Scalable Electro-Oxidation Technology for High-Purity Agrochemical Intermediate Manufacturing
Scalable Electro-Oxidation Technology for High-Purity Agrochemical Intermediate Manufacturing
The global agrochemical industry is undergoing a significant paradigm shift towards greener, more sustainable manufacturing processes, driven by stringent environmental regulations and the need for cost-effective supply chains. In this context, patent CN113308705B presents a groundbreaking advancement in the synthesis of key intermediates for strobilurin fungicides, specifically targeting the production of methyl phenylglyoxylate derivatives. This technology replaces traditional, hazardous chemical oxidants with clean electrical energy, utilizing an N-hydroxyphthalimide (NHPI) mediated electro-oxidation system. For R&D directors and procurement managers alike, this represents a critical opportunity to enhance the purity profile of active pharmaceutical ingredients (APIs) and agrochemical intermediates while simultaneously mitigating the risks associated with handling toxic reagents like cyanides or heavy metal oxidants. The method demonstrates exceptional versatility, applicable to various methyl-substituted phenylacetates, ensuring a robust supply of high-quality precursors for major fungicides such as Kresoxim-methyl and Trifloxystrobin.
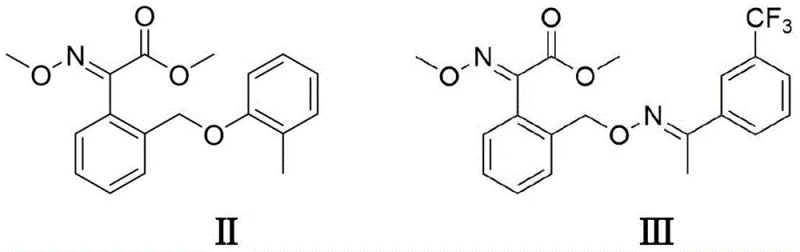
As illustrated in the structural diagrams, the target molecules serve as the foundational backbone for some of the world's most widely used agricultural fungicides. The ability to synthesize the critical alpha-keto ester motif efficiently is paramount for maintaining the continuity of the global food security supply chain. By leveraging electro-organic synthesis, manufacturers can bypass the complex waste treatment protocols associated with stoichiometric oxidants, thereby aligning their production capabilities with modern ESG (Environmental, Social, and Governance) goals. This patent not only offers a technical solution but also provides a strategic advantage for companies looking to position themselves as reliable agrochemical intermediate suppliers in a increasingly regulated market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methyl 2-methylphenylglyoxylate has relied on several chemically intensive pathways that present significant operational and environmental challenges. Early methods, such as those described in US patent 5145980, utilized potassium cyanide for cyanation followed by oxidation, introducing severe toxicity hazards that complicate worker safety and waste disposal. Alternative routes employing potassium permanganate, as seen in Chinese patents CN 1560027 and CN 1793115, suffer from poor selectivity; the strong oxidizing nature of permanganate often leads to the over-oxidation of the methyl group on the benzene ring, resulting in complex impurity profiles that are difficult and costly to purify. Furthermore, methods involving organocopper reagents or Friedel-Crafts acylation often require harsh reaction conditions, expensive catalysts, and generate substantial amounts of salt waste, all of which contribute to inflated production costs and extended lead times for high-purity agrochemical intermediates.
The Novel Approach
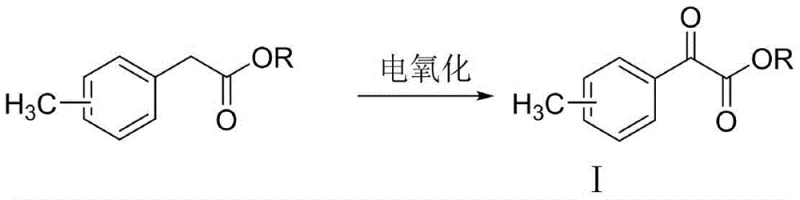
In stark contrast, the electro-oxidation method disclosed in CN113308705B offers a streamlined, catalytic alternative that fundamentally changes the reaction landscape. By employing an undivided electrolytic cell equipped with a carbon felt anode and a platinum mesh cathode, the process utilizes electrons as the primary oxidant, effectively rendering the reaction 'reagent-less' in terms of stoichiometric oxidizing agents. The system operates under mild conditions, typically between 15°C and 65°C, using acetonitrile and water as the solvent system with NHPI acting as a redox mediator. This approach not only eliminates the generation of heavy metal waste but also allows for precise control over the reaction trajectory through the adjustment of current density. As shown in the core reaction scheme below, the transformation of methyl 2-methylphenylacetate to the corresponding glyoxylate is achieved with high efficiency and selectivity, marking a substantial improvement over legacy chemical oxidation techniques.
Mechanistic Insights into NHPI-Mediated Electro-Oxidation
The core of this technological breakthrough lies in the synergistic interaction between the electrochemical cell and the NHPI catalyst. During the electrolysis process, the N-hydroxyphthalimide is anodically oxidized to generate the phthalimide-N-oxyl (PINO) radical, which acts as a potent hydrogen abstractor. This radical selectively abstracts a hydrogen atom from the benzylic position of the methyl phenylacetate substrate, generating a carbon-centered radical that is subsequently further oxidized at the anode surface to form the desired alpha-keto ester. This radical-mediated pathway is significantly milder than direct chemical oxidation, preserving the integrity of the aromatic ring and preventing the formation of ring-oxidized byproducts that plague permanganate-based methods. For R&D teams, understanding this mechanism is crucial for troubleshooting and optimizing the process, as the concentration of the NHPI mediator and the electrolyte composition directly influence the rate of PINO radical generation and overall reaction kinetics.
Furthermore, the patent highlights the critical role of current density in controlling product selectivity and conversion rates. By fine-tuning the current within the range of 5mA/cm² to 20mA/cm², operators can balance the rate of substrate oxidation against potential side reactions, ensuring a consistent impurity profile batch after batch. This level of control is particularly valuable when scaling up from laboratory to commercial production, where heat and mass transfer limitations can often degrade product quality in traditional exothermic oxidation reactions. The downstream application of this intermediate is seamless, as demonstrated by the subsequent conversion into Kresoxim-methyl through standard oximation, bromination, and etherification steps, confirming that the electro-chemically produced intermediate meets the rigorous purity specifications required for final API synthesis.
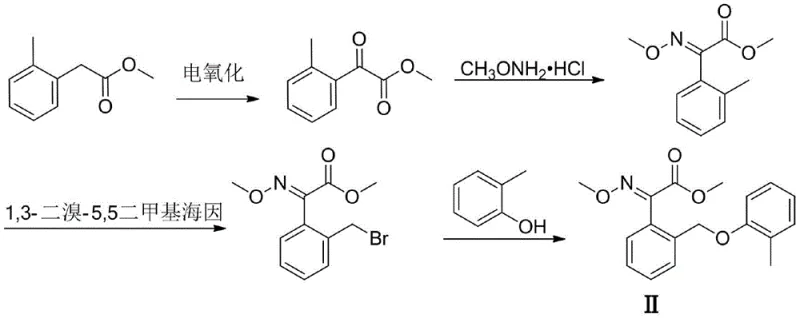
How to Synthesize Methyl 2-Methylphenylglyoxylate Efficiently
The practical implementation of this electro-oxidation protocol is designed for ease of adoption within existing fine chemical manufacturing facilities. The process begins with the preparation of the electrolyte solution, where the substrate, NHPI catalyst, and supporting electrolyte are dissolved in a mixture of acetonitrile and water. The reaction is conducted in a simple undivided cell, removing the need for complex membrane separators that often increase capital expenditure and maintenance requirements. Following the electrolysis period, which typically ranges from 4 to 10 hours depending on the scale and desired conversion, the workup involves standard organic extraction and purification techniques familiar to any process chemist. Detailed standardized synthesis steps see the guide below.
- Setup the electrolytic cell with a carbon felt anode and platinum mesh cathode, adding methyl 2-methylphenylacetate, NHPI catalyst, and electrolyte in acetonitrile/water.
- Apply a constant current density between 5mA/cm² and 20mA/cm² at a controlled temperature of 15°C to 65°C for 4 to 10 hours.
- Perform workup by rotary evaporation, extraction with ethyl acetate, washing, and drying to isolate the high-purity methyl 2-methylphenylglyoxylate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electro-oxidative methodology offers compelling economic and logistical benefits that extend beyond simple yield improvements. By shifting the oxidation burden from chemical reagents to electrical energy, manufacturers can decouple their production costs from the volatile pricing of specialized oxidants like chromium or manganese salts. This shift also drastically simplifies the waste management infrastructure required on-site, as the effluent streams are free from heavy metals and toxic cyanide residues, leading to significant reductions in environmental compliance costs and disposal fees. Moreover, the use of readily available starting materials such as methyl phenylacetate ensures a stable and resilient supply chain, reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chemical oxidants removes a major variable cost component from the production budget. Additionally, the simplified workup procedure reduces solvent consumption and labor hours associated with complex purification steps. The ability to operate at ambient or near-ambient temperatures further lowers energy consumption related to heating or cryogenic cooling, contributing to a leaner overall manufacturing cost structure without compromising on output quality.
- Enhanced Supply Chain Reliability: Relying on electricity as a reagent provides a level of supply security that chemical reagents cannot match, as power grids are generally more stable than the supply chains for niche fine chemicals. The robustness of the carbon felt and platinum electrodes ensures long equipment lifecycles with minimal replacement frequency. This reliability translates directly into more predictable delivery schedules for downstream customers, allowing supply chain planners to maintain lower safety stock levels and improve overall inventory turnover rates.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable through the addition of electrode plates or the use of flow chemistry configurations, facilitating a smooth transition from pilot scale to multi-ton commercial production. The 'green' nature of the process, characterized by the absence of toxic heavy metals and hazardous oxidants, aligns perfectly with increasingly strict global environmental regulations. This compliance advantage future-proofs the manufacturing asset, reducing the risk of regulatory shutdowns and enhancing the brand reputation of the supplier as a sustainable partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electro-oxidation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does the electro-oxidation method improve safety compared to traditional permanganate oxidation?
A: Traditional methods often utilize hazardous oxidants like potassium permanganate or toxic cyanides, which pose significant storage and disposal risks. The electro-oxidation method uses electrons as the primary reagent, eliminating the need for storing large quantities of dangerous chemical oxidants and reducing heavy metal waste.
Q: Can the selectivity of the reaction be controlled during the electro-oxidation process?
A: Yes, one of the key advantages highlighted in the patent is the ability to control conversion and selectivity by adjusting the constant current density. This tunability allows manufacturers to optimize the yield of the desired glyoxylate intermediate while minimizing over-oxidation byproducts.
Q: Is this electro-oxidation process suitable for large-scale industrial production?
A: The patent explicitly states that the method simplifies the technological process and is safe and environmentally friendly, making it suitable for large-scale promotion. The use of undivided cells and common electrode materials like carbon felt supports scalable implementation in standard industrial electrolyzers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-Methylphenylglyoxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electro-organic synthesis in the production of high-value agrochemical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113308705B can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of methyl 2-methylphenylglyoxylate meets the exacting standards required for strobilurin fungicide synthesis.
We invite forward-thinking partners to collaborate with us to leverage this green technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the highest levels of quality, sustainability, and economic efficiency available in the market.
