Advanced Graphene-Catalyzed Synthesis of 4,4'-Diaminodiphenyl Methane for Industrial Scale-Up
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable catalytic processes, particularly in the synthesis of high-value aromatic amines. Patent CN115850090B introduces a groundbreaking methodology for the preparation of 4,4'-diaminodiphenyl methane (MDA) utilizing a novel graphene-based catalyst. This technology addresses critical pain points associated with traditional manufacturing routes, such as severe equipment corrosion and the generation of hazardous saline wastewater. By leveraging the unique acidic properties of edge-carboxylated graphene, this process enables the efficient condensation of aniline and formaldehyde under mild conditions. The structural integrity and functional utility of the target molecule, 4,4'-diaminodiphenyl methane, are paramount for its downstream applications in high-performance polymers and pharmaceutical intermediates.
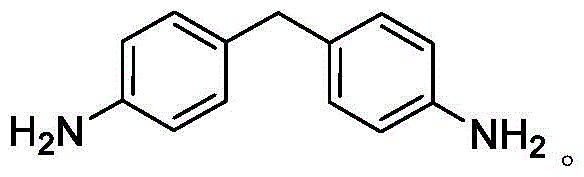
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4,4'-diaminodiphenyl methane has relied heavily on the use of strong liquid mineral acids, most notably concentrated hydrochloric acid, as catalysts. While these traditional methods offer the advantage of established protocols and relatively low raw material costs, they suffer from significant operational and environmental drawbacks. The use of concentrated hydrochloric acid necessitates the use of highly corrosion-resistant equipment, driving up capital expenditure for reactor maintenance and replacement. Furthermore, post-reaction processing requires substantial quantities of alkali liquor to neutralize the residual acid, resulting in the generation of large volumes of salt-containing wastewater that poses a severe burden on environmental treatment facilities. Alternative solid acid catalysts, such as molecular sieves or phosphotungstic acid, have been explored to mitigate these issues, but they often require excessively high reaction temperatures exceeding 160°C, leading to high energy consumption and limited catalyst recyclability.
The Novel Approach
In stark contrast to these legacy technologies, the graphene-catalyzed synthesis route described in the patent offers a streamlined, environmentally benign alternative that operates under significantly milder conditions. The core innovation lies in the utilization of edge high-carboxylation reduced graphene oxide, which acts as a robust solid acid catalyst capable of driving the condensation reaction at temperatures as low as 70-80°C. This approach eliminates the need for corrosive liquid acids entirely, thereby preserving equipment integrity and simplifying the downstream purification process. The reaction scheme is remarkably clean, utilizing aniline not only as a reactant but also as the solvent, which removes the necessity for additional volatile organic compounds. The visual representation of this efficient transformation highlights the direct conversion of aniline and paraformaldehyde into the target diamine with high atom economy.
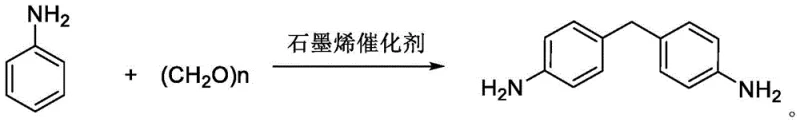
Mechanistic Insights into Graphene-Catalyzed Condensation
The efficacy of this synthetic route is rooted in the unique physicochemical properties of the graphene catalyst, specifically the abundance of carboxyl groups located at the edges of the graphene sheets. These carboxyl functionalities impart strong solid acidity to the material, mimicking the proton-donating capability of liquid mineral acids but within a heterogeneous framework. During the reaction, these acidic sites activate the formaldehyde species, facilitating the electrophilic attack on the electron-rich aromatic ring of the aniline. This mechanism promotes the formation of the methylene bridge between the two phenyl rings with high regioselectivity, favoring the para-para substitution pattern required for 4,4'-diaminodiphenyl methane. The heterogeneous nature of the catalyst ensures that the active sites are accessible to the reactants while remaining distinct from the product phase, which is crucial for easy separation.
From an impurity control perspective, the mild reaction temperature of 70-80°C plays a pivotal role in suppressing side reactions that typically plague high-temperature acid-catalyzed processes. Traditional methods operating at elevated temperatures often lead to the formation of polymeric by-products or ortho-substituted isomers, which complicate purification and reduce overall yield. The graphene catalyst's specific surface chemistry appears to stabilize the transition state for the desired para-coupling, resulting in crude product purities that exceed 91% prior to recrystallization. Furthermore, the ability to filter the catalyst while hot prevents the re-adsorption of products or the degradation of the catalyst structure during cooling, ensuring that the recovered catalyst retains its activity for subsequent cycles. This mechanistic precision translates directly into a cleaner impurity profile, reducing the burden on downstream crystallization and drying units.
How to Synthesize 4,4'-Diaminodiphenyl Methane Efficiently
The implementation of this graphene-catalyzed protocol requires precise control over stoichiometry and thermal parameters to maximize yield and catalyst longevity. The process begins with the charging of aniline, paraformaldehyde, water, and the graphene catalyst into a reactor, followed by heating to the optimal range of 70-80°C. Maintaining this temperature window is critical; temperatures that are too low may result in incomplete conversion, while excessive heat could compromise the selectivity advantages of the graphene surface. The detailed standardized synthesis steps, including specific workup procedures for catalyst recovery and product isolation, are outlined in the technical guide below.
- Charge aniline, paraformaldehyde, water, and edge high-carboxylation reduced graphene oxide catalyst into a reactor.
- Heat the mixture to 70-80°C and maintain reaction for 1-2 hours without organic solvents.
- Filter the hot mixture to recover the catalyst, distill filtrate to recover aniline, and recrystallize the heavy component to obtain pure MDA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this graphene-catalyzed technology represents a strategic opportunity to optimize both operational expenditures and supply reliability. The elimination of concentrated hydrochloric acid from the process flow fundamentally alters the cost structure of manufacturing 4,4'-diaminodiphenyl methane. By removing the need for expensive corrosion-resistant alloys in reactors and piping, capital depreciation costs are significantly reduced. Moreover, the absence of a neutralization step means that the facility no longer needs to procure large quantities of caustic soda for waste treatment, nor does it incur the disposal costs associated with tons of saline effluent. This streamlining of the chemical bill of materials leads to substantial cost savings in raw material procurement and waste management overheads.
- Cost Reduction in Manufacturing: The economic benefits of this process extend beyond simple raw material substitution. Because the graphene catalyst is a heterogeneous solid, it can be separated from the reaction mixture via simple filtration, allowing for immediate recovery and reuse. The patent data indicates that the catalyst maintains its activity for more than 10 cycles, which drastically amortizes the initial cost of the nanomaterial over a large production volume. Additionally, the process does not require organic solvents, as aniline serves as the reaction medium, further reducing solvent purchase and recovery costs. These factors combine to create a leaner manufacturing process with a lower variable cost per kilogram of finished product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the reaction conditions. Operating at mild temperatures (70-80°C) reduces the energy load on the facility, making the process less susceptible to fluctuations in utility costs or steam availability. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which shortens the overall production cycle time and increases throughput capacity. This efficiency allows manufacturers to respond more agilely to market demand spikes for high-purity pharmaceutical intermediates or polymer monomers without the bottleneck of complex purification trains.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions continue to tighten globally, the environmental profile of this synthesis route offers a distinct competitive advantage. The reduction in wastewater generation and the elimination of corrosive acid fumes simplify the permitting process for new production lines or capacity expansions. The solid waste generated is primarily the spent catalyst, which is minimal in volume compared to the sludge produced by liquid acid neutralization. This alignment with green chemistry principles not only ensures compliance with current environmental standards but also future-proofs the supply chain against increasingly stringent ecological regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this graphene-catalyzed synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring that decision-makers have access to accurate and actionable information. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing manufacturing frameworks.
Q: What are the primary advantages of using graphene catalyst over concentrated hydrochloric acid for MDA synthesis?
A: The graphene catalyst eliminates the need for corrosive liquid acids like concentrated HCl, thereby removing the requirement for extensive neutralization steps and reducing salt-containing wastewater generation. Additionally, the solid graphene catalyst can be filtered and reused more than 10 times, significantly lowering material costs and environmental impact.
Q: What represent the optimal reaction conditions for this graphene-catalyzed process?
A: The process operates efficiently at mild temperatures between 70-80°C for 1 to 2 hours. The molar ratio of paraformaldehyde to aniline is typically maintained between 1:2 and 1:5, with water added to facilitate the reaction medium without requiring additional organic solvents.
Q: How does this method impact the purity and yield of the final 4,4'-diaminodiphenyl methane product?
A: Experimental data indicates that this method achieves yields up to 91.7% with product purity reaching 99.3% after recrystallization. The high selectivity of the graphene catalyst minimizes by-product formation compared to traditional strong acid methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Diaminodiphenyl Methane Supplier
The technological advancements detailed in patent CN115850090B underscore the immense potential of nanocarbon materials in modern organic synthesis, paving the way for cleaner and more efficient production of critical chemical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory-scale protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this graphene-catalyzed route can be realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4,4'-diaminodiphenyl methane meets the exacting standards required for high-performance applications.
We invite global partners to collaborate with us to optimize their supply chains through the adoption of this advanced synthesis technology. By leveraging our process development capabilities, we can help you achieve significant efficiencies and cost reductions in your manufacturing operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your strategic sourcing decisions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
