Advanced Manufacturing of Natural Carbazole Alkaloids via Regioselective Gold Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for accessing bioactive natural product scaffolds, particularly carbazole alkaloids known for their potent antitumor and antiviral properties. A significant breakthrough in this domain is detailed in Chinese Patent CN102424666A, which discloses a highly efficient, regioselective synthetic pathway for producing valuable targets such as Clauszoline-K, Clausine-N, and 3-Formyl-7-hydroxycarbazole. Unlike traditional methods that often suffer from harsh conditions or poor selectivity, this invention leverages a sophisticated gold-catalyzed cyclization strategy. By utilizing 1-benzyl-5-bromo-2-indolealdehyde and methoxy-1,2-propadiene as primary building blocks, the process constructs the complex carbazole core with exceptional precision. This technological advancement represents a pivotal shift towards more sustainable and controllable manufacturing of these high-value pharmaceutical intermediates, offering a reliable alternative to extraction from limited natural sources.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of carbazole alkaloids like Clausine-N and Clauszoline-K has been fraught with significant logistical and chemical challenges. Conventional approaches primarily relied on the extraction of these compounds from plant sources such as Murraya japonica or Clausena lansium. This biological sourcing is inherently unstable, subject to seasonal variations, geographical limitations, and extremely low natural abundance, which drives up costs and creates supply chain bottlenecks. Furthermore, chemical synthesis routes predating this innovation often required multi-step sequences involving hazardous oxidants, strong acids, or high-temperature conditions that compromised the integrity of sensitive functional groups. These traditional synthetic pathways frequently resulted in complex mixtures of regioisomers, necessitating difficult and yield-losing purification steps that are economically unviable for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
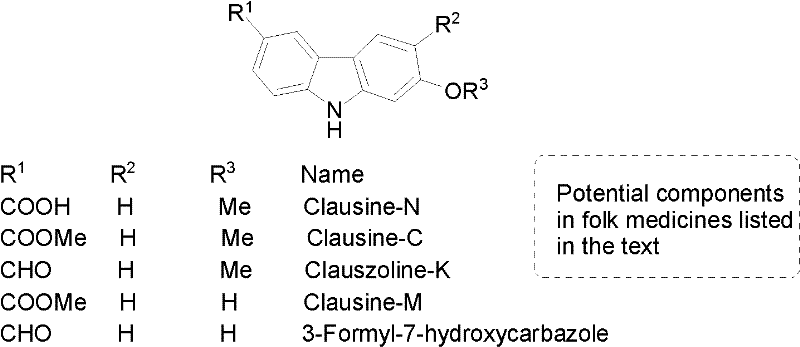
The methodology outlined in the patent data introduces a streamlined, convergent synthesis that fundamentally alters the efficiency landscape. The core innovation lies in the generation of an indole allenol intermediate, which subsequently undergoes a metal-catalyzed cycloisomerization to form the carbazole ring system. This approach bypasses the need for pre-functionalized carbazole starting materials, allowing for the construction of the heterocyclic core from simpler, more abundant indole derivatives. As illustrated in the overall reaction scheme below, the process demonstrates remarkable versatility, enabling the synthesis of multiple alkaloid variants through simple downstream modifications of a common intermediate. This modularity is crucial for manufacturers aiming to produce a diverse portfolio of high-purity carbazole derivatives without developing entirely new processes for each target molecule.
Mechanistic Insights into AuCl-Catalyzed Cyclization
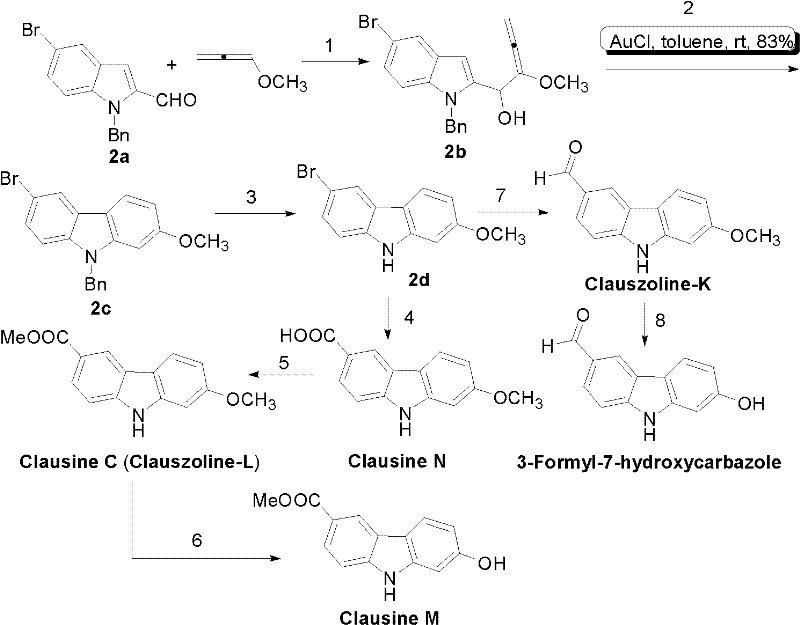
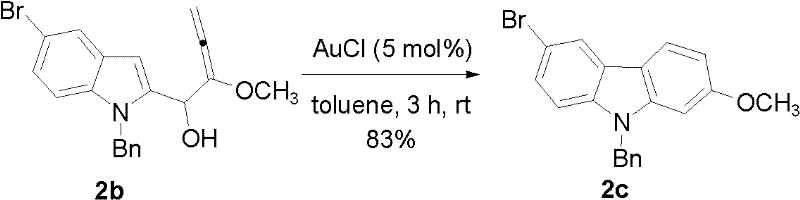
The heart of this synthetic strategy is the transition metal-catalyzed cyclization of the allenol intermediate. While the patent explores both Platinum (PtCl2) and Gold (AuCl) catalysts, the mechanistic superiority of the gold system is evident. The reaction initiates with the activation of the allene moiety by the soft Lewis acidic gold center. This activation facilitates a nucleophilic attack by the electron-rich indole ring, specifically at the C3 position, leading to the formation of the new C-C bond that closes the carbazole ring. The use of AuCl (5 mol%) in toluene at room temperature is particularly noteworthy, as it proceeds with rapid kinetics and high conversion rates. This mild activation mode prevents the degradation of sensitive substituents, such as the methoxy and bromo groups present on the scaffold, ensuring that the final product retains the necessary functionality for further derivatization into bioactive agents.
From an impurity control perspective, the high regioselectivity of this gold-catalyzed step is a major advantage for R&D teams focused on quality. Traditional electrophilic aromatic substitutions on carbazoles can often lead to mixtures of 1-, 3-, and 6-substituted isomers, which are notoriously difficult to separate. However, the intramolecular nature of this cyclization, directed by the tethered allenol side chain, locks the substitution pattern into the desired 3,7-disubstituted configuration (relative to the final numbering). This intrinsic selectivity minimizes the formation of structural impurities, thereby simplifying the purification workflow. The subsequent steps, such as the oxidative debenzylation using t-BuOK/DMSO/O2, are also designed to be chemoselective, removing the nitrogen protecting group without affecting the newly formed aromatic system or the oxygenated substituents, resulting in a clean crude profile ideal for commercial scale-up of complex natural product analogs.
How to Synthesize Clauszoline-K Efficiently
The synthesis of Clauszoline-K serves as an excellent case study for the practical application of this technology. The process begins with the lithiation of methoxy-1,2-propadiene followed by addition to the indole aldehyde to generate the allenol precursor. This intermediate is then subjected to the gold-catalyzed cyclization to form the protected carbazole core. Following the removal of the benzyl protecting group, a Vilsmeier-Haack type formylation (using DMF and t-BuLi) introduces the critical aldehyde functionality at the C3 position. The detailed standardized synthesis steps for this specific transformation, including precise stoichiometry, temperature profiles, and workup procedures, are provided in the guide below to assist technical teams in replicating these results.
- React 1-benzyl-5-bromo-2-indolealdehyde with methoxy-1,2-propadiene using n-BuLi to form the key allenol intermediate.
- Perform AuCl-catalyzed cyclization in toluene at room temperature to construct the carbazole core with high regioselectivity.
- Execute downstream functionalizations including debenzylation, carboxylation, or formylation to yield target alkaloids like Clausine-N.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction or legacy synthesis to this patented methodology offers substantial strategic benefits. The primary driver for value creation is the drastic simplification of the raw material basket. By relying on commodity chemicals like indole aldehydes and allenes rather than scarce plant extracts or highly specialized heterocycles, the process insulates the supply chain from agricultural volatility and geopolitical sourcing risks. Furthermore, the elimination of heavy oxidants and the use of catalytic amounts of gold, which can potentially be recovered, aligns with modern green chemistry principles, reducing waste disposal costs and environmental compliance burdens. This operational efficiency translates directly into a more resilient and cost-effective supply model for downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic viability of this route is underpinned by the high atom economy of the cyclization step and the avoidance of expensive stoichiometric oxidants. By achieving the core ring construction in a single catalytic step with high yield, the process significantly reduces the number of unit operations required. This consolidation of steps lowers energy consumption, solvent usage, and labor hours, driving down the overall cost of goods sold (COGS) without compromising on the quality of the pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Dependence on natural extraction subjects buyers to unpredictable lead times and batch-to-batch variability. In contrast, this fully synthetic route utilizes stable, commercially available starting materials that can be sourced from multiple global suppliers. This diversification of the supply base ensures continuous availability of critical intermediates like Clausine-N and Clauszoline-K, reducing lead time for high-purity carbazole alkaloids and preventing production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: The reaction conditions described, particularly the room temperature cyclization in toluene, are inherently safer and easier to scale than processes requiring cryogenic temperatures or high pressure. The mild nature of the chemistry reduces the risk of thermal runaways, facilitating a smoother technology transfer from laboratory to pilot and commercial plant scales. Additionally, the reduced generation of hazardous byproducts simplifies wastewater treatment and aligns with increasingly stringent environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this route for industrial production. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating these intermediates into their existing drug development pipelines.
Q: What is the primary advantage of using AuCl over PtCl2 in this carbazole synthesis?
A: According to the patent data, while both catalysts work, the AuCl-catalyzed reaction proceeds significantly faster and delivers higher yields compared to the PtCl2 variant, making it the preferred choice for efficient manufacturing.
Q: How does this method improve upon traditional extraction from plant sources?
A: Traditional extraction relies on scarce plant resources with low natural abundance. This synthetic route uses readily available chemical feedstocks, ensuring consistent supply continuity, higher purity, and independence from seasonal agricultural variations.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes mild reaction conditions, such as room temperature cyclization and standard solvents like toluene and THF, which avoids the need for extreme high-pressure or cryogenic equipment, facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clauszoline-K Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our technical team has thoroughly analyzed the potential of this gold-catalyzed pathway and is well-equipped to translate these laboratory findings into robust commercial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to market launch. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards for high-purity carbazole derivatives.
We invite you to collaborate with us to leverage this innovative chemistry for your specific needs. Whether you require custom synthesis of Clausine-N or large-scale supply of Clauszoline-K, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments that can optimize your supply chain and reduce your overall development costs.
