Scalable Synthesis of Anthracycline Toxoid Derivatives for Next-Gen ADC Manufacturing
Scalable Synthesis of Anthracycline Toxoid Derivatives for Next-Gen ADC Manufacturing
The rapidly evolving landscape of Antibody Drug Conjugates (ADCs) demands highly potent and stable cytotoxins capable of overcoming multidrug resistance in tumor cells. Patent CN115651043A introduces a groundbreaking preparation method for anthracycline toxoid derivatives, specifically targeting the synthesis of high-potency metabolites like PNU-159682. This technology addresses critical bottlenecks in the current supply chain by utilizing inexpensive, commercially available starting materials to achieve efficient synthesis under mild conditions. The core innovation lies in a streamlined reaction pathway that bypasses the cumbersome multi-step sequences traditionally associated with modifying the anthracycline sugar moiety. By enabling a direct one-step formation of the complex pyranooxazolooxazine ring system, this patent offers a robust solution for producing next-generation ADC payloads with improved cost-efficiency and scalability.
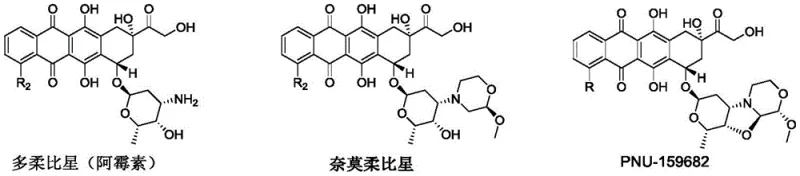
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of morpholino-anthracycline derivatives like PNU-159682 has been plagued by excessive complexity and poor scalability. Traditional routes typically commence with doxorubicin or nemorubicin, requiring a sequence of morpholine ring closure, selective N-oxidation, and subsequent oxazole ring closure. These multi-step processes often necessitate the introduction and removal of protecting groups to manage the reactivity of various hydroxyl functionalities on the anthracycline scaffold. Such lengthy synthetic pathways not only result in relatively low overall yields due to cumulative losses at each stage but also generate a complex impurity profile that is difficult to control. Furthermore, the intermediates involved in these traditional methods often exhibit poor stability, restricting reaction scales to mere milligrams and rendering them unsuitable for the industrial production volumes required by the booming ADC market.
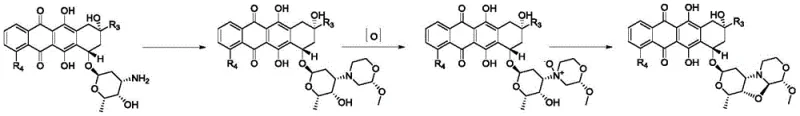
The Novel Approach
In stark contrast to the arduous conventional pathways, the method disclosed in CN115651043A employs a convergent strategy that dramatically simplifies the molecular construction. The novel approach utilizes a readily available sugar derivative, methyl-α-D-xylopyranose, which is oxidatively cleaved to generate a reactive dialdehyde intermediate in situ. This electrophilic species is then immediately engaged in a condensation reaction with an amino-substituted anthracycline precursor. This ingenious design allows for the simultaneous formation of the requisite heterocyclic rings in a single operational step, effectively collapsing what was once a multi-stage sequence into a concise transformation. By strictly controlling reaction parameters such as temperature, pH, and reagent stoichiometry, this method ensures high regioselectivity and minimizes side reactions, thereby securing a reliable supply of the target toxoid derivative.
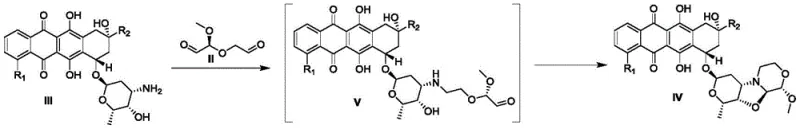
Mechanistic Insights into Reductive Amination and Cyclization
The chemical elegance of this synthesis rests on the precise orchestration of oxidative cleavage followed by reductive amination and spontaneous cyclization. Initially, the vicinal diol system of the methyl-α-D-xylopyranose starting material undergoes oxidative cleavage mediated by periodate salts, generating a highly reactive dialdehyde species. This intermediate possesses the exact carbon framework required to bridge the amino and hydroxyl groups present on the anthracycline substrate. Upon mixing, the primary amine of the anthracycline attacks one of the aldehyde carbons to form an imine or iminium ion, while the proximal hydroxyl group participates in hemiacetal formation with the second aldehyde. The presence of a mild reducing agent, such as sodium cyanoborohydride or sodium triacetoxyborohydride, stabilizes the newly formed C-N bond, locking the structure into the desired fused pyranooxazolooxazine architecture. This cascade mechanism proceeds efficiently under mild acidic to neutral conditions, preserving the sensitive quinone and glycosidic linkages of the anthracycline core.
From an impurity control perspective, the mechanism offers significant advantages by limiting the exposure of the drug substance to harsh oxidative environments. In traditional routes, the N-oxidation step is particularly prone to generating over-oxidized byproducts and degrading the anthracycline chromophore. The new method avoids discrete oxidation steps on the drug molecule itself, confining oxidative chemistry to the inexpensive sugar precursor. Consequently, the impurity spectrum is markedly simplified, with fewer degradation products related to the anthracycline aglycone. This mechanistic cleanliness translates directly to easier downstream processing, as the crude reaction mixture contains fewer structurally similar impurities that are notoriously difficult to separate via standard chromatographic techniques, ultimately ensuring higher final product purity.
How to Synthesize Anthracycline Toxoid Derivatives Efficiently
The execution of this synthesis requires careful attention to the preparation of the unstable dialdehyde intermediate and the subsequent coupling conditions. The process is designed to be telescoped, meaning the intermediate generated in the first step is used directly in the second step without isolation, which mitigates stability issues and reduces handling time. Operators must maintain strict temperature control during the oxidative cleavage to prevent over-oxidation or decomposition of the sugar derivative. Following the generation of the dialdehyde, the addition of the anthracycline substrate and the reducing agent must be managed to ensure complete conversion while minimizing the formation of reduced alcohol byproducts. Detailed standardized operating procedures for this specific pathway are outlined below to ensure reproducibility and safety.
- Oxidative Cleavage: React methyl-α-D-xylopyranose with sodium periodate in a suitable solvent system to generate the reactive dialdehyde intermediate.
- One-Pot Condensation: Combine the crude dialdehyde intermediate with the amino-anthracycline substrate in the presence of a reducing agent and buffer.
- Workup and Purification: Quench the reaction, extract the organic phase, and purify via chromatography to isolate the target pyranooxazolooxazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this patented methodology represents a strategic opportunity to optimize the cost structure and reliability of ADC cytotoxin sourcing. The most immediate impact is the drastic reduction in raw material costs, as the process replaces expensive, specialized intermediates with commodity chemicals like methyl-α-D-xylopyranose and bulk anthracyclines. This shift decouples production from the volatile supply chains of niche fine chemicals, ensuring a more stable and predictable input flow. Furthermore, the consolidation of multiple synthetic steps into a single pot operation significantly lowers labor costs and reduces the consumption of solvents and energy per kilogram of product. These efficiencies compound to offer a substantially lower cost of goods sold (COGS), providing a competitive edge in the pricing of final ADC therapeutics.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps removes the need for additional reagents and the associated waste disposal costs. By avoiding the isolation of unstable intermediates, the process reduces material loss and equipment occupancy time. The use of common, non-proprietary solvents further drives down operational expenses, making the overall manufacturing economics significantly more favorable compared to legacy routes that rely on exotic reagents and complex purification sequences.
- Enhanced Supply Chain Reliability: Reliance on widely available starting materials mitigates the risk of supply disruptions often associated with custom-synthesized building blocks. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor variations in environmental parameters. This resilience ensures consistent delivery timelines, which is critical for maintaining the production schedules of downstream ADC conjugation facilities that operate on tight just-in-time inventories.
- Scalability and Environmental Compliance: The simplified workflow inherently supports scale-up from pilot batches to commercial tonnage without the need for specialized high-pressure or cryogenic equipment. The reduction in synthetic steps leads to a lower E-factor (mass of waste per mass of product), aligning with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile facilitates smoother regulatory approvals and reduces the burden on waste treatment infrastructure, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement teams negotiating supply agreements.
Q: What are the primary advantages of this new synthesis route over traditional methods?
A: The new route eliminates the need for multiple protection and deprotection steps required in traditional morpholine ring closures and N-oxidations. It utilizes cheap, commercially available starting materials and achieves the target structure in significantly fewer steps, enhancing scalability.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent emphasizes the use of mild reaction conditions (-20°C to 40°C) and common solvents like dichloromethane and acetonitrile. The simplification of the synthetic pathway reduces operational complexity, making it highly amenable to scale-up from kilogram to tonnage levels.
Q: How does this method impact the impurity profile of the final ADC cytotoxin?
A: By reducing the number of synthetic steps and avoiding harsh oxidation conditions on the sensitive anthracycline core, the method effectively controls the complexity of the impurity spectrum. This leads to a cleaner crude product and simplifies downstream purification efforts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anthracycline Toxoid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this streamlined synthesis route for the ADC industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reductive conditions required for this chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee the quality of every batch. We are committed to delivering high-purity anthracycline toxoid intermediates that meet the exacting standards of global pharmaceutical developers.
We invite you to engage with our technical procurement team to discuss how this innovative method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your ADC development timelines and optimize your manufacturing budget.
