Advanced Synthesis of Fluorinated Azobenzene Esters for High-Performance Liquid Crystal Displays
Advanced Synthesis of Fluorinated Azobenzene Esters for High-Performance Liquid Crystal Displays
The rapid evolution of the flat-panel display industry demands liquid crystal materials that possess superior electro-optical properties, including broad operating temperature ranges, low driving voltages, and exceptional chemical stability. Patent CN1928007A introduces a sophisticated synthetic route for a novel substituted azobenzene carboxylate liquid crystal compound, specifically 4'-n-heptyloxy-4-phenylazobenzoic acid-3-fluoro-4-cyanobenzene ester. This molecule integrates both fluorine atoms and cyano groups into its structure, a strategic design choice that significantly enhances the dielectric anisotropy and reduces the rotational viscosity of the final liquid crystal mixture. For R&D directors and procurement specialists in the electronic chemicals sector, understanding the nuances of this synthesis is critical, as it represents a shift towards more specialized, high-value intermediates capable of meeting the rigorous standards of modern TFT and STN display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for complex liquid crystal esters often rely on harsh reaction conditions that can compromise the integrity of sensitive functional groups. Conventional esterification methods frequently utilize acid chlorides generated by thionyl chloride or oxalyl chloride, which require elevated temperatures and generate corrosive byproducts that are difficult to remove completely. In the context of azobenzene derivatives, exposure to strong acids or high thermal stress can lead to the decomposition of the azo linkage (-N=N-) or the hydrolysis of the cyano group, resulting in colored impurities that degrade the optical performance of the display. Furthermore, older alkylation strategies for introducing the heptyloxy chain often suffered from poor regioselectivity or required excessive reaction times, leading to lower overall yields and increased waste generation, which poses significant challenges for cost-effective commercial manufacturing.
The Novel Approach
The methodology outlined in the patent data presents a refined, two-step strategy that mitigates these risks through precise control of reaction parameters. The process initiates with a Williamson etherification to install the n-heptyloxy chain under mildly alkaline conditions, followed by a dehydration condensation using dicyclohexylcarbodiimide (DCC) as a coupling agent. This approach allows the final esterification to proceed at near-ambient temperatures (0-30°C), effectively preserving the delicate fluorine and cyano substituents. By avoiding the formation of reactive acid chlorides and utilizing DMAP as a nucleophilic catalyst, the reaction achieves high conversion rates with minimal side reactions. This gentle yet efficient pathway ensures that the final product maintains the high purity levels required for electronic grade applications, directly addressing the quality concerns of downstream liquid crystal blenders.
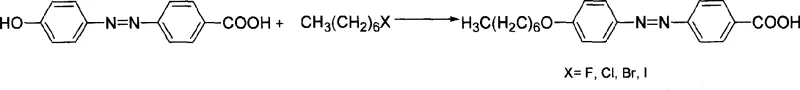
Mechanistic Insights into DCC-Mediated Dehydration Condensation
The core of this synthesis lies in the activation of the carboxylic acid moiety using DCC, a carbodiimide dehydrating agent that facilitates amide or ester bond formation under neutral conditions. In the presence of the catalyst 4-dimethylaminopyridine (DMAP), the carboxylic acid first reacts with DCC to form an unstable O-acylisourea intermediate. This activated species is highly electrophilic and is rapidly attacked by the nucleophilic hydroxyl group of the 2-fluoro-4-hydroxybenzonitrile. The DMAP catalyst plays a pivotal role by forming an even more reactive acylpyridinium intermediate, which accelerates the acylation of the sterically hindered phenol. This mechanism is particularly advantageous for substrates containing electron-withdrawing groups like fluorine and cyano, which typically reduce the nucleophilicity of the phenol, ensuring that the reaction proceeds to completion without requiring excessive heat that could trigger decomposition.
Impurity control is inherently built into this mechanistic pathway through the formation of dicyclohexylurea (DCU) as the sole stoichiometric byproduct. Unlike inorganic salts generated in traditional acid-base reactions, DCU is largely insoluble in many organic solvents, allowing it to be removed efficiently via simple filtration. This physical separation step significantly simplifies the downstream purification process, reducing the burden on chromatographic columns or recrystallization steps. Moreover, the mild alkaline conditions used in the preceding etherification step (pH > 10) ensure that any unreacted starting materials or acidic byproducts are converted into water-soluble salts, which can be washed away during the aqueous workup. This multi-layered approach to impurity management results in a final product with a clean impurity profile, essential for maintaining the high voltage holding ratio (VHR) required in active matrix displays.
How to Synthesize 4'-n-heptyloxy Azobenzene Ester Efficiently
The synthesis of this high-performance liquid crystal intermediate requires strict adherence to the patented protocol to ensure optimal yield and purity. The process begins with the preparation of the ether intermediate, where precise control of the solvent composition (ethanol-water ratio) and pH is essential to drive the nucleophilic substitution to completion while minimizing hydrolysis of the alkyl halide. Following the isolation of the pure acid intermediate, the subsequent esterification must be conducted under anhydrous conditions to prevent the hydrolysis of the DCC reagent. Detailed standardized operating procedures regarding reagent addition rates, temperature ramping, and workup protocols are critical for reproducibility. For a comprehensive guide on the specific molar ratios, solvent volumes, and purification techniques validated in the patent examples, please refer to the technical instructions below.
- Perform Williamson etherification by reacting p-hydroxyazobenzenecarboxylic acid with 1-haloheptane in an ethanol-water solvent system with alkali metal hydroxide at 50-80°C.
- Purify the intermediate 4'-n-heptyloxy-4-azobenzenecarboxylic acid via hydrolysis of byproducts and recrystallization.
- Conduct dehydration condensation using DCC and DMAP catalyst in anhydrous THF at 0-30°C with 2-fluoro-4-hydroxybenzonitrile to form the final ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits in terms of cost structure and operational reliability. The reliance on commodity chemicals such as ethanol, tetrahydrofuran, and standard alkyl halides ensures that raw material sourcing remains stable and unaffected by the volatility associated with exotic reagents. Furthermore, the elimination of hazardous chlorinating agents reduces the safety risks and environmental compliance costs associated with waste disposal. The mild reaction conditions also translate to lower energy consumption, as the process does not require prolonged heating at high temperatures or cryogenic cooling, aligning with modern green chemistry initiatives and reducing the overall carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The use of DCC coupling, while involving a specific reagent cost, eliminates the need for expensive and corrosive acid chloride formation steps, thereby reducing equipment maintenance costs and extending reactor lifespan. The efficient removal of the DCU byproduct via filtration minimizes the need for complex and solvent-intensive purification steps like column chromatography on a large scale, leading to substantial savings in solvent recovery and waste treatment. Additionally, the high selectivity of the reaction reduces the formation of difficult-to-separate isomers, improving the overall mass balance and reducing the loss of valuable starting materials during purification.
- Enhanced Supply Chain Reliability: The starting materials, including p-hydroxyazobenzenecarboxylic acid and fluoro-hydroxybenzonitriles, are established intermediates in the fine chemical industry with multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the synthetic method allows for flexible production scheduling, as the reactions are not overly sensitive to minor fluctuations in ambient conditions, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply lines to liquid crystal blenders who operate on tight just-in-time delivery schedules for the electronics sector.
- Scalability and Environmental Compliance: The process utilizes standard unit operations such as reflux, filtration, and recrystallization, which are easily scalable from pilot plants to multi-ton commercial production facilities without requiring specialized high-pressure or high-temperature equipment. The aqueous workup steps generate waste streams that are relatively straightforward to treat compared to those containing heavy metals or persistent organic pollutants, facilitating easier compliance with increasingly stringent environmental regulations. This scalability ensures that the supply of this critical intermediate can grow in tandem with the expanding demand for advanced display technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated azobenzene derivative. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's potential. Understanding these details helps stakeholders make informed decisions regarding the integration of this intermediate into their supply chains and product formulations.
Q: What are the key advantages of this fluorinated azobenzene ester for LCD applications?
A: This compound offers a high clearing point, a wide liquid crystal working temperature range, and low viscosity, making it ideal for TN, STN, and TFT display technologies requiring fast response times and stability.
Q: How does the DCC coupling method improve product quality compared to traditional acid chloride methods?
A: The DCC/DMAP mediated esterification occurs under mild conditions (0-30°C), preventing thermal degradation of the sensitive azo (-N=N-) and cyano (-CN) functional groups, thereby ensuring higher chemical purity and color stability.
Q: Is this synthesis route scalable for industrial production of liquid crystal intermediates?
A: Yes, the process utilizes common solvents like ethanol and THF and standard unit operations such as filtration and recrystallization, facilitating straightforward scale-up from laboratory to commercial manufacturing without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-n-heptyloxy Azobenzene Ester Supplier
As the demand for high-performance liquid crystal materials continues to surge, partnering with a chemically proficient manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major display panel manufacturers. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 4'-n-heptyloxy-4-phenylazobenzoic acid-3-fluoro-4-cyanobenzene ester meets the exacting standards required for electronic applications. We understand the critical nature of impurity profiles in liquid crystals and employ advanced analytical techniques to verify the absence of ionic contaminants and colored impurities.
We invite you to collaborate with our technical team to explore how this advanced intermediate can enhance your liquid crystal formulations. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can deliver both superior quality and economic value to your organization.
