Scalable Synthesis of Gamma-Hydroxy-Gamma-Perfluoromethylbutenolides via Organocatalysis
Introduction to Advanced Fluorinated Butenolide Synthesis
The development of efficient synthetic routes for fluorinated heterocycles remains a critical frontier in modern medicinal chemistry, particularly for the construction of complex pharmaceutical intermediates. Patent CN114605361B introduces a groundbreaking methodology for the synthesis of γ-hydroxy-γ-perfluoromethylbutenolide compounds, utilizing a metal-free organocatalytic strategy that addresses long-standing challenges in the field. This innovation leverages Morita-Baylis-Hillman (MBH) carbonates and 2-perfluoroalkyl-oxazol-5(2H)-ones as key building blocks, catalyzed by the inexpensive and readily available organic base DABCO. The significance of this technology lies in its ability to construct highly functionalized butenolide scaffolds containing trifluoromethyl or pentafluoroethyl groups without the reliance on precious transition metals or harsh stoichiometric oxidants. For global pharmaceutical manufacturers, this represents a paradigm shift towards more sustainable and cost-effective manufacturing processes for high-value active pharmaceutical ingredient (API) precursors.
Furthermore, the introduction of perfluoroalkyl groups into the butenolide core significantly enhances the metabolic stability and lipophilicity of the resulting molecules, properties that are highly sought after in the design of next-generation therapeutics. The patent details a robust protocol that operates under mild reaction conditions, typically ranging from 0°C to 60°C, and tolerates an air atmosphere, which drastically simplifies operational requirements compared to inert gas-sensitive metal catalysis. By eliminating the need for complex ligand systems and expensive metal salts, this method not only reduces the direct cost of goods but also mitigates the environmental burden associated with heavy metal waste disposal. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative synthetic pathways is essential for maintaining competitiveness in the rapidly evolving landscape of fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of γ-hydroxy-γ-perfluoromethylbutenolides has relied heavily on transition metal catalysis or the use of strong stoichiometric oxidants, both of which present significant drawbacks for large-scale industrial application. Traditional methods often require palladium, rhodium, or copper catalysts, which are not only prohibitively expensive due to their scarcity but also pose severe contamination risks that necessitate rigorous and costly purification steps to meet regulatory limits for residual metals in drug substances. Additionally, many conventional protocols demand strictly anhydrous and anaerobic conditions, requiring specialized equipment and increasing the complexity of process engineering. The use of stoichiometric oxidants further complicates the safety profile of the reaction, generating substantial amounts of chemical waste that must be treated before disposal, thereby inflating the overall environmental footprint and operational costs of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a simple organocatalytic system driven by DABCO, which acts as a nucleophilic catalyst to activate the MBH carbonate electrophile. This metal-free strategy allows the reaction to proceed smoothly in common organic solvents such as dichloromethane or ethanol under ambient air conditions, removing the need for expensive inert gas protection. The subsequent hydrolysis and cyclization steps are mediated by mild inorganic bases like sodium bicarbonate, ensuring a gentle transformation that preserves sensitive functional groups on the aromatic ring. This streamlined two-step one-pot procedure significantly shortens the production timeline and minimizes solvent consumption. 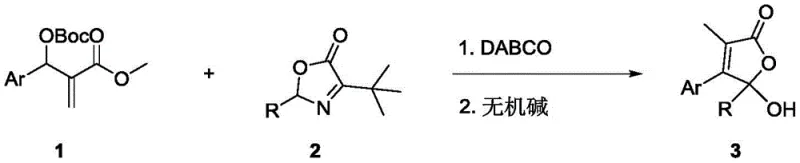 As illustrated in the reaction scheme, the transformation efficiently couples the MBH carbonate with the oxazolone derivative to yield the target butenolide with high regioselectivity and excellent yields, demonstrating a clear advantage over legacy technologies in terms of both economic and operational efficiency.
As illustrated in the reaction scheme, the transformation efficiently couples the MBH carbonate with the oxazolone derivative to yield the target butenolide with high regioselectivity and excellent yields, demonstrating a clear advantage over legacy technologies in terms of both economic and operational efficiency.
Mechanistic Insights into DABCO-Catalyzed Cyclization
The mechanistic pathway of this transformation begins with the nucleophilic attack of the DABCO catalyst on the MBH carbonate, generating a reactive zwitterionic intermediate that serves as a potent electrophile. This activated species is then attacked by the nucleophilic carbon of the 2-perfluoroalkyl-oxazol-5(2H)-one, forming a new carbon-carbon bond and establishing the core skeleton of the butenolide framework. The presence of the perfluoroalkyl group on the oxazolone ring enhances the acidity of the adjacent proton, facilitating the deprotonation and subsequent elimination of the carbonate leaving group. This cascade sequence occurs rapidly at room temperature, highlighting the high catalytic efficiency of DABCO in promoting C-C bond formation without the need for external energy input or metal coordination. The robustness of this mechanism is evidenced by its tolerance to a wide variety of electronic environments on the aryl ring of the MBH carbonate.
Following the initial coupling, the reaction mixture undergoes a base-mediated hydrolysis and cyclization sequence upon the addition of an inorganic base and alcohol solvent at elevated temperatures (e.g., 55°C). The inorganic base, such as sodium carbonate or sodium bicarbonate, facilitates the hydrolysis of the ester moiety and promotes the intramolecular cyclization to close the five-membered lactone ring. This step is crucial for establishing the final γ-hydroxy-γ-perfluoromethyl substitution pattern, which is critical for the biological activity of the molecule. The mild basic conditions prevent the decomposition of the sensitive fluorinated intermediates, ensuring high product purity and minimizing the formation of side products. From a quality control perspective, the absence of metal catalysts means that the impurity profile is significantly cleaner, primarily consisting of organic byproducts that are easily removed via standard silica gel chromatography or crystallization, thus ensuring high-purity pharmaceutical intermediates suitable for downstream processing.
How to Synthesize Gamma-Hydroxy-Gamma-Perfluoromethylbutenolides Efficiently
The practical implementation of this synthesis involves a straightforward protocol that can be easily adapted for both laboratory screening and pilot plant operations. The process begins by dissolving the MBH carbonate and the oxazolone derivative in a suitable organic solvent, followed by the addition of the DABCO catalyst. After stirring at room temperature to complete the coupling, the solvent is removed, and the residue is subjected to the cyclization conditions using an aqueous base and ethanol. This simplicity allows for rapid optimization and scale-up, making it an ideal candidate for contract development and manufacturing organizations (CDMOs) looking to expand their fluorinated chemistry capabilities. The detailed standardized synthesis steps are outlined in the guide below.
- Dissolve MBH carbonate and 2-perfluoroalkyl-oxazol-5(2H)-one in an organic solvent such as dichloromethane, then add DABCO catalyst (10 mol%) and stir at room temperature.
- Monitor the reaction progress via TLC until the starting materials are fully consumed, typically requiring approximately 1 hour.
- Concentrate the mixture, then add an aqueous inorganic base solution (e.g., NaHCO3) and ethanol, heating to 55°C to facilitate hydrolysis and cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DABCO-catalyzed methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of transition metals from the synthetic route directly translates to a significant reduction in raw material costs, as DABCO is a commodity chemical compared to precious metal catalysts like palladium or rhodium. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents required for metal scavenging, leading to lower waste treatment costs and a smaller environmental footprint. This aligns perfectly with the industry's growing emphasis on green chemistry and sustainable manufacturing practices, potentially qualifying the process for various environmental incentives and improving the corporate sustainability profile of the manufacturing entity.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive organic amines like DABCO results in a drastic decrease in the bill of materials for the synthesis of these complex intermediates. Since the reaction does not require stoichiometric oxidants or specialized ligands, the overall reagent cost is minimized, allowing for more competitive pricing in the global market. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, further contributing to operational expenditure savings. The high yields reported across a broad substrate scope ensure that material throughput is maximized, reducing the cost per kilogram of the final API intermediate and enhancing the overall profitability of the production campaign.
- Enhanced Supply Chain Reliability: The starting materials, specifically MBH carbonates and 2-perfluoroalkyl-oxazol-5(2H)-ones, are commercially available or easily synthesized from abundant feedstocks, ensuring a stable and continuous supply chain. Unlike metal catalysts, which are subject to geopolitical supply constraints and price volatility, organic catalysts like DABCO are produced in vast quantities globally, mitigating the risk of production delays due to raw material shortages. The robustness of the reaction under air atmosphere also means that manufacturing does not rely on complex infrastructure for inert gas generation, making it easier to execute in diverse geographic locations and ensuring consistent delivery timelines for international clients seeking a reliable pharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable under mild temperatures and the absence of hazardous reagents that pose explosion risks upon scale-up. The use of common solvents like dichloromethane and ethanol simplifies solvent recovery and recycling systems, which is critical for large-scale commercial production. Moreover, the absence of heavy metal waste simplifies regulatory compliance regarding effluent discharge and solid waste disposal, reducing the administrative burden and potential fines associated with environmental violations. This ease of scale-up from 100 kgs to 100 MT annual commercial production ensures that the technology can meet the demands of late-stage clinical trials and commercial launch without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this chemistry into their existing pipelines and for procurement teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of using DABCO over transition metal catalysts for this synthesis?
A: The use of DABCO eliminates the need for expensive and toxic transition metals, significantly reducing raw material costs and simplifying the removal of metal residues to meet stringent pharmaceutical purity standards.
Q: Does this method support a broad range of substrate substituents?
A: Yes, the protocol demonstrates excellent tolerance for various functional groups including halogens (F, Cl, Br), alkyl, alkoxy, and heterocycles like thiophene and furan, ensuring versatility for diverse drug discovery programs.
Q: How does the reaction condition impact scalability?
A: The reaction proceeds under mild conditions (0-60°C) and air atmosphere without stoichiometric oxidants, making it inherently safer and easier to scale up from gram to multi-ton production compared to sensitive metal-catalyzed processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Hydroxy-Gamma-Perfluoromethylbutenolides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free organocatalytic technology in accelerating the development of fluorinated pharmaceuticals. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of high-purity fluorinated intermediates meets the exacting standards required by global regulatory agencies. We are committed to leveraging our technical expertise to optimize this DABCO-catalyzed route for your specific needs, ensuring maximum yield and minimal impurity profiles.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can drive cost reduction in API manufacturing for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain resilience and accelerate your time to market with next-generation therapeutic agents.
