Advanced Pentafluoropropylene Ether Liquid Crystals: Technical Breakthroughs and Commercial Scalability
Introduction to Next-Generation Liquid Crystal Materials
The rapid evolution of display technology, particularly in the realm of Thin Film Transistor Liquid Crystal Displays (TFT-LCDs), demands materials that can simultaneously offer high response speeds, low operating voltages, and exceptional stability. Patent CN103756688A introduces a groundbreaking class of pentafluoropropylene ether liquid crystal compounds that address these critical performance bottlenecks. Unlike traditional biphenyl-based liquid crystals which often suffer from limited dielectric anisotropy and slower response times, these novel compounds incorporate a unique pentafluoropropylene ether bridge. This structural modification not only disrupts the conjugation of the biphenyl system to lower melting points and enhance solubility but also significantly increases the dipole moment. For R&D directors and procurement specialists in the electronic chemical sector, this represents a pivotal shift towards materials that can enable thinner, more energy-efficient, and higher-resolution display panels without compromising on the charge retention rate essential for image quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of liquid crystal mixtures has been constrained by the inherent trade-offs between viscosity, dielectric anisotropy, and voltage holding ratio. Conventional compounds often relied on simple alkyl or alkoxy chains attached to rigid core structures, which resulted in high rotational viscosity and consequently slow response times. Furthermore, many existing fluorinated compounds struggled with maintaining high charge retention rates under prolonged UV exposure, leading to image sticking and reduced display lifespan. The synthesis of complex fluorinated intermediates was also notoriously difficult, often requiring harsh conditions that generated significant impurities, thereby driving up purification costs and reducing overall yield. These limitations created a substantial barrier for manufacturers aiming to produce next-generation displays that meet the rigorous standards of modern consumer electronics and automotive applications.
The Novel Approach
The innovative strategy outlined in the patent data leverages a sophisticated molecular architecture featuring a pentafluoropropylene ether linkage (-OCF2-CF=CF2). This specific functional group serves as a powerful electron-withdrawing unit that drastically enhances the dielectric anisotropy (Δε) of the molecule, allowing for lower threshold voltages (V10) in the final display module. By strategically placing fluorine atoms on the phenyl rings and utilizing the ether bridge, the inventors have successfully decoupled the relationship between viscosity and dielectric properties. The result is a compound that maintains fluidity for fast switching while providing the strong dipole interactions necessary for efficient electric field alignment. This approach effectively overcomes the sluggish response and high voltage requirements that plagued earlier generations of liquid crystal materials, offering a clear pathway for cost reduction in electronic chemical manufacturing through improved device efficiency.
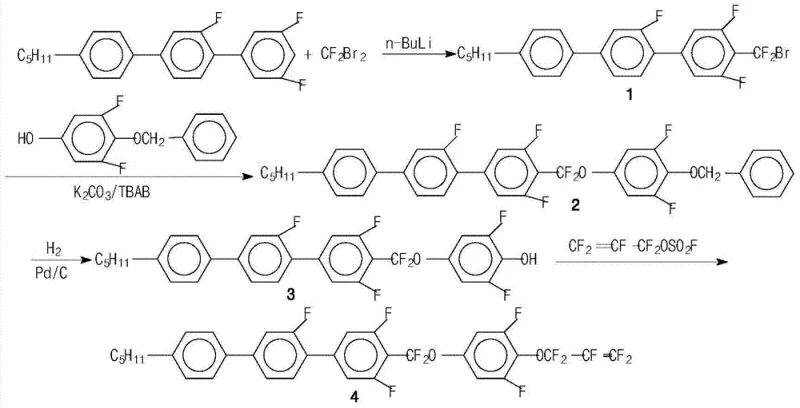
Mechanistic Insights into the Four-Step Synthesis Strategy
The synthesis of these high-performance liquid crystal monomers relies on a precise four-step sequence that balances reactivity with selectivity to ensure high purity. The process initiates with a low-temperature lithiation reaction, where the precursor is treated with n-butyllithium (n-BuLi) in tetrahydrofuran (THF) at temperatures ranging from -75°C to -85°C. This cryogenic condition is critical for generating the reactive lithium species without inducing side reactions or decomposition. Subsequently, difluorodibromomethane (CF2Br2) is introduced to install the bromodifluoromethyl group, a key building block for the subsequent ether linkage. The control of temperature during this exothermic addition is paramount, as the reaction mixture is allowed to warm naturally to -30°C before quenching, ensuring complete conversion while minimizing the formation of polymeric byproducts that could complicate downstream purification.
Following the initial functionalization, the route proceeds through a phase-transfer catalyzed etherification and a selective hydrogenation step. The bromodifluoromethyl intermediate reacts with a benzyloxy-protected phenol in the presence of potassium carbonate and tetrabutylammonium bromide (TBAB). This phase-transfer system facilitates the reaction between the organic halide and the phenolic salt in an aqueous-organic interface, significantly accelerating the rate of ether bond formation. The resulting protected intermediate is then subjected to catalytic hydrogenation using palladium on carbon (Pd/C) at mild temperatures of 20-30°C. This step cleanly removes the benzyl protecting group to reveal the free phenol without affecting the sensitive fluorinated olefinic bonds elsewhere in the molecule. The final step involves reacting this phenol with pentafluoropropenesulfonyl fluoride under basic conditions, locking in the terminal pentafluoropropenyl ether moiety that defines the material's superior electro-optical properties.
How to Synthesize Pentafluoropropylene Ether Liquid Crystal Efficiently
The preparation of these advanced electronic chemicals requires strict adherence to the patented protocol to achieve the reported yields and purity levels. The synthesis is designed to be scalable, utilizing common solvents like THF, toluene, and DMF, which simplifies solvent recovery and waste management in a commercial setting. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide process engineers in replicating this high-value chemistry.
- Perform lithiation of the precursor at -75°C to -85°C using n-BuLi, followed by reaction with CF2Br2 to form the bromodifluoromethyl intermediate.
- Conduct etherification with a benzyloxy-protected phenol using potassium carbonate and TBAB as a phase transfer catalyst under reflux conditions.
- Execute catalytic hydrogenation using Pd/C in a toluene/ethanol mixture at 20-30°C to remove the benzyl protecting group.
- Complete the synthesis by reacting the phenol intermediate with pentafluoropropenesulfonyl fluoride in DMF with triethylamine at 10-20°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits regarding cost stability and supply continuity. The high yields reported in the patent examples, such as 97.3% in the lithiation step and 84.5% in the final coupling, indicate a highly efficient process that minimizes raw material waste. This efficiency translates directly into a more predictable cost structure, as less starting material is required to produce a given mass of the final active pharmaceutical ingredient or electronic chemical. Furthermore, the use of robust, well-understood reaction types like lithiation and hydrogenation means that the process can be easily transferred to existing multipurpose reactors without the need for specialized or exotic equipment, thereby reducing capital expenditure barriers for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts in the final coupling steps, relying instead on readily available bases like triethylamine and potassium carbonate. This substitution significantly lowers the cost of goods sold (COGS) by removing the necessity for costly metal scavenging processes often required to meet stringent ppm limits in electronic grade materials. Additionally, the high selectivity of the reactions reduces the burden on purification units, allowing for simpler distillation or crystallization protocols that consume less energy and time compared to complex chromatographic separations typically associated with fluorinated compounds.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted biphenyls and fluorinated reagents like CF2Br2, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, particularly the tolerance of the hydrogenation step to mild temperatures, ensures consistent batch-to-batch quality even when scaling up from pilot plants to full commercial production. This reliability is crucial for maintaining the continuous supply lines required by major display panel manufacturers who operate on just-in-time inventory models and cannot afford interruptions due to synthesis failures.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the byproducts are primarily inorganic salts like lithium bromide and potassium fluoride, which are easier to treat and dispose of compared to heavy metal waste streams. The use of standard organic solvents allows for established recycling loops, aligning with increasingly strict environmental regulations in the chemical industry. The scalability is further evidenced by the successful execution of the reactions in multi-liter setups described in the examples, demonstrating that the heat transfer and mixing requirements are manageable at larger scales, facilitating a smoother transition from laboratory discovery to industrial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pentafluoropropylene ether liquid crystals. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: What are the key performance advantages of these pentafluoropropylene ether liquid crystals?
A: According to patent CN103756688A, these compounds exhibit high charge retention rates, large dielectric anisotropy, and improved mutual solubility. When applied to liquid crystal compositions, they significantly reduce threshold voltage and increase the Δn value, leading to faster response speeds in TFT-LCD displays.
Q: How is the purity of the final liquid crystal monomer controlled during synthesis?
A: The synthesis protocol emphasizes rigorous purification at each stage. The final step involves acidification, separation, drying, column chromatography, and recrystallization. Example 1 in the patent reports achieving a final purity of 99.7% for the target compound, ensuring minimal ionic impurities that could affect display performance.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes standard organic synthesis techniques such as lithiation, phase-transfer catalysis, and hydrogenation which are well-established in the fine chemical industry. The reported yields are consistently high (e.g., 97.3% in the first step, 84.5% in the final step), indicating a robust pathway amenable to scale-up from kilogram to tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pentafluoropropylene Ether Liquid Crystal Supplier
As the demand for high-performance display materials continues to surge, partnering with an experienced chemical manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and optical property testing, which are critical for electronic grade liquid crystals. We understand the nuances of handling fluorinated intermediates and the strict safety protocols required for lithiation and hydrogenation processes, guaranteeing a safe and compliant production environment.
We invite you to collaborate with our technical team to explore how these advanced liquid crystal monomers can enhance your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the electronic materials sector.
