Advanced Synthesis of Bisphenol Imide Monomers for High-Performance Polyimide Production
Advanced Synthesis of Bisphenol Imide Monomers for High-Performance Polyimide Production
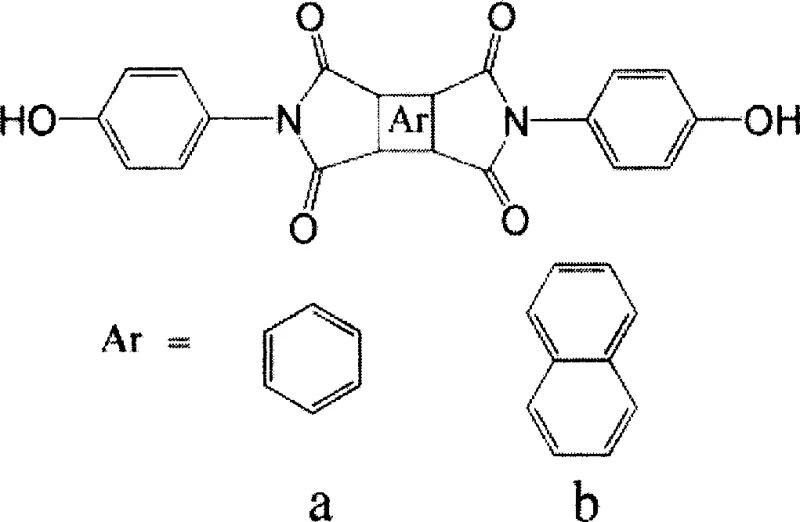
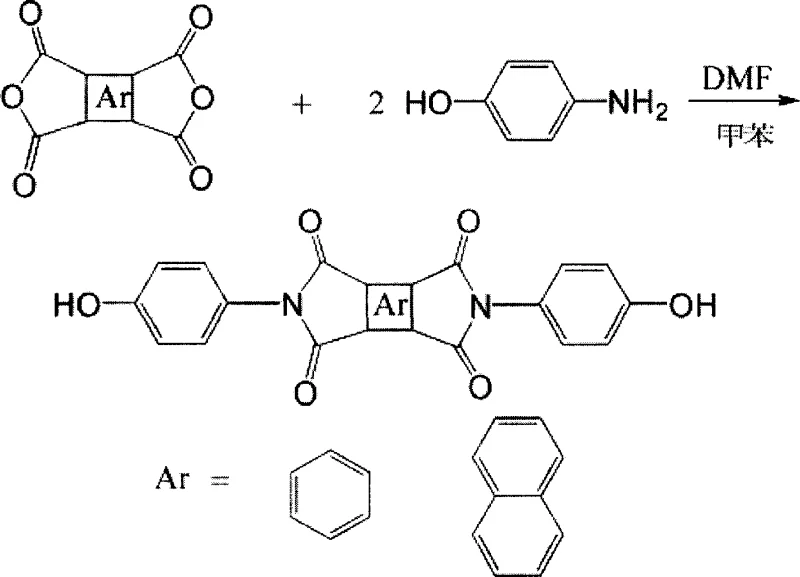
The development of high-performance polymers for aerospace and electronic applications relies heavily on the availability of specialized monomers with exceptional thermal stability. Patent CN100351254C introduces a robust synthetic methodology for producing bisphenol monomers containing an imide structure, which serve as critical precursors for advanced polyimides. Unlike traditional routes that often suffer from instability or harsh conditions, this novel approach utilizes a condensation reaction between dianhydrides and p-aminophenol in a controlled solvent system. The process leverages azeotropic dehydration to drive the equilibrium towards the desired imide formation, achieving yields ranging from 50% to 90%. For R&D directors and procurement specialists seeking a reliable polyimide monomer supplier, understanding the nuances of this synthesis is vital for securing a consistent supply of high-quality electronic chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyimides has been dominated by single-stage or two-stage methods that present significant logistical and technical challenges for large-scale manufacturing. The single-stage method, while conceptually simple, requires extremely harsh reaction conditions involving strong polarity and severe corrosive solvents such as meta-cresol, which pose serious safety and environmental hazards in a commercial plant setting. Furthermore, the two-stage method, which involves the initial formation of polyamic acid followed by thermal or chemical cyclization, suffers from inherent instability issues. The polyamic acid intermediate is notoriously sensitive to moisture and tends to decompose during storage, leading to batch-to-batch variability and compromised mechanical properties in the final polymer. These drawbacks necessitate strict inventory control and immediate usage, complicating supply chain management and increasing the risk of production delays due to precursor degradation.
The Novel Approach
The methodology described in the patent offers a transformative alternative by focusing on the synthesis of a stable, terminal hydroxyl imide monomer rather than the polymer itself. By reacting pyromellitic dianhydride or naphthalene-1,4,5,8-dianhydride directly with p-aminophenol, the process bypasses the unstable polyamic acid stage entirely. The use of DMF as a primary solvent, combined with a toluene azeotropic distillation step, allows for the efficient removal of water generated during the reaction, driving the equilibrium forward without requiring excessive temperatures initially. This results in a clear transition from solution to suspension as the product precipitates, simplifying isolation. This route not only enhances the stability of the intermediate monomer but also facilitates cost reduction in electronic chemical manufacturing by eliminating the need for corrosive solvents and reducing waste associated with unstable intermediates.
Mechanistic Insights into Thermal Imidization Condensation
The core of this synthesis lies in the nucleophilic attack of the amino group of p-aminophenol on the carbonyl carbon of the dianhydride, followed by a thermal dehydration cyclization to form the imide ring. The reaction begins at room temperature where the dianhydride and p-aminophenol form a soluble amic acid intermediate in DMF. The subsequent addition of toluene and heating to 110°C-120°C initiates the azeotropic removal of water, which is the byproduct of the initial amidation. As the water is removed, the system transitions from a clear solution to a suspension, indicating the onset of imidization and reduced solubility of the forming imide rings. The final high-temperature reflux at 140°C-150°C ensures the complete closure of the imide rings, a critical step for achieving the thermal stability required in aerospace applications. This mechanistic pathway ensures that the resulting monomer possesses the rigid imide structure necessary for high glass transition temperatures in the final polymer matrix.
Impurity control is intrinsically managed through the solvent selection and workup procedure detailed in the patent. The choice of DMF over other solvents like DMSO or NMP is not arbitrary; experimental data within the patent demonstrates that DMF yields significantly higher purity and conversion rates (up to 90% for naphthalene derivatives) compared to alternatives which dropped to near 50%. The rigorous washing protocol involving ethanol followed by boiling water effectively removes residual organic solvents and unreacted starting materials. Ethanol washes eliminate non-polar organic impurities, while the boiling water wash hydrolyzes and removes any remaining soluble salts or polar byproducts. This multi-step purification ensures that the final high-purity polyimide monomer meets the stringent specifications required for dielectric applications, minimizing the risk of ionic contamination that could degrade electronic performance.
How to Synthesize Bisphenol Imide Monomer Efficiently
Implementing this synthesis requires precise control over stoichiometry and temperature profiles to maximize yield and minimize side reactions. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors with standard agitation and heating capabilities. The key to success lies in the management of the azeotropic distillation phase, where the removal of water must be balanced against the thermal stability of the reactants. Operators must monitor the transition from a clear solution to a suspension closely, as this visual cue indicates the progress of the reaction. The following guide outlines the standardized operational parameters derived from the patent examples to ensure reproducible results.
- Mix dianhydride (pyromellitic or naphthalene-based) with p-aminophenol in DMF solvent at room temperature and stir vigorously for 2-3 hours until a clear solution forms.
- Add toluene as a water-carrying agent (1/3 to 1/2 of solvent volume) and reflux at 110°C-120°C for 3-4 hours under nitrogen protection to remove water.
- Evaporate the toluene, then continue refluxing the remaining mixture at 140°C-150°C for 4-5 hours to complete the imidization cyclization.
- Cool the suspension, wash the solid product with ethanol and boiling water to remove organic residues, then filter and dry at 80°C-100°C to obtain the pure monomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits regarding cost stability and operational reliability. The elimination of corrosive solvents like meta-cresol reduces the need for specialized corrosion-resistant equipment, thereby lowering capital expenditure (CAPEX) for manufacturing facilities. Additionally, the stability of the synthesized monomer allows for longer storage times compared to polyamic acid solutions, providing greater flexibility in production scheduling and inventory management. This flexibility translates directly into reducing lead time for high-purity polyimide monomers, as batches can be produced in advance and held in stock without significant degradation, ensuring continuity of supply even during fluctuations in raw material availability.
- Cost Reduction in Manufacturing: The process utilizes readily available raw materials such as pyromellitic dianhydride and p-aminophenol, which are commodity chemicals with stable pricing structures. By avoiding the use of expensive catalysts or complex multi-step purification sequences, the overall variable cost of production is significantly minimized. The high yield reported in the patent examples, particularly when using DMF, means that less raw material is wasted per unit of product, further enhancing the economic efficiency of the process. This efficiency allows suppliers to offer competitive pricing without compromising on the quality required for high-end applications.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate slight variations in temperature and stirring speed without catastrophic failure, makes the process highly suitable for contract manufacturing organizations (CMOs). The use of common solvents like DMF and toluene ensures that supply chain disruptions for exotic reagents are unlikely to halt production. Furthermore, the solid nature of the final product simplifies logistics; unlike liquid prepolymers that may require cold chain shipping, the powdered monomer can be transported in standard packaging, reducing shipping costs and expanding the geographic reach of the supply network.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, as demonstrated by the straightforward transition from gram-scale examples to potential tonnage production. The workup procedure relies on filtration and washing rather than complex chromatographic separations, which are difficult to scale. From an environmental perspective, the ability to recover and recycle solvents like toluene and DMF through distillation aligns with modern green chemistry principles. This reduces the volume of hazardous waste generated, simplifying compliance with environmental regulations and lowering waste disposal costs for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on solvent selection, reaction kinetics, and product quality assurance. Understanding these factors is essential for technical teams evaluating the feasibility of integrating this monomer into their existing polymerization workflows.
Q: Why is DMF preferred over other polar aprotic solvents for this synthesis?
A: While solvents like DMSO, NMP, and DMAc can be used, the patent data indicates that Dimethylformamide (DMF) provides the optimal balance of solubility and reaction kinetics, resulting in the highest yields (up to 90%) compared to alternatives which yielded as low as 51%.
Q: What is the critical function of adding toluene during the reaction?
A: Toluene acts as an azeotropic agent to continuously remove water generated during the condensation reaction. This shifts the chemical equilibrium towards the product side, ensuring high conversion rates and preventing the hydrolysis of the sensitive anhydride intermediates.
Q: How does this method improve upon traditional polyamic acid routes?
A: Traditional two-step methods involve unstable polyamic acid intermediates that are sensitive to moisture and storage conditions. This direct condensation method produces a stable, terminal hydroxyl imide monomer that simplifies storage and allows for more controlled nucleophilic polycondensation in downstream polymerization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphenol Imide Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of next-generation polyimides for aerospace and electronics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of bisphenol imide monomer meets the exacting standards required for dielectric and thermal applications. Our commitment to quality ensures that your downstream polymerization processes proceed without interruption or variability.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that can help you streamline your manufacturing operations. Contact us today to discuss how our advanced synthesis capabilities can support your long-term strategic goals in the high-performance materials sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
