Scalable Manufacturing of Ruxolitinib Phosphate Intermediates via Novel Nickel-Catalyzed Routes
Scalable Manufacturing of Ruxolitinib Phosphate Intermediates via Novel Nickel-Catalyzed Routes
The pharmaceutical industry is constantly seeking robust, cost-effective pathways for the production of critical oncology therapeutics, and the recent disclosure in patent CN114044777A presents a transformative approach to synthesizing Ruxolitinib Phosphate. This document details a sophisticated six-step synthetic strategy that circumvents the limitations of traditional methods, specifically addressing the high costs associated with noble metal catalysts and the inefficiencies of chiral separation technologies. By leveraging a Nickel-catalyzed Suzuki coupling and a highly selective chiral resolution step using camphoric acid, this methodology delivers exceptional optical purity exceeding 99.8 percent while simplifying the downstream processing requirements. For global supply chain stakeholders, this represents a significant opportunity to secure a more reliable pharmaceutical intermediate supplier capable of delivering high-purity JAK inhibitor precursors with improved economic viability.
![Chemical structure of Ruxolitinib Phosphate showing the pyrrolo[2,3-d]pyrimidine core and cyclopentyl side chain](/insights/img/ruxolitinib-phosphate-nickel-catalysis-pharma-supplier-20260309062758-01.png)
The structural complexity of Ruxolitinib Phosphate, characterized by its fused pyrrolo[2,3-d]pyrimidine core and chiral cyclopentyl-propionitrile side chain, has historically posed significant challenges for process chemists aiming for commercial scale-up. The innovation described in this patent lies not just in the sequence of reactions, but in the strategic selection of reagents that balance reactivity with cost-efficiency. The route begins with the protection of the pyrrole nitrogen, proceeds through a boronation and coupling sequence, and culminates in a precise resolution and deprotection strategy. This comprehensive technical breakthrough ensures that the final active pharmaceutical ingredient meets stringent regulatory standards for impurity profiles, making it an ideal candidate for integration into existing GMP manufacturing lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Ruxolitinib intermediates have been plagued by inherent inefficiencies that hinder large-scale production. One prominent historical route relied on asymmetric induction using chiral micro-molecules to establish the critical stereocenter early in the synthesis. However, this approach suffers from the use of chiral induction reagents with high molecular weights and exorbitant preparation costs, alongside harsh reaction conditions that compromise safety and scalability. Furthermore, the selectivity of the asymmetric Michael addition in these legacy routes is often suboptimal, leading to difficult separations and reduced overall yields. Another documented method utilized Rhodium-catalyzed reactions with chiral ligands, which introduced the dual burden of expensive noble metals and starting materials that are not readily commercially available, thereby creating bottlenecks in the supply chain.
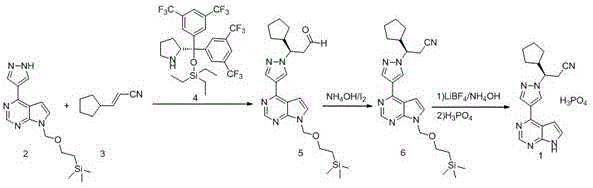
Additionally, some existing patents describe routes where the key intermediate requires purification via chiral preparation columns, a technique that is notoriously inefficient for ton-scale manufacturing. The reliance on preparative HPLC or column chromatography significantly increases solvent consumption, extends cycle times, and introduces substantial operational costs that are unsustainable for generic API production. These conventional methods often result in optical purities that hover near the acceptance threshold, necessitating additional recrystallization steps that further erode yield. The cumulative effect of these drawbacks is a manufacturing process that is fragile, costly, and difficult to control, prompting the urgent need for the innovative solution presented in the current patent data.
The Novel Approach
The novel synthetic route disclosed in CN114044777A fundamentally reengineers the production logic by deferring chirality introduction to a later stage where it can be managed more economically through resolution rather than asymmetric synthesis. This strategy employs a robust Nickel-catalyzed Suzuki coupling to construct the carbon-carbon bond between the pyrimidine core and the pyrazole ring, replacing the traditionally expensive Palladium systems. The use of Ni(PCy3)2Cl2 provides a catalyst system that is not only more stable but also significantly more cost-effective, allowing for high conversion rates under mild conditions. Following the coupling, a Michael addition with 3-cyclopentylacrylonitrile installs the side chain, setting the stage for the pivotal resolution step.
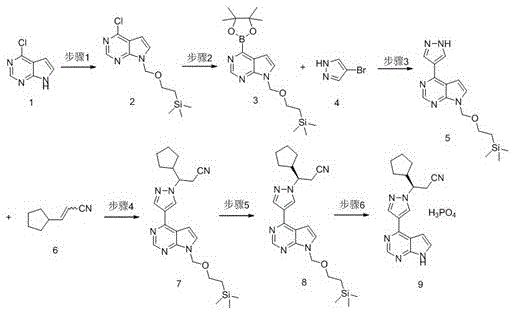
Perhaps the most critical advancement in this new approach is the implementation of a chiral resolution step using D-(+)-camphoric acid. This specific resolving agent demonstrates superior selectivity compared to alternatives, enabling the isolation of the desired enantiomer with an optical purity reaching 99.8 percent and chemical purity exceeding 99.5 percent. Crucially, this high level of purity is achieved without the need for column chromatography, relying instead on crystallization and filtration techniques that are inherently scalable. The final deprotection and salt formation with phosphoric acid are conducted in common solvents like acetone, ensuring that the entire process remains grounded in practical, industrial chemistry principles that facilitate seamless technology transfer.
Mechanistic Insights into Nickel-Catalyzed Suzuki Coupling and Chiral Resolution
The mechanistic elegance of this synthesis is anchored in the substitution of Palladium with Nickel for the Suzuki-Miyaura coupling step. In this transformation, the nickel catalyst, specifically Ni(PCy3)2Cl2, facilitates the oxidative addition into the carbon-chlorine bond of the pyrimidine intermediate, followed by transmetallation with the boronic ester species generated in the preceding step. The use of a Nickel center is particularly advantageous here because it exhibits distinct electronic properties that can enhance reactivity towards heteroaryl chlorides, which are often less reactive than their bromide or iodide counterparts in Palladium systems. This allows the reaction to proceed efficiently at moderate temperatures of 65-70 ℃, minimizing thermal degradation of the sensitive heterocyclic core. The catalyst's stability ensures consistent performance across batches, reducing the risk of residual metal contamination in the final product, a critical parameter for regulatory compliance.
Following the construction of the core scaffold, the control of stereochemistry is achieved through a classical yet highly optimized diastereomeric salt formation. The racemic nitrile intermediate reacts with D-(+)-camphoric acid to form diastereomeric salts, which possess different solubility profiles in methyl acetate. By carefully controlling the temperature profile—dissolving at ambient temperature, heating to 55-60 ℃, and then cooling—the less soluble salt of the desired (R)-enantiomer precipitates selectively. This thermodynamic control allows for the exclusion of the (S)-enantiomer and other impurities into the mother liquor. The subsequent treatment with triethylamine liberates the free base, which retains the high optical purity established during crystallization. This mechanism effectively bypasses the kinetic limitations of asymmetric catalysis, offering a more predictable and robust path to high enantiomeric excess.
How to Synthesize Ruxolitinib Phosphate Efficiently
The execution of this synthetic pathway requires precise adherence to the reaction parameters outlined in the patent to maximize yield and purity. The process begins with the protection of the pyrrolo[2,3-d]pyrimidine nitrogen using a SEM group, which serves as a robust protecting group that withstands the subsequent Grignard and coupling conditions. The formation of the boronic ester via Grignard reagent and pinacol diboron must be conducted at low temperatures to prevent side reactions, ensuring the integrity of the boron species for the coupling step. The subsequent Nickel-catalyzed coupling and Michael addition are performed in polar aprotic solvents like DMF or DMAc, which solubilize the intermediates effectively. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and workup procedures, are provided in the technical guide below.
- Protect 4-chloro-7H-pyrrolo[2,3-d]pyrimidine with SEM group using sodium hydride and SEM-Cl in N,N-diethylphenylacetamide.
- Perform Grignard reaction followed by boronation with pinacol diboron to generate the key boronic ester intermediate.
- Execute Suzuki coupling with 4-bromo-1H-pyrazole using a stable Nickel catalyst (Ni(PCy3)2Cl2) instead of Palladium.
- Conduct Michael addition with 3-cyclopentylacrylonitrile using DMAP as a base to form the racemic nitrile intermediate.
- Resolve the racemic mixture using D-(+)-camphoric acid in methyl acetate to achieve high optical purity (>99.8%).
- Remove the SEM protecting group and form the phosphate salt using phosphoric acid in acetone to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the need for column chromatography, the process removes a major bottleneck that typically consumes vast quantities of silica gel and organic solvents, thereby significantly reducing waste disposal costs and environmental impact. This streamlining of operations translates directly into shorter batch cycle times and increased throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts like Rhodium and Palladium with a stable Nickel catalyst represents a direct material cost saving that compounds over large production volumes. Furthermore, the use of D-(+)-camphoric acid as a resolving agent is economically superior to proprietary chiral ligands or micro-molecules, as it is a commodity chemical with a stable supply chain. The elimination of preparative chiral HPLC steps further drives down operational expenditures by removing the need for specialized equipment and high-purity mobile phases, resulting in substantial overall cost optimization for the final API.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 4-chloro-7H-pyrrolo[2,3-d]pyrimidine and 3-cyclopentylacrylonitrile mitigates the risk of raw material shortages that often plague complex synthetic routes. Unlike methods requiring custom-synthesized chiral auxiliaries or rare earth metals, the reagents in this protocol are sourced from established bulk chemical suppliers, ensuring continuity of supply. This robustness makes the manufacturing process less susceptible to geopolitical or logistical disruptions, providing a secure foundation for long-term procurement planning and inventory management.
- Scalability and Environmental Compliance: The mild reaction conditions, typically ranging from ambient temperature to 70 ℃, reduce the energy load required for heating and cooling, aligning with modern green chemistry initiatives. The simplified workup procedures, which rely on crystallization and filtration rather than complex extractions or chromatographic separations, generate less hazardous waste and simplify effluent treatment. This ease of scale-up means that the transition from pilot plant to commercial production involves fewer engineering challenges, facilitating faster time-to-market for generic versions of the drug while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this methodology for their own production lines.
Q: Why is the Nickel catalyst preferred over Palladium in this Ruxolitinib synthesis route?
A: The patent specifies the use of Ni(PCy3)2Cl2 because it offers higher stability and significantly lower cost compared to traditional Palladium catalysts, while maintaining high coupling yields (up to 95%) without requiring expensive ligand systems.
Q: How does the new chiral resolution method improve upon previous asymmetric induction techniques?
A: Unlike prior art relying on expensive chiral micro-molecules or harsh asymmetric Michael additions, this route utilizes D-(+)-camphoric acid for resolution. This achieves superior optical purity (>99.8%) and chemical purity (>99.5%) with simpler post-treatment, eliminating the need for preparative chiral HPLC.
Q: Is column chromatography required for the purification of intermediates in this process?
A: No, a major advantage of this patented method is that column chromatography is not needed. The process relies on crystallization and simple filtration steps, which drastically reduces solvent consumption and processing time, making it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruxolitinib Phosphate Supplier
As the global demand for JAK inhibitors continues to rise, securing a manufacturing partner with deep expertise in complex heterocyclic synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of Nickel-catalyzed reactions and chiral resolutions, ensuring that every batch of Ruxolitinib Phosphate intermediate meets stringent purity specifications. With our rigorous QC labs and commitment to process excellence, we guarantee a supply of high-purity pharmaceutical intermediates that adhere to the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this Nickel-catalyzed process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both cost and reliability in the competitive oncology market.
