Revolutionizing Vitamin A Intermediate Production via Novel Rh-Catalyzed Hydroformylation
Revolutionizing Vitamin A Intermediate Production via Novel Rh-Catalyzed Hydroformylation
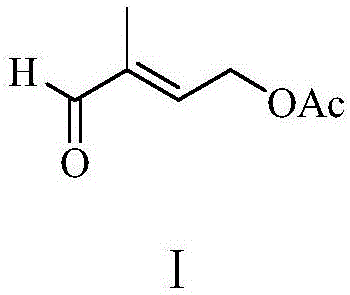
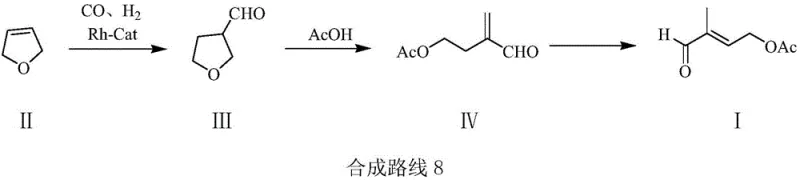
The global demand for high-purity Vitamin A acetate and carotenoids continues to drive innovation in the synthesis of their critical precursors. Patent CN110734374A introduces a groundbreaking preparation method for 2-methyl-4-acetoxyl-2-butenal, a pivotal intermediate in this value chain. This technology leverages a concise three-step sequence starting from 2,5-dihydrofuran and synthesis gas, utilizing advanced rhodium-catalyzed hydroformylation followed by acid-catalyzed ring opening and selective isomerization. Unlike traditional multi-step processes that suffer from low atom economy and hazardous reagents, this novel approach achieves a total yield of approximately 90 percent while significantly reducing environmental impact. For R&D directors and procurement strategists, this represents a paradigm shift towards greener, more cost-efficient manufacturing of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-4-acetoxyl-2-butenal has relied on cumbersome pathways such as the 1,1-dimethoxyacetone method or the ethylene oxide process, both of which present severe industrial bottlenecks. The dimethoxyacetone route, for instance, necessitates the use of expensive raw materials that are difficult to source consistently, alongside Grignard reagents that generate substantial volumes of hazardous wastewater. Furthermore, the ethylene oxide pathway involves the oxidation of 2-acetoxyethanol using silver catalysts, a reaction notorious for poor selectivity and the formation of unstable intermediates that complicate storage and handling. These legacy methods often require extreme reaction conditions, such as pressures exceeding 130 atm in the 1,4-butanediol route, demanding specialized high-pressure equipment that escalates capital expenditure and operational risk.
The Novel Approach
In stark contrast, the methodology disclosed in CN110734374A streamlines production into a robust three-step protocol that maximizes atom economy and minimizes waste. By initiating the synthesis with the hydroformylation of 2,5-dihydrofuran, the process bypasses the need for complex protecting group strategies and unstable aldehyde intermediates common in older routes. The subsequent acetylation and isomerization steps are conducted under moderate temperatures and pressures, utilizing readily available acetic acid and standard heterogeneous catalysts. This strategic simplification not only enhances the overall yield to nearly 90 percent but also drastically reduces the E-factor of the process, aligning perfectly with modern green chemistry principles and regulatory requirements for sustainable pharmaceutical manufacturing.
Mechanistic Insights into Rh-Catalyzed Hydroformylation and Selective Isomerization
The cornerstone of this innovative synthesis is the initial hydroformylation step, where 2,5-dihydrofuran reacts with synthesis gas (CO and H2) in the presence of a rhodium-based catalyst system. The absence of carbonyl functional groups in the starting 2,5-dihydrofuran is a critical design feature, as it prevents the competitive coordination of the substrate with the rhodium center, thereby facilitating high regioselectivity towards the desired 3-formyltetrahydrofuran intermediate. The catalyst system, typically comprising tris(triphenylphosphine)rhodium carbonyl hydride alongside a phosphine cocatalyst, operates efficiently at pressures between 2.0 and 10.0 MPa, which is significantly lower than the extreme conditions required by competing technologies. This mechanistic efficiency ensures that the reaction proceeds with high conversion rates while maintaining the integrity of the furan ring for the subsequent transformation.
Following the ring-opening acetylation, the final transformation involves a delicate double bond isomerization catalyzed by palladium on carbon. A key mechanistic advantage of this patent is the inclusion of a specific poisoning agent, such as tetramethyl thiourea, which modulates the activity of the palladium catalyst. Without this additive, there is a significant risk of over-hydrogenation, where the carbon-carbon double bond would be fully saturated to form a single bond, destroying the conjugated system essential for the molecule's reactivity. The poisoning agent selectively inhibits the hydrogenation activity while permitting the isomerization of the double bond to the thermodynamically stable 2,3-position. This precise control over catalyst selectivity is what enables the process to achieve gas phase purities exceeding 98 percent, minimizing the burden on downstream purification units.
How to Synthesize 2-Methyl-4-Acetoxyl-2-Butenal Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing fine chemical manufacturing facilities, requiring only standard stainless steel autoclaves and distillation setups. The process begins with the hydroformylation of 2,5-dihydrofuran in a solvent like toluene, followed by a straightforward workup to isolate the formyl-tetrahydrofuran intermediate. This intermediate is then subjected to acid-catalyzed reaction with acetic acid to effect ring opening, and finally, the crude product undergoes isomerization. The detailed standardized operating procedures, including specific molar ratios, temperature ramps, and safety protocols for handling synthesis gas, are critical for replicating the high yields reported in the patent examples.
- Perform hydroformylation of 2,5-dihydrofuran with synthesis gas (CO/H2) using a Rhodium catalyst to form 3-formyltetrahydrofuran.
- React the intermediate with acetic acid under acidic catalysis to effect ring-opening acetylation, yielding 2-formyl-4-acetoxy-1-butene.
- Execute double bond isomerization using a Palladium catalyst with a specific poisoning agent to obtain the final 2-methyl-4-acetoxyl-2-butenal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical superiority. The shift away from exotic raw materials like 1,1-dimethoxyacetone to commodity chemicals like 2,5-dihydrofuran and synthesis gas fundamentally alters the cost structure of the supply chain. By eliminating the need for Grignard reagents and high-pressure specialized reactors, the process reduces both the direct material costs and the capital intensity required for production. Furthermore, the significant reduction in wastewater generation and the avoidance of chlorinated byproducts simplify environmental compliance, lowering the overhead costs associated with waste treatment and regulatory reporting.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of low-cost, bulk-available feedstocks and the elimination of expensive stoichiometric reagents. By replacing complex multi-step sequences with a direct hydroformylation pathway, the number of unit operations is minimized, leading to reduced labor costs and energy consumption per kilogram of product. Additionally, the high selectivity of the rhodium and palladium catalyst systems means that raw material waste is negligible, maximizing the yield of valuable intermediates and directly improving the gross margin profile for manufacturers.
- Enhanced Supply Chain Reliability: Relying on stable, non-hazardous intermediates like 3-formyltetrahydrofuran mitigates the risks associated with the storage and transport of unstable aldehydes found in older ethylene oxide routes. The robustness of the catalyst system ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed reactions or off-spec material. This reliability is crucial for maintaining uninterrupted supply lines to downstream Vitamin A producers, ensuring that contractual obligations are met without the volatility often seen in complex fine chemical synthesis.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed to operate under moderate pressures that do not require bespoke high-pressure infrastructure. This facilitates rapid scale-up from pilot plant to commercial tonnage production without significant engineering hurdles. Moreover, the green nature of the synthesis, characterized by low waste generation and the absence of heavy metal contaminants in the final product, aligns with increasingly stringent global environmental regulations, future-proofing the supply chain against potential regulatory crackdowns on polluting manufacturing methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this synthesis route. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios.
Q: What are the primary advantages of the hydroformylation route over the dimethoxyacetone method?
A: The hydroformylation route utilizes cheaper, more stable raw materials like 2,5-dihydrofuran compared to expensive 1,1-dimethoxyacetone. It avoids the use of Grignard reagents, significantly reducing wastewater generation and operational complexity while achieving higher overall yields.
Q: How does the new process ensure selectivity during the isomerization step?
A: The process employs a specific poisoning agent, such as tetramethyl thiourea, alongside the Palladium catalyst. This additive prevents the over-hydrogenation of the carbon-carbon double bond, ensuring high selectivity for the desired conjugated aldehyde structure without forming saturated byproducts.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial scalability. It operates at moderate pressures (2.0-10.0 MPa) compared to older high-pressure methods, uses stable intermediates, and generates minimal wastewater, making it environmentally compliant and economically viable for ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-4-Acetoxyl-2-Butenal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient production of vitamins and nutraceuticals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of 2-methyl-4-acetoxyl-2-butenal. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch adheres to the highest industry standards for pharmaceutical intermediates.
We invite forward-thinking organizations to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your transition to this superior manufacturing method is seamless and economically advantageous.
