Advanced Synthesis of Fluorinated Biphenyl Liquid Crystals for High-Performance Display Manufacturing
Advanced Synthesis of Fluorinated Biphenyl Liquid Crystals for High-Performance Display Manufacturing
The rapid evolution of the electronic display industry demands materials that can withstand rigorous operational conditions while delivering superior optical performance. Patent CN1800154A introduces a significant advancement in the field of organic synthesis, specifically targeting the production of substituted 4-biphenylcarboxylate liquid crystal compounds containing fluorine atoms and cyano groups. This specific chemical architecture, 4′-(3-methylbutoxy-1)-4-biphenylcarboxylic acid-2-fluoro-4-hydroxybenzonitrile ester, represents a critical component for next-generation liquid crystal displays (LCDs). The integration of fluorine and cyano functional groups into the biphenyl backbone is not merely a structural modification but a strategic engineering choice designed to enhance dielectric anisotropy and lower viscosity. For R&D Directors and Technical Procurement Managers, understanding the provenance of this technology is essential, as it offers a pathway to materials that meet the stringent requirements of wide operating temperature ranges and low driving voltages. The patent outlines a robust synthetic route that moves away from traditional, often hazardous methods, proposing a streamlined process that balances chemical efficiency with practical manufacturability. This report analyzes the technical depth of this innovation, providing a comprehensive view of its potential impact on the supply chain for high-purity electronic chemicals.
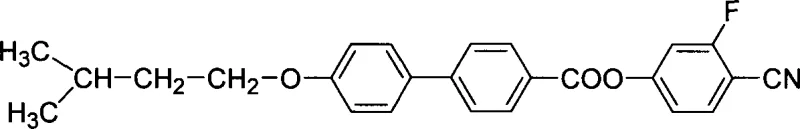
The limitations of conventional liquid crystal synthesis methods often revolve around the use of aggressive reagents and complex purification protocols that can compromise yield and purity. Traditional approaches to introducing alkoxy chains onto biphenyl cores frequently rely on anhydrous conditions that are sensitive to moisture, leading to inconsistent reaction outcomes and the formation of difficult-to-remove impurities. Furthermore, older esterification techniques might utilize acid chlorides, which generate corrosive by-products and require specialized handling equipment, increasing the overall cost of ownership for manufacturing facilities. In contrast, the novel approach detailed in the patent leverages a Williamson etherification reaction in a mixed ethanol-water solvent system. This shift allows for the use of soluble alkali metal hydroxides to maintain a pH greater than 10, creating a stable environment for the nucleophilic substitution of 1-halo-3-methylbutane onto the 4'-hydroxy-4-biphenylcarboxylic acid core. The ability to conduct this reaction at moderate temperatures between 50°C and 80°C significantly reduces energy consumption and thermal stress on the reactants. By avoiding extreme conditions, the novel approach minimizes side reactions such as elimination or polymerization, which are common pitfalls in high-temperature organic synthesis. This methodological refinement ensures that the intermediate 4′-(3-methylbutoxy-1)-4-biphenylcarboxylic acid is generated with a higher degree of structural integrity, setting a solid foundation for the subsequent coupling steps.
Mechanistic Insights into Williamson Etherification and DCC-Mediated Esterification
The core of this synthetic strategy lies in the precise control of reaction kinetics and thermodynamics during the etherification and coupling phases. The initial step involves the deprotonation of the phenolic hydroxyl group on the biphenyl carboxylic acid by the alkali metal hydroxide, generating a phenoxide ion that acts as a potent nucleophile. This ion attacks the electrophilic carbon of the 1-halo-3-methylbutane, displacing the halide ion and forming the ether linkage. The use of an ethanol-water mixture is particularly ingenious; it solubilizes both the organic substrate and the inorganic base, creating a homogeneous phase that accelerates the reaction rate without the need for expensive phase transfer catalysts. Following this, the patent describes a critical purification step where the crude reaction mixture is treated with additional alkali hydroxide solution. This hydrolysis step is designed to decompose any unreacted haloalkane or ester by-products that may have formed, ensuring that the isolated intermediate is chemically pure before proceeding. The final transformation utilizes N,N-dicyclohexylcarbodiimide (DCC) as a dehydrating agent and N,N-dimethylaminopyridine (DMAP) as a nucleophilic catalyst. This coupling mechanism activates the carboxylic acid group of the intermediate, allowing it to react efficiently with 2-fluoro-4-hydroxybenzonitrile. The reaction is conducted in anhydrous tetrahydrofuran at low temperatures ranging from 0°C to 30°C, which is crucial for preserving the stereochemical integrity and preventing the decomposition of the sensitive cyano and fluorine functionalities.
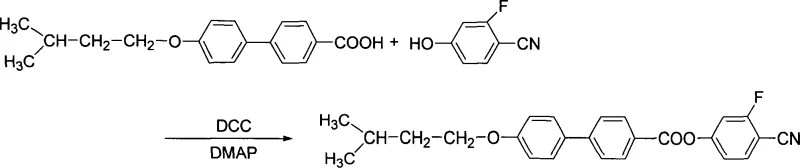
Impurity control is a paramount concern for R&D teams focusing on the performance of liquid crystal mixtures, as even trace contaminants can disrupt the alignment of liquid crystal molecules and degrade display quality. The synthetic route described in the patent incorporates multiple checkpoints to mitigate impurity formation. The hydrolysis step mentioned earlier serves as a chemical filter, removing species that do not possess the desired acid functionality. Furthermore, the use of DCC in the final step generates dicyclohexylurea as a by-product, which is insoluble in many organic solvents and can be easily removed by filtration. This physical separation method is far more efficient than chromatographic purification on a large scale, reducing solvent waste and processing time. The patent also specifies the use of column chromatography for final purification in the experimental examples, indicating a commitment to achieving the highest possible purity levels for characterization. For industrial applications, this suggests that the process is amenable to optimization where crystallization or recrystallization techniques could replace column chromatography to further enhance cost efficiency. The presence of the fluorine atom and the cyano group introduces specific electronic effects that must be preserved; the mild conditions of the DCC coupling ensure that these groups remain intact, preventing the formation of defluorinated or hydrolyzed nitrile impurities that would otherwise compromise the dielectric properties of the final material.
How to Synthesize 4'-(3-methylbutoxy-1)-4-biphenylcarboxylic Acid-2-fluoro-4-hydroxybenzonitrile Ester Efficiently
The synthesis of this advanced liquid crystal monomer requires a disciplined approach to reaction conditions and reagent stoichiometry to ensure reproducibility and high yield. The process begins with the preparation of the ether intermediate, where precise control of the pH and solvent composition is vital to drive the Williamson etherification to completion. Detailed standardized synthesis steps see the guide below.
- Perform Williamson etherification using 4'-hydroxy-4-biphenylcarboxylic acid and 1-halo-3-methylbutane in an ethanol-water solvent system with alkali metal hydroxide at 50-80°C.
- Purify the intermediate 4'-(3-methylbutoxy-1)-4-biphenylcarboxylic acid via hydrolysis with soluble alkali metal hydroxide to remove by-products.
- Conduct dehydration condensation using DCC and DMAP in anhydrous tetrahydrofuran with 2-fluoro-4-hydroxybenzonitrile at 0-30°C to yield the final ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route outlined in patent CN1800154A offers substantial advantages over legacy methods, primarily driven by the selection of reagents and the simplicity of the workup procedures. The reliance on ethanol-water mixtures for the initial etherification step significantly reduces the dependency on expensive, anhydrous organic solvents that require rigorous drying and storage protocols. This shift not only lowers the direct material costs but also simplifies the safety infrastructure required in the manufacturing plant, as ethanol is less hazardous than many chlorinated or aromatic solvents. The ability to operate at moderate temperatures between 50°C and 80°C further contributes to cost reduction in electronic chemical manufacturing by lowering energy consumption for heating and cooling systems. For Supply Chain Heads, the availability of starting materials such as 4'-hydroxy-4-biphenylcarboxylic acid and 2-fluoro-4-hydroxybenzonitrile is a critical factor; these are established intermediates in the fine chemical industry, ensuring a stable and reliable supply chain without the risk of bottlenecks associated with exotic reagents. The process design inherently supports scalability, as the unit operations involved—reflux, filtration, and crystallization—are standard in chemical engineering and can be easily transferred from pilot scale to commercial production volumes.
- Cost Reduction in Manufacturing: The elimination of harsh acid chlorides and the use of DCC coupling instead of traditional acid-catalyzed esterification reduces the need for corrosion-resistant equipment, leading to lower capital expenditure and maintenance costs. The by-product of the DCC reaction, dicyclohexylurea, is a solid that can be filtered off easily, simplifying the purification workflow and reducing the volume of solvent required for washing and extraction. This streamlined downstream processing translates directly into reduced operational expenses and shorter batch cycle times. Furthermore, the high selectivity of the Williamson etherification minimizes the formation of side products, which means less raw material is wasted on impurities that must be discarded. The overall atom economy of the process is optimized by the direct coupling strategy, avoiding multi-step protection and deprotection sequences that are common in older synthetic routes. These factors combine to create a manufacturing profile that is both economically efficient and environmentally friendlier, aligning with modern green chemistry principles.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis are commodity chemicals or widely available fine chemical intermediates, which mitigates the risk of supply disruptions. Unlike processes that rely on specialized catalysts or rare earth metals, this route uses alkali metal hydroxides and organic bases like DMAP, which are readily sourced from multiple global suppliers. This diversification of the supply base ensures that production can continue even if one vendor faces logistical challenges. The robustness of the reaction conditions, particularly the tolerance of the ethanol-water system, means that variations in raw material quality can be managed more effectively without compromising the final product specification. For Supply Chain Heads, this reliability is crucial for maintaining consistent delivery schedules to downstream display manufacturers. The ability to produce the compound with high purity using standard equipment also means that contract manufacturing organizations (CMOs) can adopt this process quickly, expanding the potential manufacturing network and reducing lead time for high-purity electronic chemicals.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are safe and manageable in large reactors. The use of ethanol, which can be recovered and recycled through distillation, reduces the environmental footprint of the manufacturing process and lowers waste disposal costs. The absence of heavy metal catalysts eliminates the need for complex metal scavenging steps and reduces the burden of hazardous waste treatment, ensuring compliance with strict environmental regulations. The mild reaction temperatures reduce the risk of thermal runaway incidents, enhancing plant safety and insurance profiles. As production scales from kilograms to tons, the efficiency of the filtration and crystallization steps becomes even more pronounced, offering a clear path to commercial scale-up of complex polymer additives and liquid crystal monomers. The process generates minimal aqueous waste compared to acid-heavy routes, simplifying wastewater treatment requirements. This environmental compatibility is increasingly important for suppliers aiming to partner with major electronics brands that have stringent sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this fluorinated liquid crystal compound, based on the detailed specifications and experimental data provided in the patent documentation. These answers are derived from the chemical principles and process descriptions to assist technical decision-makers in evaluating the feasibility of this technology for their specific applications.
Q: What are the thermal stability characteristics of this liquid crystal compound?
A: The compound exhibits a high clearing point and a wide nematic phase range, maintaining stability from approximately 95°C to over 200°C, which is critical for display reliability.
Q: How does the synthesis method impact impurity profiles?
A: The process includes a specific hydrolysis step using alkali metal hydroxides to remove etherification by-products, ensuring high chemical purity before the final coupling reaction.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the use of common solvents like ethanol-water and mild reaction temperatures between 50°C and 80°C facilitates safer and more scalable manufacturing operations compared to harsh alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-(3-methylbutoxy-1)-4-biphenylcarboxylic acid-2-fluoro-4-hydroxybenzonitrile ester Supplier
The technical potential of the synthesis route described in patent CN1800154A is immense, offering a pathway to high-performance liquid crystal materials that meet the demanding specifications of the modern display industry. NINGBO INNO PHARMCHEM, as a leading CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facilities are equipped with state-of-the-art reaction vessels capable of handling the specific solvent systems and temperature profiles outlined in the patent, ensuring that the transition from lab scale to mass production is seamless. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the critical optical and thermal standards required for LCD applications. Our team understands the nuances of fluorinated chemistry and the importance of impurity control in electronic materials, allowing us to optimize the process for maximum yield and consistency. By leveraging our infrastructure, clients can bypass the lengthy development phase and accelerate their time-to-market for new display technologies.
We invite procurement and R&D leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this synthesis can be integrated into your supply chain. By partnering with us, you gain access to a reliable source of advanced electronic chemicals that combine technical excellence with commercial viability. Contact us today to discuss how we can support your next-generation display projects with high-quality, scalable manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
