Advanced Synthesis of Rigid Phenylquinoline Iridium Complexes for High-Performance Red OLEDs
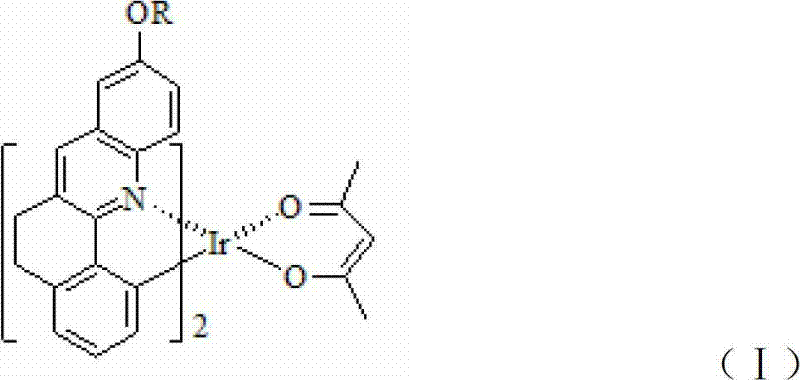
The landscape of organic light-emitting diode (OLED) technology has been fundamentally transformed by the introduction of phosphorescent materials, which harness both singlet and triplet excitons to achieve theoretically 100% internal quantum efficiency. Within this domain, patent CN102690293A presents a significant breakthrough in the development of red phosphorescent emitters, specifically focusing on a novel class of iridium complexes featuring rigid phenylquinoline ligands. As a leading entity in the fine chemical sector, we recognize that the transition from fluorescent to phosphorescent materials represents a critical pivot point for display manufacturers seeking higher brightness and lower power consumption. This patent details the synthesis of an iridium complex, denoted as (DHBA)2Ir(acac), where the ligand structure is engineered to maximize rigidity through a unique methylene-bridged connection between the benzene and quinoline rings. The technical implications of this structural modification are profound, offering a pathway to overcome the efficiency roll-off and color purity issues that have historically plagued red OLED emitters. By leveraging low-valent titanium chemistry to construct the core heterocyclic framework, the inventors have established a robust synthetic route that balances high performance with chemical feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industry relied heavily on various rigid ligand systems to enhance the luminescent properties of iridium complexes. However, historical data indicates that simply increasing rigidity does not guarantee superior device performance. For instance, earlier complexes based on rigid ligands such as DBA and BA, while capable of achieving saturated red emission, demonstrated disappointing device metrics with maximum brightness capped at approximately 9,540 cd/m² and external quantum efficiencies hovering around 4.66%. These limitations stem from non-radiative decay pathways that are not sufficiently suppressed by the ligand architecture alone. Furthermore, many conventional synthesis routes for these complex heterocycles involve harsh conditions or multiple protection-deprotection steps that introduce impurities difficult to remove via standard purification techniques. The presence of such impurities acts as quenching sites within the emissive layer, drastically reducing the operational lifetime and efficiency of the final OLED device. Consequently, there has been a persistent demand for a reliable OLED material supplier who can provide emitters that break through these efficiency ceilings without compromising on color saturation or stability.
The Novel Approach
The methodology outlined in CN102690293A addresses these deficiencies through a strategic molecular design coupled with an efficient synthetic strategy. The core innovation lies in the specific arrangement of the phenylquinoline ligand, where two methylene groups link the benzene and quinoline moieties. This structural feature significantly enhances the rigidity of the entire coordination sphere around the iridium center, thereby restricting molecular vibration and rotation that typically lead to energy loss. The result is a material that, when doped into a host matrix like CBP at concentrations between 7% and 10%, yields devices with maximum brightness ranging from 55,800 to 66,512 cd/m². This represents a magnitude of improvement over previous generations. Moreover, the synthetic route avoids overly complex multistep sequences by utilizing a direct intramolecular reductive coupling. This approach not only streamlines the production workflow but also facilitates cost reduction in electronic chemical manufacturing by minimizing solvent usage and reaction time. The ability to tune the alkoxy group (R = Me, Et, i-Pr, t-Bu) on the phenyl ring further allows for fine-tuning of the solubility and sublimation properties, which is critical for the vacuum thermal evaporation processes used in panel fabrication.
Mechanistic Insights into Low-Valent Titanium Mediated Cyclization
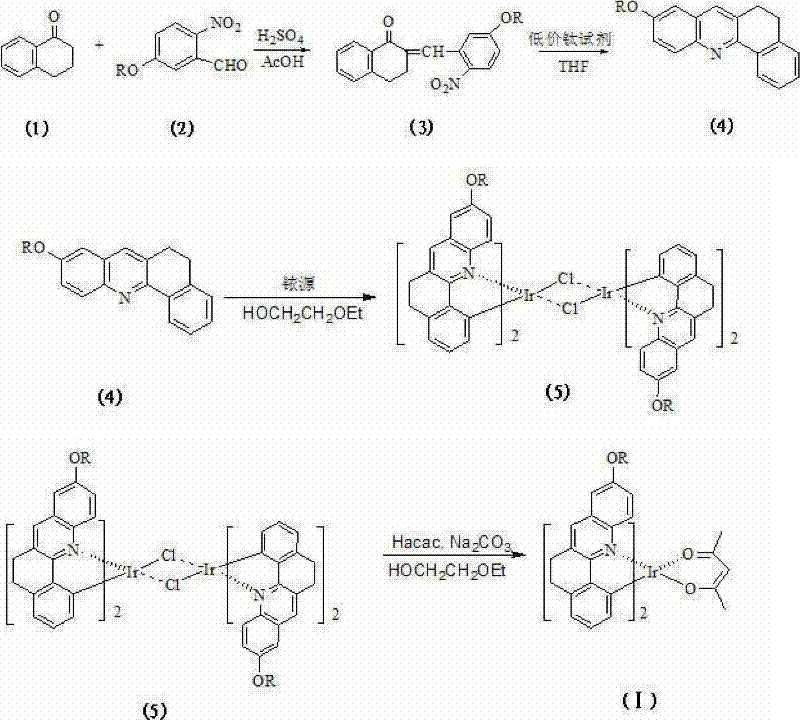
The heart of this synthesis lies in the construction of the phenylquinoline backbone via a low-valent titanium-mediated reductive coupling, a reaction mechanism that offers exceptional chemoselectivity. The process begins with an aldol condensation between a 2-nitrobenzaldehyde derivative and tetralone, catalyzed by sulfuric acid in glacial acetic acid. This step generates a chalcone-like intermediate containing both a nitro group and an alpha,beta-unsaturated ketone. The subsequent transformation is where the true chemical elegance is displayed: the addition of a low-valent titanium reagent, prepared in situ from zinc powder and titanium tetrachloride (TiCl4) in a precise 2:1 molar ratio. This reagent system acts as a powerful oxygen scavenger and reductant, facilitating the intramolecular cyclization of the nitro group with the adjacent aromatic system to form the quinoline ring. Unlike traditional reduction methods that might reduce the nitro group to an amine without cyclization, the low-valent titanium species promotes the specific dehydration and aromatization required to close the ring efficiently. This mechanistic pathway ensures that the conjugation degree of the nitrogen-containing hybrid part is maximized, which is directly correlated with the observed enhancement in luminous efficiency. The reaction proceeds under mild conditions, typically at room temperature overnight, which preserves the integrity of sensitive functional groups and prevents the formation of polymeric byproducts.
Impurity control is paramount in the production of high-purity OLED material, and the patent describes a rigorous purification protocol integrated directly into the synthetic workflow. Following the reductive coupling, the crude phenylquinoline ligand is isolated and subjected to recrystallization from 95% ethanol. This step is crucial for removing residual titanium salts and unreacted starting materials that could otherwise coordinate with the iridium in later stages, leading to off-color emission or reduced quantum yield. In the subsequent metallation step, the purified ligand reacts with iridium trichloride hydrate in 2-ethoxyethanol to form a chloro-bridged dimer. The use of a slight excess of ligand (molar ratio of ligand to iridium source of approximately 2.1 to 2.4) drives the equilibrium towards the formation of the bis-cyclometalated species, minimizing the presence of mono-cyclometalated impurities. Finally, the displacement of the chloride bridges with acetylacetone (acac) in the presence of anhydrous sodium carbonate yields the neutral, sublimable complex. The final product is recrystallized from absolute ethanol, ensuring that the material meets the stringent purity specifications required for commercial display applications. This comprehensive approach to impurity management underscores the feasibility of scaling this chemistry for industrial production.
How to Synthesize (DHBA)2Ir(acac) Efficiently
The synthesis of this high-performance emitter follows a logical progression from simple commodity chemicals to a sophisticated organometallic complex. The process is designed to be robust, utilizing reagents that are readily available in the global chemical supply chain. The initial formation of the low-valent titanium reagent requires careful control of stoichiometry and atmosphere, typically under nitrogen protection to prevent oxidation of the active titanium species. Once generated, this reagent is immediately utilized to cyclize the nitro-chalcone intermediate, a step that defines the structural rigidity of the final emitter. The subsequent coordination with iridium is performed under reflux conditions, ensuring complete conversion to the chloro-bridged dimer before the final ligand exchange with acetylacetone.
- Perform aldol condensation between 2-nitrobenzaldehyde derivatives and tetralone under sulfuric acid catalysis to form the intermediate chalcone derivative.
- Execute intramolecular reductive coupling using a low-valent titanium reagent (TiCl4-Zn) to cyclize the intermediate into the rigid phenylquinoline ligand.
- React the purified ligand with an iridium source (IrCl3) to form a chloro-bridged dimer, followed by substitution with acetylacetone to yield the final complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this specific iridium complex technology offers distinct strategic advantages beyond mere performance metrics. The synthetic route described eliminates the need for exotic catalysts or extremely low-temperature cryogenic conditions, which are often bottlenecks in the manufacturing of specialty electronic chemicals. By relying on standard unit operations such as reflux, filtration, and recrystallization, the process is inherently more adaptable to large-scale reactor systems. This adaptability translates directly into enhanced supply chain reliability, as the production of the key phenylquinoline ligand does not depend on single-source proprietary catalysts that might face availability issues. Furthermore, the high efficiency of the resulting material means that less dopant is required to achieve the same luminance levels in the final OLED panel. This reduction in material loading can lead to substantial cost savings in the overall bill of materials for display manufacturers, effectively lowering the cost per unit of the finished display without sacrificing quality.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the ligand synthesis step, replaced by the cost-effective TiCl4-Zn system, significantly lowers the raw material input costs. Additionally, the high yield and selectivity of the reductive coupling reaction minimize waste generation and the need for extensive chromatographic purification, which is notoriously expensive and difficult to scale. The use of ethanol for recrystallization steps further reduces solvent costs compared to processes requiring chlorinated solvents or specialized ethers. These factors combine to create a manufacturing profile that supports significant cost optimization, making high-efficiency red phosphorescence more economically viable for mass-market applications.
- Enhanced Supply Chain Reliability: The precursors for this synthesis, including tetralone, nitrobenzaldehyde derivatives, zinc powder, and iridium chloride, are commodity chemicals with well-established global supply networks. This diversity of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the stability of the intermediate chloro-bridged dimer allows for potential stockpiling or semi-finished goods storage, providing flexibility in production scheduling. For a reliable OLED material supplier, this means the ability to maintain consistent delivery schedules even during periods of fluctuating demand, ensuring that downstream panel manufacturers can meet their own production targets without interruption.
- Scalability and Environmental Compliance: The process operates primarily in alcoholic and glycol ether solvents, which are easier to recover and recycle compared to halogenated alternatives, aligning with increasingly strict environmental regulations in chemical manufacturing. The absence of heavy metal catalysts in the organic synthesis phase simplifies waste treatment protocols, as the primary metal concern is limited to the valuable iridium itself, which is recovered and recycled in standard precious metal recovery loops. The straightforward nature of the reaction conditions—mostly atmospheric pressure and moderate temperatures—facilitates the commercial scale-up of complex electronic chemicals from pilot plant quantities to multi-ton annual production capacities with minimal engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphorescent material technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance expectations and integration requirements. Understanding these nuances is essential for R&D teams evaluating this material for next-generation display architectures.
Q: What distinguishes this iridium complex from previous rigid ligand emitters?
A: Unlike earlier rigid ligands like DBA or BA which suffered from lower brightness and efficiency despite saturated red emission, this complex utilizes a specific methylene-bridged structure between the benzene and quinoline rings. This unique architecture significantly increases rigidity and conjugation, resulting in maximum brightness exceeding 60,000 cd/m² and external quantum efficiencies up to 17.7%.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the process relies on standard industrial operations such as reflux in 2-ethoxyethanol and recrystallization from ethanol. The use of commercially available reagents like zinc powder, titanium tetrachloride, and iridium trichloride ensures that the supply chain remains robust and the method is adaptable for commercial scale-up of complex electronic chemicals.
Q: How does the low-valent titanium reagent impact product purity?
A: The use of the TiCl4-Zn system allows for a highly selective intramolecular reductive coupling. This specificity minimizes side reactions common in other reduction methods. Furthermore, the protocol includes specific recrystallization steps using 95% ethanol for the ligand and absolute ethanol for the final complex, ensuring high-purity OLED material suitable for vacuum deposition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (DHBA)2Ir(acac) Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent CN102690293A can be realized in your supply chain. We are committed to delivering high-purity OLED material that meets the rigorous demands of the display industry, utilizing our state-of-the-art rigorous QC labs to verify every batch against stringent purity specifications. Our infrastructure is designed to handle the sensitivities of organometallic synthesis, providing the controlled environments necessary to maintain the integrity of these high-value emitters throughout the production lifecycle.
We invite you to engage with our technical procurement team to discuss how this advanced iridium complex can enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this more efficient emitter. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your specific volume requirements. Let us collaborate to bring the next generation of high-brightness, energy-efficient red OLED displays to market faster and more cost-effectively.
