Advanced Synthesis of Spiro Indole Compounds for High-Performance Chiral Catalysts
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex chiral scaffolds that serve as the backbone for next-generation asymmetric catalysts. Patent CN112442042A discloses a groundbreaking preparation method for spiro indole compounds, specifically targeting the synthesis of spiro indole diphenols with unique skeletal structures. This innovation addresses a critical gap in medicinal chemistry by providing a versatile platform for developing high-performance chiral ligands. Unlike traditional spiro diphenols which possess a pure carbon ring framework limiting their functionalization potential, this novel approach strategically incorporates nitrogen atoms and ortho-hydroxyl groups on the benzene rings. These structural features significantly enhance the molecule's capacity for diverse derivatization, allowing chemists to fine-tune steric and electronic properties for optimal catalytic performance in asymmetric hydrogenation and other transformations. The disclosed process is characterized by its operational simplicity, utilizing economically viable reagents and mild conditions that are highly conducive to industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spirocyclic compounds has often relied on frameworks composed entirely of carbon rings, which present significant challenges for downstream modification. The absence of heteroatoms or reactive functional groups in close proximity to the spiro center restricts the chemical space available for ligand optimization. Conventional routes frequently involve harsh reaction conditions, expensive transition metal catalysts, or multi-step sequences that suffer from low overall yields and poor atom economy. Furthermore, the purification of intermediates in traditional methods can be cumbersome, often requiring extensive chromatography which is impractical for large-scale manufacturing. The inability to easily introduce diversity at key positions on the aromatic rings means that screening for improved catalytic activity becomes a slow and resource-intensive process, hindering the rapid development of new chiral drugs and agrochemicals that rely on these sophisticated catalysts.
The Novel Approach
The methodology outlined in the patent offers a transformative solution by streamlining the construction of the spiro indole core through a sequence of highly efficient transformations. The process begins with the condensation of readily available aniline derivatives with malonic acid or malonyl chloride to form bis-amide precursors, followed by a strategic oxidation or acid treatment to activate the central bridge. A key innovation lies in the use of Eaton reagent or similar dehydrating agents to effect the intramolecular cyclization, forging the challenging spiro carbon-carbon bond under relatively mild thermal conditions. This approach not only simplifies the synthetic route but also ensures high regioselectivity. Subsequent deprotection steps reveal the critical ortho-hydroxyl groups, yielding the final spiro indole diphenol ready for resolution. By integrating nitrogen into the scaffold, the resulting compounds offer superior coordination capabilities for metal centers, making them ideal candidates for advanced asymmetric catalysis applications.
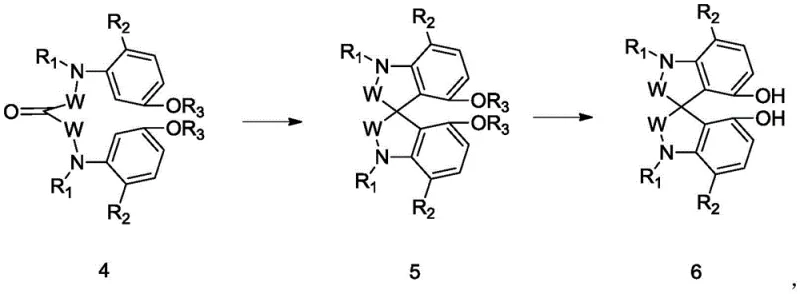
Mechanistic Insights into Eaton Reagent-Catalyzed Cyclization
The formation of the quaternary spiro center is the mechanistic cornerstone of this synthesis, driven by the powerful dehydrating capability of the Eaton reagent, which is a mixture of methanesulfonic acid and phosphorus pentoxide. In this acidic medium, the carbonyl oxygen of the precursor is protonated, increasing its electrophilicity and facilitating an intramolecular Friedel-Crafts-type alkylation onto the electron-rich aromatic ring. The presence of the methoxy groups on the benzene rings plays a crucial role in directing this cyclization by activating the ortho-positions towards electrophilic attack, ensuring that the spiro junction forms at the desired location with high fidelity. This acid-mediated cyclization avoids the need for expensive Lewis acids or transition metals, thereby reducing the risk of metal contamination in the final product, a critical parameter for pharmaceutical intermediates. The reaction proceeds through a stabilized carbocation intermediate, which is rapidly trapped by the aromatic nucleophile to close the ring, demonstrating excellent tolerance to various substituents on the nitrogen and aromatic rings.
Following the cyclization, the revelation of the active hydroxyl groups is achieved through demethylation, typically using boron tribromide. This step is mechanistically straightforward yet vital, as the free phenolic hydroxyls are essential for the subsequent coordination with metal centers in catalytic applications. The use of boron tribromide allows for selective cleavage of the methyl ethers without disturbing the sensitive spiro architecture or the amide functionalities. Impurity control is inherently managed by the crystallinity of the intermediates; for instance, the spiro ketone intermediates often precipitate from the reaction mixture upon workup, allowing for simple filtration to remove soluble byproducts. This inherent purity enhancement reduces the burden on downstream purification processes, ensuring that the final chiral ligands meet the stringent quality standards required for asymmetric synthesis in drug manufacturing.
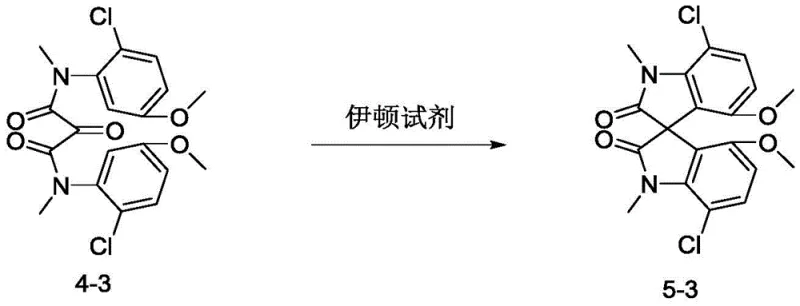
How to Synthesize Spiro Indole Diphenol Efficiently
The synthesis of these high-value intermediates follows a logical progression from simple anilines to the complex spiro architecture. The process is designed to maximize yield while minimizing waste, starting with the acylation of the amine to protect the nitrogen and set up the cyclization precursor. The subsequent oxidation or activation step prepares the molecule for the critical ring-closing event. Finally, the deprotection reveals the functional groups necessary for catalytic activity. For detailed operational parameters, stoichiometry, and specific workup procedures tailored to different substituents, please refer to the standardized guide below.
- Condense substituted aniline derivatives with malonyl chloride or malonic acid to form the bis-amide precursor (Compound 3a/3b).
- Oxidize the methylene bridge to a carbonyl group using chromium trioxide or treat with acid to prepare the cyclization substrate (Compound 4).
- Perform intramolecular cyclization using Eaton reagent to form the spiro skeleton (Compound 5), followed by demethylation with boron tribromide to yield the final diphenol (Compound 6).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic benefits that directly impact the bottom line and operational resilience. The reliance on commodity chemicals such as substituted anilines, malonyl chloride, and common solvents like dichloromethane and acetic acid ensures a stable and diversified supply base, mitigating the risks associated with sourcing exotic or single-source reagents. The elimination of precious metal catalysts in the key cyclization step removes a significant cost driver and simplifies the regulatory compliance landscape regarding heavy metal residues in the final API. Furthermore, the shortness of the synthetic sequence translates to reduced manufacturing lead times and lower inventory holding costs, allowing for a more agile response to market demands for chiral intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive dehydrating agents like Eaton reagent instead of costly transition metal complexes. The mild reaction conditions reduce energy consumption associated with heating and cooling, while the high selectivity minimizes the formation of difficult-to-separate byproducts, leading to higher overall yields and reduced raw material waste. Additionally, the ability to purify intermediates via simple crystallization or pulping rather than extensive column chromatography drastically lowers solvent usage and processing time, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: By employing robust chemistry that tolerates a wide range of substituents, manufacturers can maintain consistent production schedules even when specific raw material grades vary slightly. The scalability of the reaction, demonstrated by the successful execution of steps on multi-gram scales in the patent examples, indicates a smooth path to tonnage production without the need for specialized equipment. This reliability ensures a continuous flow of high-quality intermediates to downstream customers, preventing bottlenecks in the synthesis of final chiral drugs and securing the supply chain against disruptions.
- Scalability and Environmental Compliance: The synthetic route is inherently green by design, avoiding the generation of toxic heavy metal waste streams that require expensive disposal protocols. The use of recyclable solvents and the potential for solvent recovery systems aligns with modern environmental, social, and governance (ESG) goals. The straightforward workup procedures, involving aqueous quenches and phase separations, are easily adapted to large-scale reactors, ensuring that the process remains safe and manageable as production volumes increase from pilot plant to full commercial scale.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spiro indole compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for their own manufacturing pipelines.
Q: What is the primary advantage of this spiro indole synthesis method?
A: The method introduces nitrogen atoms and ortho-hydroxyl groups into the spiro framework, enabling diverse derivatization that is difficult with pure carbon ring skeletons, thus facilitating the screening of more effective chiral catalysts.
Q: Are the reaction conditions suitable for industrial scale-up?
A: Yes, the process utilizes readily available raw materials and mild reaction conditions, such as using Eaton reagent for cyclization and standard solvents like dichloromethane, which simplifies operation and lowers production costs for large-scale implementation.
Q: How is chirality controlled in the final spiro indole product?
A: The racemic spiro indole diphenol can be resolved using chemical resolving agents such as L-proline, quaternary ammonium quinine salts, or menthyl chloroformate to obtain products with single R or S configurations and high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Indole Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in complex organic transformations to deliver high-purity pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We understand the critical nature of chiral purity and structural integrity in catalyst precursors, which is why our facilities are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to verify stringent purity specifications for every batch we release.
We invite global partners to collaborate with us to optimize their supply chains and reduce time-to-market for their chiral drug candidates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the competitive landscape of asymmetric catalysis.
