscosity, and response speed. As a leading provider in the fine chemical sector, we recognize that the structural integrity of these molecules—characterized by a unique four-ring backbone and conjugated diene systems—is paramount for achieving the high clearing points and large elastic constants required for next-generation STN and TFT displays. This technical insight report analyzes the proprietary synthesis route disclosed in the patent, highlighting its potential for commercial scale-up and its strategic value for procurement teams seeking reliable electronic chemical suppliers.
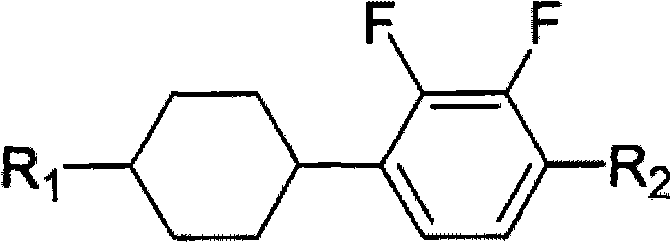
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional liquid crystal materials often rely on simpler biphenyl or phenyl-cyclohexane structures which, while stable, frequently suffer from insufficient dielectric anisotropy or excessively high viscosity. In the context of high-speed switching displays, high viscosity translates directly to slower response times, a critical bottleneck for modern multimedia applications. Furthermore, conventional synthetic routes for complex polycyclic mesogens often involve harsh reaction conditions, the use of expensive transition metal catalysts that require rigorous removal to meet electronic grade purity standards, or multi-step sequences with poor overall yields. These factors contribute to volatile supply chains and inflated manufacturing costs, creating significant friction for procurement managers aiming for cost reduction in display material manufacturing.
The Novel Approach
The methodology outlined in CN102134183B offers a sophisticated yet pragmatic solution by leveraging a convergent synthesis strategy centered on the Wittig reaction. This approach allows for the precise construction of the carbon-carbon double bonds essential for the diene functionality while maintaining the rigid rod-like geometry necessary for liquid crystallinity. By utilizing readily available starting materials such as substituted benzyl ring hexanones and protected dicyclohexanediones, the process circumvents the need for exotic precursors. The strategic inclusion of an isomerization step ensures the exclusive formation of the trans-configuration, which is thermodynamically more stable and exhibits superior mesomorphic properties compared to its cis-counterparts. This robust chemical design not only enhances the physical performance of the final mixture but also streamlines the production workflow.
Mechanistic Insights into Wittig Olefination and Isomerization
The core of this synthesis lies in the meticulous control of stereochemistry during the olefination steps. The formation of the double bond is achieved through the reaction of a phosphonium ylide with a carbonyl compound. In the preparation of Intermediate A, for instance, a substituted phenyl cyclohexyl ketone reacts with a chloromethyl ether phosphonium salt. The mechanism proceeds through the formation of a betaine intermediate which collapses into an oxaphosphetane ring, subsequently decomposing to yield the alkene and triphenylphosphine oxide. Crucially, the initial Wittig product may exist as a mixture of cis and trans isomers. To rectify this, the process employs a base-catalyzed isomerization using potassium hydroxide in methanol at controlled temperatures below 10°C. This thermodynamic equilibration drives the equilibrium towards the more stable trans-isomer, which is essential for the linear molecular shape required in nematic phases.
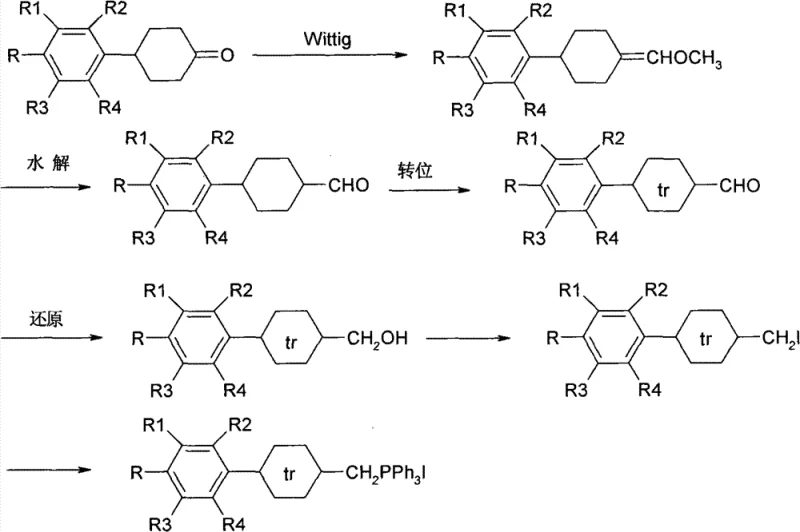
Furthermore, the impurity profile is tightly managed through the use of protecting groups, specifically the neopentyl glycol acetal moiety used in the synthesis of Intermediate B. This protecting group masks the ketone functionality during the initial chain extension, preventing unwanted side reactions such as self-condensation or over-alkylation. The subsequent deprotection under acidic conditions reveals the aldehyde functionality precisely when needed for the final coupling. This level of chemoselectivity is vital for R&D directors focused on purity and杂质谱 (impurity profiles), as it minimizes the formation of homologous impurities that are notoriously difficult to separate via crystallization. The final coupling of Intermediate A and Intermediate B via a second Wittig reaction, followed by a final isomerization in refluxing toluene with sodium benzenesulfinate, ensures the complete establishment of the conjugated diene system with the correct stereochemistry.
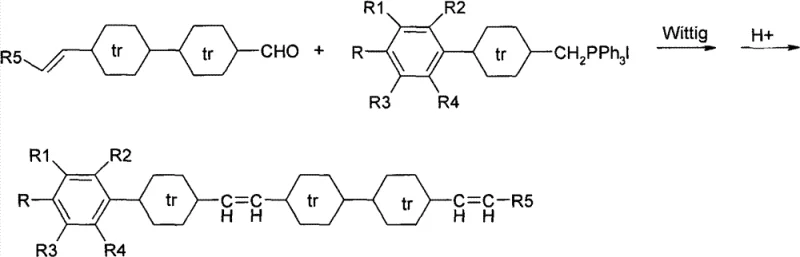
How to Synthesize Tetracyclic Diene Liquid Crystal Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these high-value intermediates. It begins with the preparation of two distinct fragments: a phosphonium salt derived from a fluorinated or alkyl-substituted phenyl cyclohexane, and an aldehyde derived from a bicyclic cyclohexane system. The convergence of these fragments allows for modular variation of the terminal groups (R and R5), enabling the fine-tuning of physical properties such as birefringence and viscosity. The detailed standardized synthesis steps, including specific molar ratios, solvent systems like tetrahydrofuran and dichloromethane, and precise temperature controls, are outlined below to guide process development teams in replicating this high-yield route.
- Preparation of Intermediate A: Synthesize trans-substituted phenyl cyclohexyl methyl iodide triphenyl phosphine salt via Wittig reaction, hydrolysis, isomerization, reduction, and iodination.
- Preparation of Intermediate B: Convert single neopentyl glycol contracting-4,4'-dicyclo hexanedione into vinyl dicyclohexyl formaldehyde through protection, Wittig reaction, and deprotection.
- Final Coupling: Connect Intermediate A and Intermediate B via Wittig reaction followed by double bond translocation to obtain the final trans-tetracyclic diene product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical performance. The reliance on commodity chemicals such as cyclohexanone derivatives and triphenylphosphine ensures a stable and resilient supply base, mitigating the risks associated with sourcing specialized organometallic reagents. For procurement managers, this translates to reduced exposure to price volatility and shorter lead times for raw material acquisition. The process is designed to be operationally simple, avoiding the need for extreme pressures or cryogenic conditions that would otherwise necessitate specialized reactor infrastructure. This operational simplicity is a key driver for cost reduction in electronic chemical manufacturing, as it lowers both capital expenditure (CAPEX) and operating expenditure (OPEX) associated with production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, which are often required in cross-coupling reactions like Suzuki or Heck couplings, significantly reduces the raw material cost burden. Additionally, the avoidance of heavy metals simplifies the downstream purification process, removing the need for costly scavenging resins or extensive washing protocols to meet ppm-level metal specifications. The high yields reported in the embodiments, such as the 97% yield in the initial Wittig step and 85.4% in the subsequent hydrolysis, indicate a highly atom-economical process that minimizes waste disposal costs and maximizes output per batch.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes robust chemistry that is less sensitive to trace moisture or oxygen compared to organolithium or Grignard-based alternatives. This robustness enhances batch-to-b consistency, a critical factor for supply chain heads who prioritize continuity of supply. The ability to synthesize the key intermediates in large quantities with high purity (>98% in several steps) ensures that downstream formulation houses receive consistent quality material, reducing the risk of production stoppages due to off-spec ingredients. The modular nature of the synthesis also allows for flexible production scheduling, where different R-group variants can be produced on the same equipment train with minimal changeover time.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions, with most reactions occurring between 0°C and room temperature, except for the final isomerization which uses refluxing toluene. This thermal profile is easily manageable in standard glass-lined or stainless steel reactors, facilitating seamless scale-up from pilot plant to commercial tonnage. Furthermore, the solvents used, primarily THF, methanol, and dichloromethane, are well-understood and can be efficiently recovered and recycled, aligning with modern green chemistry principles and environmental regulations. The absence of highly toxic reagents reduces the regulatory burden and safety risks associated with handling hazardous materials, contributing to a safer and more sustainable manufacturing environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetracyclic diene liquid crystals. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating these materials into existing liquid crystal mixtures.
Q: What are the key advantages of this tetracyclic diene structure for LCD applications?
A: The novel tetracyclic diene structure combines the advantages of alkene and cyclohexane liquid crystals, offering a wide working temperature range, high clearing point, excellent chemical stability, and improved elastic constants (K33/K11) suitable for STN and TFT displays.
Q: How does the synthesis method ensure high trans-isomer purity?
A: The process utilizes a specific isomerization step under alkaline conditions (potassium hydroxide/methanol system) at controlled low temperatures (below 10°C) to convert cis-alkene mixtures into the thermodynamically stable trans-configuration, ensuring high optical purity.
Q: Is this manufacturing process scalable for industrial production?
A: Yes, the method uses cheap and readily available starting materials like substituted benzyl ring hexanone and avoids toxic or irritating reagents where possible. The reaction conditions are mild and controllable, making it highly suitable for large-scale industrial suitability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetracyclic Diene Liquid Crystal Supplier
The commercialization of advanced display technologies relies heavily on the availability of high-performance liquid crystal monomers with precise physical properties. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in organic synthesis to deliver complex intermediates like the tetracyclic dienes described in CN102134183B. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global display manufacturers. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for electronic applications, including low ion content and high chemical stability.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific formulation needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of new molecular structures, our experts are ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality and efficiency can drive value for your organization.
