Advanced Recovery of 1-(3,3-Dimethylcyclohexyl) Ethanol for Sustainable Musk Eden Production
Advanced Recovery of 1-(3,3-Dimethylcyclohexyl) Ethanol for Sustainable Musk Eden Production
The global fragrance industry is increasingly driven by the dual mandates of cost efficiency and environmental sustainability, particularly in the synthesis of high-value musk compounds. A pivotal development in this sector is detailed in patent CN112898156A, which introduces a sophisticated method for recovering 1-(3,3-dimethylcyclohexyl) ethanol, a critical intermediate in the manufacture of Musk Eden. This technology addresses a longstanding inefficiency in fragrance synthesis where significant quantities of expensive starting materials are lost to side reactions, ending up as heavy distillation residues. By implementing a novel hydrolysis protocol utilizing molecular sieve catalysts, manufacturers can now reclaim these trapped intermediates with exceptional purity. This breakthrough not only optimizes the atom economy of the process but also establishes a new benchmark for sustainable chemical manufacturing in the fine fragrance sector, offering a compelling value proposition for procurement and supply chain leaders seeking to reduce their carbon footprint while securing raw material availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
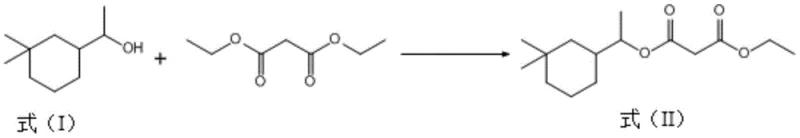
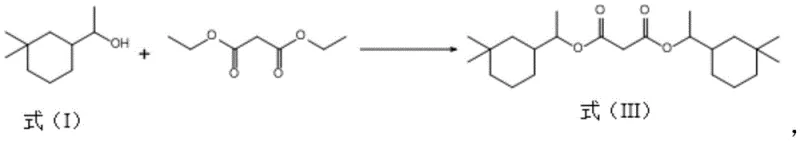
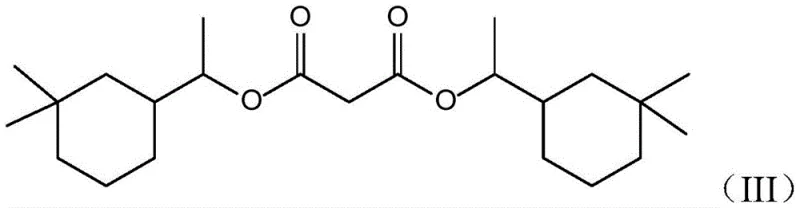
Traditionally, the synthesis of Musk Eden involves the esterification of 1-(3,3-dimethylcyclohexyl) ethanol with diethyl malonate. However, this reaction is prone to competitive side pathways that generate heavy byproducts, specifically the bis-substituted diester known as Formula (III). In conventional processing setups, these heavy ends accumulate in the distillation residues after the target mono-ester is collected. Historically, these residues were often treated as hazardous waste or low-value fuel, representing a direct loss of the expensive cyclohexyl alcohol starting material. Furthermore, traditional methods for breaking down these esters often relied on harsh homogeneous catalysts like concentrated sulfuric acid or strong alkalis. These aggressive reagents necessitate extensive neutralization and washing steps, generating large volumes of saline wastewater and complicating the purification of the recovered alcohol, thereby eroding the economic viability of any recovery attempt.
The Novel Approach
The methodology disclosed in CN112898156A fundamentally shifts this paradigm by introducing a selective catalytic hydrolysis step tailored for these specific distillation residues. Instead of discarding the heavy ends containing Formula (III), the process subjects them to a controlled hydrolysis reaction using solid acid catalysts, specifically various types of molecular sieves such as 4A, ZSM-5, or Beta zeolites. This approach allows for the cleavage of the ester bonds in the byproduct under mild thermal conditions (60°C to 100°C), effectively regenerating the free 1-(3,3-dimethylcyclohexyl) ethanol. The use of heterogeneous molecular sieve catalysts eliminates the need for corrosive liquid acids and the subsequent neutralization waste streams. As illustrated in the reaction pathway below, this targeted recovery transforms a waste stream into a high-value feedstock, closing the loop on material usage and significantly enhancing the overall process economics.
Mechanistic Insights into Molecular Sieve Catalyzed Hydrolysis
The core of this technological advancement lies in the unique properties of the molecular sieve catalysts employed. Unlike traditional liquid acids that promote non-selective degradation and charring, molecular sieves possess a defined pore structure and tunable acidity that facilitates the specific hydrolysis of the bulky bis-ester molecules. The silicon-to-aluminum ratio of the catalyst, which can range from 1:2 to 1:500, is a critical parameter that governs the acid strength and hydrophobicity of the active sites. This tunability ensures that the catalyst is active enough to drive the hydrolysis of the sterically hindered ester groups in Formula (III) to completion—achieving conversion rates up to 100%—while minimizing side reactions such as dehydration of the alcohol or polymerization. The solid nature of the catalyst also simplifies the work-up procedure; post-reaction, the catalyst can be easily separated from the reaction mixture via filtration, leaving a clean organic phase ready for purification.
From an impurity control perspective, this mechanism offers distinct advantages for R&D teams focused on product quality. The selectivity of the molecular sieve prevents the formation of complex tarry byproducts often associated with high-temperature acid hydrolysis. Consequently, the recovered 1-(3,3-dimethylcyclohexyl) ethanol exhibits a remarkably clean impurity profile, with gas chromatography data indicating purities exceeding 98%. This high level of purity is essential because the recovered alcohol is intended for recycling back into the primary synthesis of Musk Eden. Any carryover of impurities could poison the primary synthesis catalyst or affect the olfactory profile of the final fragrance. The gentle yet effective nature of the molecular sieve catalysis ensures that the regenerated alcohol retains its structural integrity and sensory characteristics, making it indistinguishable from virgin material in downstream applications.
How to Synthesize 1-(3,3-Dimethylcyclohexyl) Ethanol Efficiently
The implementation of this recovery protocol requires precise control over distillation and hydrolysis parameters to maximize yield. The process begins with the careful fractionation of the crude reaction mass to isolate the heavy residue containing the bis-ester. Following isolation, the residue is subjected to hydrolysis with a selected molecular sieve catalyst at optimized temperatures.
- Perform reduced pressure distillation on the crude reaction mixture from the Musk Eden synthesis to separate the target product and collect distillation residues containing the bis-ester byproduct (Formula III).
- Subject the collected distillation residues to a hydrolysis reaction using a specific molecular sieve catalyst (e.g., 4A, ZSM-5) at temperatures between 60°C and 100°C.
- Purify the resulting hydrolysis product via rectification under reduced pressure (360-580 Pa) to isolate high-purity 1-(3,3-dimethylcyclohexyl) ethanol for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recovery technology translates into tangible strategic benefits beyond simple technical metrics. The primary advantage is the substantial reduction in raw material volatility. By internalizing the recovery of 1-(3,3-dimethylcyclohexyl) ethanol, manufacturers decrease their dependence on external suppliers for this specific intermediate, thereby insulating the production line from market price fluctuations and supply disruptions. This self-sufficiency enhances supply chain resilience, ensuring consistent production schedules even when upstream feedstock availability is constrained. Moreover, the ability to recycle a significant portion of the starting alcohol effectively lowers the net consumption of raw materials per kilogram of finished Musk Eden, driving down the variable cost of goods sold (COGS) without compromising product quality.
- Cost Reduction in Manufacturing: The elimination of expensive waste disposal fees and the recovery of high-value alcohol create a dual financial benefit. Traditional disposal of chemical residues incurs significant logistical and regulatory costs, which are entirely avoided here. Furthermore, by converting what was once a waste liability into a productive asset, the effective yield of the overall process is dramatically improved. This efficiency gain means that less fresh raw material needs to be purchased to achieve the same output volume, directly improving the gross margin of the fragrance manufacturing operation.
- Enhanced Supply Chain Reliability: Integrating this recovery step creates a more robust circular economy within the plant. It reduces the total volume of materials that need to be sourced externally, simplifying inventory management and reducing the risk of production stoppages due to raw material shortages. The simplicity of the molecular sieve catalyst handling also means that the process is less susceptible to delays caused by the procurement of hazardous reagents like concentrated sulfuric acid, streamlining the operational workflow and ensuring a steady flow of intermediates to the synthesis reactors.
- Scalability and Environmental Compliance: The use of solid molecular sieve catalysts aligns perfectly with modern green chemistry principles and stringent environmental regulations. Unlike liquid acid processes that generate vast amounts of acidic wastewater requiring neutralization, this method produces minimal aqueous waste. This significantly reduces the burden on wastewater treatment facilities and lowers the risk of environmental compliance violations. The scalability of the process is further supported by the ease of catalyst separation and reuse, making it highly suitable for large-scale industrial production ranging from pilot batches to multi-ton annual capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recovery technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational realities and benefits.
Q: What is the primary advantage of using molecular sieves over traditional acid/base catalysts for this recovery?
A: Molecular sieves offer superior selectivity and catalytic activity without generating hazardous waste alkali liquor or requiring complex neutralization steps, significantly simplifying the downstream purification process and reducing environmental impact.
Q: Can the recovered 1-(3,3-dimethylcyclohexyl) ethanol be reused directly in Musk Eden synthesis?
A: Yes, the patent data indicates that the recovered alcohol achieves high purity (over 98%) after rectification, making it chemically equivalent to fresh raw material and fully suitable for recycling back into the primary esterification reaction.
Q: How does this recovery process impact the overall cost structure of Musk Eden production?
A: By converting high-boiling distillation residues (previously treated as waste) back into valuable starting alcohol, the process drastically reduces raw material consumption per unit of final product and lowers waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Musk Eden Supplier
At NINGBO INNO PHARMCHEM, we recognize that the future of fine chemical manufacturing lies in the integration of innovative process technologies with rigorous quality standards. Our technical team has extensively analyzed advanced recovery protocols like those described in CN112898156A to optimize our own production lines for fragrance intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch of Musk Eden we deliver meets the highest standards of consistency and purity. Our state-of-the-art rigorous QC labs utilize advanced analytical techniques to verify the identity and purity of our intermediates, guaranteeing that our products perform flawlessly in your final formulations.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates how our optimized manufacturing processes can reduce your total procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project requirements, ensuring a secure and efficient supply of high-quality fragrance ingredients.
