Revolutionizing Alpha-Quaternary Carbon Synthesis: A Scalable Route for High-Value Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust methodologies to construct sterically hindered quaternary carbon centers, a structural motif prevalent in numerous bioactive natural products and synthetic drugs. Patent CN102295582A introduces a groundbreaking preparation method for alpha-position quaternary carbon alpha,beta-diamino acid derivatives, addressing a long-standing challenge in asymmetric synthesis. This technology leverages a sophisticated three-component reaction involving diazo compounds, alkoxyamides, and imines, catalyzed by a dual system of rhodium acetate and chiral phosphoric acids. The significance of this innovation lies in its ability to access all four stereoisomers—(2R,3R)-trans, (2R,3S)-cis, (2S,3S)-trans, and (2S,3R)-cis—with exceptional stereocontrol. For research and development teams, this represents a paradigm shift from multi-step sequences to a concise, one-pot transformation that delivers high optical purity. The versatility of this approach not only accelerates lead optimization but also provides a reliable pathway for generating diverse compound libraries essential for modern drug discovery programs targeting oncology and other therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active alpha,beta-diamino acid derivatives has relied heavily on the Mannich reaction between glycine esters and imines. While effective for certain substrates, this conventional approach suffers from severe limitations regarding stereochemical diversity. The most critical defect is the inherent difficulty in selectively obtaining trans-configured derivatives; traditional methods predominantly yield cis-isomers, thereby restricting the chemical space available for biological evaluation. Furthermore, existing literature indicates that synthesizing optical active alpha-position quaternary carbon derivatives is particularly arduous, often requiring harsh reaction conditions, excessive catalyst loading, and substrates with very narrow applicability. These constraints result in low atom economy and generate significant waste, posing challenges for both process chemistry and environmental compliance. Consequently, the inability to efficiently access both cis and trans isomers has hindered the comprehensive structure-activity relationship studies required to develop next-generation therapeutics with improved efficacy and safety profiles.
The Novel Approach
In stark contrast to legacy techniques, the methodology disclosed in CN102295582A offers a highly flexible and efficient solution through a rhodium-catalyzed three-component coupling. By employing different substituted chiral phosphoric acids in conjunction with an acid additive like L-tartaric acid, chemists can precisely tune the diastereoselectivity to favor either cis or trans configurations on demand. This level of control is achieved under mild conditions, typically at -20°C in toluene, utilizing molecular sieves to manage water content and drive the equilibrium forward. The reaction exhibits remarkable substrate tolerance, accommodating various aryl groups on the imine and diazo components, including electron-withdrawing and electron-donating substituents. 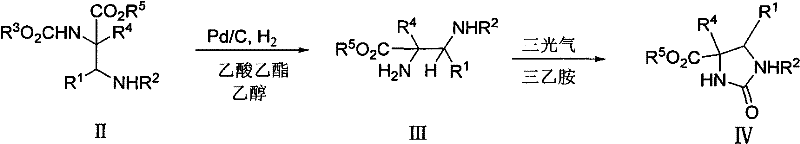 Moreover, the resulting alpha,beta-diamino acid derivatives serve as versatile building blocks for constructing imidazoline heterocycles, which are known for their potent anticancer and anti-arthritic properties. This strategic advantage transforms a complex synthetic problem into a streamlined operation, significantly enhancing the speed at which new drug candidates can be advanced from the bench to preclinical evaluation.
Moreover, the resulting alpha,beta-diamino acid derivatives serve as versatile building blocks for constructing imidazoline heterocycles, which are known for their potent anticancer and anti-arthritic properties. This strategic advantage transforms a complex synthetic problem into a streamlined operation, significantly enhancing the speed at which new drug candidates can be advanced from the bench to preclinical evaluation.
Mechanistic Insights into Rhodium and Chiral Phosphoric Acid Co-Catalysis
The success of this transformation hinges on the synergistic interaction between the rhodium catalyst and the chiral phosphoric acid. The reaction initiates with the decomposition of the diazo compound by the rhodium acetate to generate a reactive metal-carbene intermediate. This electrophilic species then undergoes nucleophilic attack by the alkoxyamide, forming an ylide or similar activated intermediate. Simultaneously, the chiral phosphoric acid activates the imine substrate through hydrogen bonding interactions, organizing the transition state within a well-defined chiral pocket. 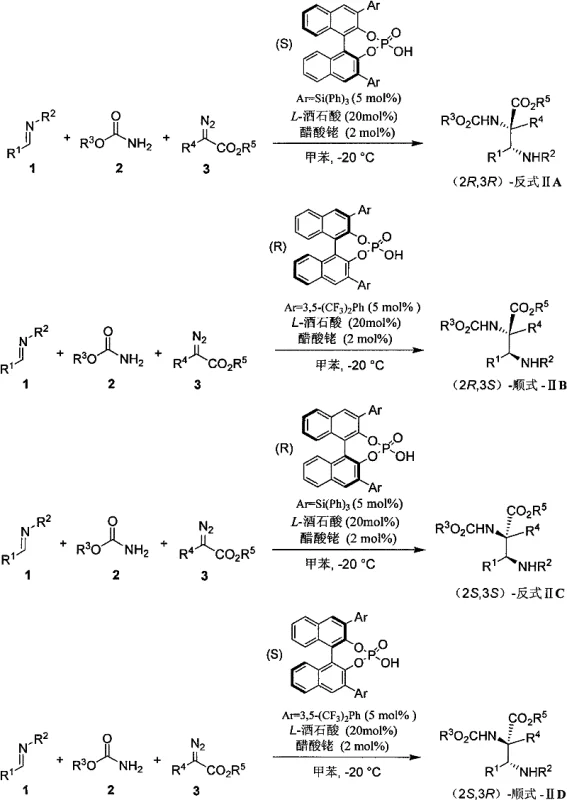 The specific stereochemical outcome—whether cis or trans—is dictated by the steric bulk and electronic properties of the 3,3'-substituents on the BINOL-derived phosphoric acid catalyst. For instance, bulky triphenylsilyl groups favor the formation of trans-isomers, while 3,5-bis(trifluoromethyl)phenyl groups promote cis-selectivity. This dual activation strategy ensures that the nucleophilic addition occurs with high facial selectivity, minimizing the formation of unwanted enantiomers. Understanding this mechanistic nuance is crucial for process optimization, as it allows for the rational selection of catalysts to match specific target molecules without the need for extensive empirical screening.
The specific stereochemical outcome—whether cis or trans—is dictated by the steric bulk and electronic properties of the 3,3'-substituents on the BINOL-derived phosphoric acid catalyst. For instance, bulky triphenylsilyl groups favor the formation of trans-isomers, while 3,5-bis(trifluoromethyl)phenyl groups promote cis-selectivity. This dual activation strategy ensures that the nucleophilic addition occurs with high facial selectivity, minimizing the formation of unwanted enantiomers. Understanding this mechanistic nuance is crucial for process optimization, as it allows for the rational selection of catalysts to match specific target molecules without the need for extensive empirical screening.
From an impurity control perspective, the use of molecular sieves plays a pivotal role in maintaining reaction integrity. Water can hydrolyze the diazo compound or deactivate the Lewis acidic rhodium center, leading to reduced yields and the formation of side products such as homodimers of the diazo species. By rigorously excluding moisture, the process ensures that the reactive carbene intermediate is consumed exclusively by the desired three-component pathway. Additionally, the mild temperature of -20°C suppresses non-catalytic background reactions that could erode enantiomeric excess. The high diastereomeric ratios observed, often exceeding 97:3, indicate that the kinetic control of the reaction is extremely effective. This precision in stereochemistry translates directly to simplified downstream purification, as the separation of diastereomers becomes trivial compared to resolving racemic mixtures, thereby reducing the overall processing time and solvent consumption associated with isolation.
How to Synthesize Alpha-Quaternary Carbon Diamino Acid Derivatives Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory-scale synthesis and potential industrial adoption. The procedure involves a straightforward sequence where the imine, alkoxyamide, catalysts, and drying agents are combined in a reactor prior to the controlled addition of the diazo component. Detailed standard operating procedures for this synthesis are provided in the guide below, ensuring reproducibility and safety during handling of energetic diazo materials.
- Prepare the reaction mixture by combining imine, alkoxyamide, chiral phosphoric acid catalyst, L-tartaric acid additive, and rhodium acetate in an organic solvent with molecular sieves.
- Cool the reaction system to -20°C and slowly add the diazo compound solution via a peristaltic pump over a period of 4 hours to control exothermicity and selectivity.
- After completion, remove the solvent via rotary evaporation and purify the crude product using column chromatography to isolate the optically active target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the economic implications of adopting this technology are substantial. The reliance on readily available starting materials such as aromatic aldehydes, amines, and standard diazo precursors eliminates the need for exotic or proprietary reagents that often bottleneck production schedules. This accessibility ensures a stable supply chain, reducing the risk of delays caused by raw material shortages. Furthermore, the one-step nature of the reaction drastically reduces the number of unit operations required compared to traditional multi-step syntheses. Fewer reaction steps mean less equipment occupancy time, lower labor costs, and reduced consumption of solvents and energy. The high atom economy of the three-component coupling ensures that a maximum proportion of the input mass is incorporated into the final product, aligning with green chemistry principles and minimizing waste disposal costs. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), making the final pharmaceutical intermediates more competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and the use of low-loading catalysts (as low as 2 mol%) directly lower material costs. By avoiding the need for resolution of racemic mixtures, which typically discards half of the produced material, this asymmetric synthesis maximizes yield efficiency. The ability to produce both cis and trans isomers from the same general platform avoids the capital expenditure of developing entirely separate process lines for different stereoisomers. This flexibility allows manufacturers to respond rapidly to changing market demands for specific enantiomers without retooling, providing a distinct financial advantage in contract manufacturing scenarios.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups, reduces the sensitivity of the process to minor variations in raw material quality. This tolerance minimizes batch failures and ensures consistent output quality, a critical metric for maintaining long-term supply agreements with major pharmaceutical clients. Additionally, the use of common organic solvents like toluene simplifies logistics and storage requirements compared to specialized or hazardous solvents. The scalability of the process from milligram to kilogram scales has been demonstrated through the variation of substrate scope, indicating that technology transfer to large-scale reactors can be achieved with minimal risk of performance degradation.
- Scalability and Environmental Compliance: The process adheres to strict environmental standards by minimizing waste generation through high selectivity and atom economy. The absence of heavy metal contaminants in the final product, due to the low catalyst loading and efficient workup, simplifies regulatory filing and quality control testing. This compliance reduces the administrative burden and time-to-market for new drug applications. Furthermore, the mild reaction temperatures reduce energy consumption for heating or extreme cooling, contributing to a lower carbon footprint for the manufacturing facility. These sustainability credentials are increasingly important for multinational corporations aiming to meet their corporate social responsibility goals while securing a reliable source of high-quality intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method for your specific production needs.
Q: What is the primary advantage of this three-component reaction over traditional Mannich reactions?
A: Unlike traditional Mannich reactions which are often limited to producing only cis-configured derivatives, this novel method allows for the tunable synthesis of both cis and trans optical isomers with high diastereoselectivity and enantioselectivity by simply changing the chiral phosphoric acid catalyst.
Q: Can this process be scaled for commercial production of anticancer intermediates?
A: Yes, the process utilizes commercially available raw materials such as diazo compounds and imines, operates under mild conditions (-20°C), and demonstrates high atom economy, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What are the biological applications of the synthesized alpha-quaternary carbon derivatives?
A: These derivatives serve as critical precursors for imidazoline heterocyclic compounds, which have demonstrated significant anticancer and anti-arthritic activities, including inhibition of human breast cancer cell lines like MDA-MB-468.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Quaternary Carbon Diamino Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing complex chiral building blocks for the development of advanced therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of alpha-quaternary carbon diamino acid derivatives meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for pharmaceutical companies seeking to secure their supply chain for next-generation anticancer agents.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your current pipeline. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits specific to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you accelerate your drug development timeline with our proven expertise in asymmetric catalysis and process optimization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
