Advanced Synthesis of Eribulin Intermediates for Commercial Scale-Up and Cost Reduction
The global demand for advanced oncology therapeutics continues to drive innovation in the synthesis of complex marine natural product derivatives, specifically focusing on the production of Eribulin mesylate. Patent CN108689795B introduces a groundbreaking preparation method for a critical eribulin intermediate, addressing long-standing challenges in stereochemical control and process efficiency. This technology represents a significant leap forward for pharmaceutical manufacturers seeking to optimize their supply chains for high-value anticancer agents. By leveraging a novel sulfonate-based protecting group strategy, the invention circumvents the need for hazardous reagents and extreme low-temperature conditions that have historically plagued this synthetic pathway. The strategic implementation of this patent allows for a more robust and economically viable production model, ensuring consistent quality for high-purity pharmaceutical intermediates. As we analyze the technical specifics, it becomes clear that this methodology is not merely an academic exercise but a commercially actionable solution for reliable eribulin intermediate supplier networks aiming to secure long-term contracts with major drug developers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical chiral fragment known as Formula II has been dominated by routes established by academic institutions, such as the pioneering work from Harvard University. These conventional methods, while chemically elegant, suffer from severe drawbacks when translated to an industrial setting. The reliance on specialized reagents like B-I-9-BBN for iodine addition introduces prohibitive costs and supply chain vulnerabilities, as these materials are not readily available in bulk quantities. Furthermore, the necessity of maintaining reaction temperatures below -65°C during ester reduction imposes immense energy burdens and requires specialized cryogenic equipment, which drastically increases capital expenditure. The frequent manipulation of hydroxyl protecting groups throughout the linear sequence leads to yield erosion and complicates the purification process, resulting in a lower overall throughput. Such inefficiencies make the traditional Kishi route unsuitable for the commercial scale-up of complex pharmaceutical intermediates required to meet global market demand. The safety hazards associated with dangerous organometallic reagents further complicate regulatory compliance and operational risk management.
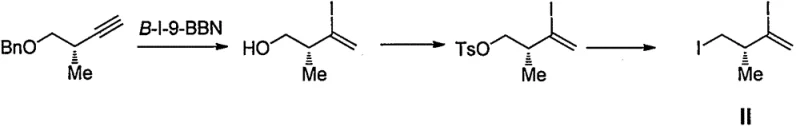
The Novel Approach
In stark contrast, the methodology disclosed in CN108689795B offers a streamlined and pragmatic alternative that directly addresses the bottlenecks of previous generations. The core innovation lies in the utilization of a substituted sulfonate as a hydroxyl protecting group, which serves a dual purpose by stabilizing the molecule and acting as a leaving group in subsequent steps. This strategic choice eliminates the need for repetitive protection and deprotection cycles, thereby shortening the synthetic timeline and improving material throughput. The adoption of the Corey-Fuchs reaction for alkyne generation provides a mild and reliable transformation that avoids the use of hazardous diazotization reagents. Moreover, the iodine addition step utilizes inexpensive and widely available iodinating agents, completely removing the dependency on costly boron species. This holistic redesign of the synthetic pathway ensures cost reduction in pharmaceutical intermediates manufacturing while maintaining rigorous stereochemical integrity. The result is a process that is inherently safer, more economical, and perfectly suited for reducing lead time for high-purity pharmaceutical intermediates.
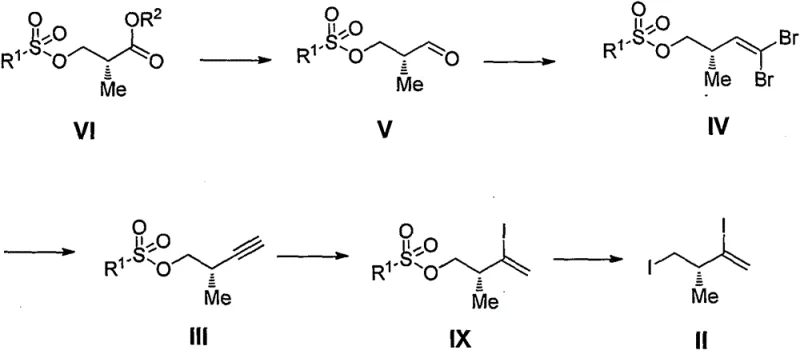
Mechanistic Insights into Sulfonate-Stabilized Alkyne Synthesis
The mechanistic elegance of this new pathway is rooted in the stability and reactivity profile of the sulfonate-protected intermediates. By employing a tosyl or similar sulfonate group early in the sequence, the synthesis establishes a robust framework that withstands the rigors of subsequent transformations without racemization. The conversion of the ester or amide precursor to the aldehyde via controlled reduction is critical, as it sets the stage for the Corey-Fuchs olefination. This reaction proceeds through a dibromoalkene intermediate, which is then subjected to base-mediated elimination to generate the terminal alkyne with high fidelity. The presence of the sulfonate moiety facilitates the final iodination steps by providing a suitable electronic environment for nucleophilic substitution. This careful orchestration of functional group interconversions ensures that the chiral center remains intact throughout the sequence, delivering the target Formula II with exceptional enantiomeric excess. Understanding these mechanistic nuances is vital for R&D Directors who must validate the feasibility of transferring this technology from the laboratory to the pilot plant.
Impurity control is another paramount aspect where this mechanism excels, particularly in the context of regulatory submissions for oncology drugs. The avoidance of enzymatic resolution steps, which often introduce biological contaminants and require complex downstream processing, significantly simplifies the impurity profile. The chemical specificity of the Corey-Fuchs reaction and the subsequent iodine addition minimizes the formation of side products that are difficult to separate. Furthermore, the mild reaction conditions reduce the likelihood of thermal degradation or polymerization, which are common sources of genotoxic impurities in aggressive synthetic routes. By maintaining a clean reaction profile, manufacturers can achieve stringent purity specifications with fewer crystallization or chromatography steps. This level of control is essential for ensuring the safety and efficacy of the final API, aligning with the rigorous quality standards expected by global health authorities and procurement teams alike.
How to Synthesize Eribulin Intermediate Efficiently
The practical implementation of this synthesis involves a series of well-defined unit operations that can be standardized across different manufacturing sites. The process begins with the preparation of the sulfonate-protected starting material, followed by a controlled reduction to the aldehyde phase. Subsequent bromination and elimination reactions are conducted under monitored conditions to ensure complete conversion before proceeding to the final iodination. Detailed standard operating procedures regarding stoichiometry, solvent selection, and quenching protocols are essential for reproducibility. For a comprehensive breakdown of the specific reaction parameters and workup procedures, please refer to the technical guide below.
- Reduce compound VI to aldehyde V using mild reducing agents.
- Perform Corey-Fuchs reaction on compound V to generate dibromoalkene IV.
- Execute elimination and iodination steps to finalize the chiral fragment II.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis offers transformative benefits for procurement strategies and supply chain resilience. The elimination of exotic reagents like B-I-9-BBN removes a significant single point of failure in the raw material supply chain, ensuring continuous production even during market fluctuations. The shift away from cryogenic conditions reduces energy consumption and infrastructure requirements, leading to substantial operational cost savings over the lifecycle of the product. Additionally, the simplified workflow reduces the total processing time, allowing for faster batch turnover and improved responsiveness to customer demand. These factors collectively enhance the reliability of supply, making it easier to secure long-term agreements with key stakeholders in the pharmaceutical industry.
- Cost Reduction in Manufacturing: The replacement of expensive boron reagents with commodity iodine chemicals results in a drastic decrease in raw material expenditures. Furthermore, the reduction in synthetic steps lowers labor costs and solvent usage, contributing to a leaner manufacturing budget. The avoidance of enzymatic resolution also eliminates the cost associated with specialized biocatalysts and their recovery processes. Overall, the process economics are significantly improved, allowing for more competitive pricing models without compromising margin.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents and standard equipment, the risk of supply disruption is minimized. The robustness of the sulfonate protecting group ensures that intermediates can be stored and transported with greater stability, reducing waste due to degradation. This reliability is crucial for maintaining just-in-time inventory levels and meeting strict delivery schedules required by downstream API manufacturers. The simplified logistics also facilitate easier qualification of multiple supply sources.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced solvent intensity make this process highly scalable from pilot to commercial volumes. The absence of hazardous heavy metals and toxic reagents simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This green chemistry approach not only mitigates regulatory risk but also aligns with the sustainability goals of modern pharmaceutical companies, enhancing the brand value of the supply partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and practical manufacturing considerations. They are designed to provide clarity for decision-makers evaluating the feasibility of adopting this route for their specific production needs. Understanding these details is key to forming a successful partnership for technology transfer.
Q: How does this new method improve upon the Kishi route?
A: The new method eliminates the need for expensive B-I-9-BBN reagents and avoids cryogenic conditions below -65°C, significantly lowering operational costs and safety risks.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of stable sulfonate protecting groups and simplified purification steps makes this route highly adaptable for commercial scale-up from kilograms to metric tons.
Q: What are the key purity advantages of this synthesis?
A: By avoiding frequent protecting group manipulations and enzymatic resolutions, the process minimizes impurity generation, ensuring high-purity intermediates for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eribulin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring this advanced synthesis route to life on a global scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of eribulin intermediate meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for navigating the complexities of oncology drug manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative process can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your development timeline. Let us collaborate to secure a sustainable and efficient supply chain for your critical pharmaceutical intermediates.
