Advanced FeCl3-Catalyzed Bromination for High-Purity Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for functionalized intermediates that enable rapid diversification of lead compounds. Patent CN101948405B introduces a significant advancement in this domain by detailing a preparation method for bromoaryl azide compounds, which serve as critical building blocks in modern medicinal chemistry. This technology leverages a Lewis acid-catalyzed bromination strategy that overcomes the limitations of historical methods, providing a pathway to high-purity intermediates essential for click chemistry and heterocycle synthesis. For R&D directors and procurement specialists, understanding the nuances of this patent is vital, as it represents a shift towards safer, more efficient manufacturing protocols that align with green chemistry principles while maintaining rigorous quality standards required for GMP production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the bromination of aryl azides has been a challenging transformation plagued by inefficiency and safety concerns. Early literature, such as the work by Smith in 1962, relied on the use of elemental liquid bromine, a hazardous reagent that requires stringent safety controls and often results in poor regioselectivity and modest yields, reported as low as 54 percent. Furthermore, alternative methods utilizing reagents like 1,4-dioxane dibromide have shown severe limitations in substrate scope, effectively restricting their utility to aryl azides containing only electron-donating groups. These conventional approaches often necessitate harsh reaction conditions that can compromise the stability of the sensitive azide functionality, leading to decomposition or the formation of complex impurity profiles that are difficult to remove during downstream processing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a catalytic system comprising anhydrous ferric chloride (FeCl3) and N-bromosuccinimide (NBS) in 1,2-dichloroethane (DCE). This novel approach operates under significantly milder thermal conditions, typically around 80°C, which preserves the integrity of the azide group while ensuring high conversion rates. The use of NBS as a solid brominating agent simplifies handling and dosing compared to liquid bromine, thereby enhancing operational safety and reproducibility on a commercial scale. This system demonstrates exceptional versatility, successfully accommodating a wide array of substituents including electron-withdrawing groups like esters and halogens, which were previously difficult to functionalize efficiently.
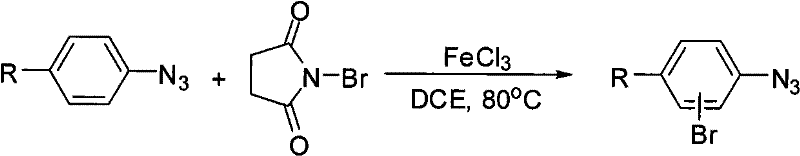
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological breakthrough lies in the synergistic interaction between the Lewis acid catalyst and the brominating agent. Anhydrous ferric chloride acts as a potent Lewis acid, coordinating with the carbonyl oxygen of N-bromosuccinimide to enhance the electrophilicity of the bromine atom. This activation facilitates a controlled electrophilic aromatic substitution on the aryl azide ring. The mechanism ensures that the bromination occurs selectively at the position ortho to the azide group or directed by other substituents, minimizing the formation of poly-brominated byproducts. This precise control over regioselectivity is paramount for pharmaceutical applications, where isomeric purity directly impacts the biological activity and safety profile of the final active pharmaceutical ingredient (API).
Furthermore, the reaction conditions are optimized to suppress side reactions that typically plague azide chemistry, such as thermal decomposition or reduction. The choice of 1,2-dichloroethane as the solvent provides an ideal medium that solubilizes both the organic substrates and the inorganic catalyst, ensuring homogeneous reaction kinetics. The patent data indicates that this system is robust enough to handle diverse structural motifs, including heterocycles like thiazoles and fused ring systems like naphthalenes, expanding the chemical space accessible to process chemists. This broad substrate tolerance suggests a fundamental reliability in the catalytic cycle, making it a dependable tool for synthesizing complex molecular architectures.
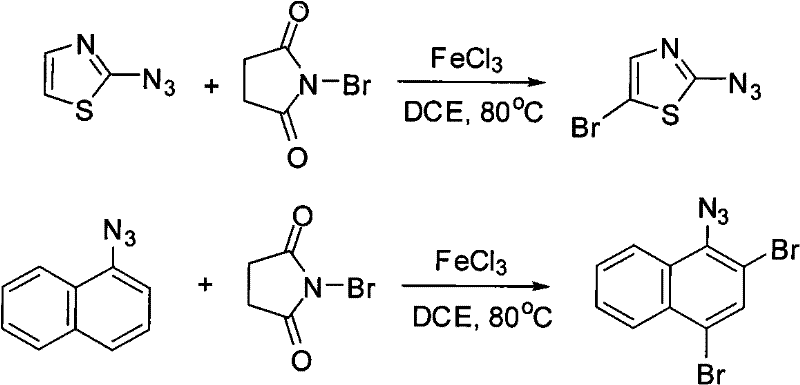
How to Synthesize Bromoaryl Azide Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process is designed to be straightforward, involving the mixing of reagents in a standard reactor followed by a controlled heating phase. The simplicity of the workup procedure, which involves aqueous quenching and standard organic extraction, further underscores its suitability for industrial adoption. For detailed operational parameters and specific molar ratios tailored to different substrates, please refer to the standardized synthesis guide below which encapsulates the critical steps derived from the patent examples.
- Charge a reactor with 1,2-dichloroethane (DCE), anhydrous ferric chloride (catalyst), N-bromosuccinimide (NBS), and the aryl azide substrate.
- Stir the reaction mixture at 80°C for 4 to 24 hours to allow complete bromination.
- Quench with water, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented method offers substantial advantages that resonate deeply with procurement managers and supply chain heads focused on cost optimization and risk mitigation. The transition from hazardous liquid bromine to solid NBS not only reduces the regulatory burden associated with storing and transporting dangerous goods but also streamlines the logistics of raw material sourcing. The use of iron-based catalysts, which are abundant and inexpensive compared to precious metal alternatives like palladium or rhodium, drives down the direct material costs significantly. Additionally, the mild reaction conditions reduce energy consumption and minimize the wear and tear on reactor vessels, contributing to lower overall operational expenditures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like NBS and FeCl3 result in a drastically simplified cost structure. By avoiding the need for specialized corrosion-resistant equipment required for liquid bromine handling, capital expenditure for new production lines is also minimized. The high efficiency of the reaction reduces solvent usage per kilogram of product, further enhancing the economic viability of the process for large-scale manufacturing.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including aryl azides and NBS, are widely available from multiple global suppliers, reducing the risk of supply chain disruptions. The robustness of the reaction means that batch-to-batch variability is minimized, ensuring consistent delivery schedules to downstream customers. This reliability is crucial for maintaining continuous production flows in the fast-paced pharmaceutical and agrochemical sectors where delays can be costly.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional bromination methods, aligning with increasingly strict environmental regulations. The workup involves standard aqueous washes and distillation, which are easily scalable from pilot plant to multi-ton production without requiring complex purification technologies. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand without compromising on quality or compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bromination technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on its practical application in industrial settings. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing manufacturing portfolios.
Q: Why is the FeCl3/NBS system superior to traditional liquid bromine methods?
A: Traditional methods using liquid bromine often suffer from low yields (around 54%) and harsh handling requirements. The FeCl3/NBS system operates under milder conditions (80°C), offers broader substrate scope including electron-withdrawing groups, and eliminates the safety hazards associated with handling elemental bromine.
Q: What is the substrate scope for this bromination protocol?
A: This protocol is highly versatile, successfully brominating aryl azides with various substituents such as chloro, bromo, carboxylate, methyl, and methoxy groups. It also extends to heterocyclic systems like thiazoles and polycyclic aromatics like naphthalenes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses readily available raw materials and standard solvents like 1,2-dichloroethane. The workup involves simple extraction and distillation, making it easily adaptable for commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromoaryl Azide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the FeCl3-catalyzed bromination described in CN101948405B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of bromoaryl azide meets the exacting standards required for drug development and commercial manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in chemical innovation and supply chain excellence.
