Revolutionizing Avibactam Intermediate Production: A Scalable Green Chemistry Approach
The pharmaceutical industry is constantly seeking robust, scalable, and economically viable pathways for synthesizing complex beta-lactamase inhibitors, particularly for next-generation antibiotics like Avibactam. Patent CN109400521B introduces a groundbreaking improvement in the preparation of 5R-benzyloxyaminopiperidine-2S-formate and its oxalate salt, which serve as critical precursors in this therapeutic class. This technology shifts the paradigm from expensive, low-yielding legacy methods to a streamlined process originating from readily available L-glutamic acid. By leveraging high-selectivity intramolecular condensation and efficient chiral resolution, this method addresses the urgent need for reliable pharmaceutical intermediates supplier capabilities that can support global demand without compromising on purity or environmental standards. The structural relationship between the intermediate and the final active pharmaceutical ingredient is clearly defined, highlighting the stereochemical precision required for biological activity.
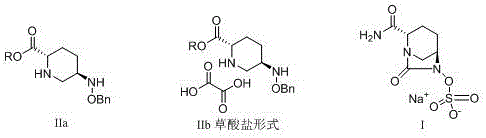
The limitations of conventional methods have long plagued the supply chain for Avibactam intermediates, primarily due to reliance on prohibitively expensive starting materials and hazardous reagents. Traditional routes, such as those described in earlier patents, typically commence with N-protected L-pyroglutamic acid esters, requiring carbon chain elongation via trimethyl sulfoxide iodide, a reagent known for its instability and high cost. Furthermore, alternative pathways often necessitate the use of precious metal catalysts, such as iridium complexes, to achieve the necessary stereoselectivity, which introduces significant purification challenges and potential heavy metal contamination risks. These legacy processes are characterized by low total yields, often hovering around 15%, and involve multiple protection and deprotection steps that amplify solvent consumption and waste generation. The operational complexity of these methods makes them ill-suited for large-scale industrial production, creating bottlenecks that threaten commercial scale-up of complex heterocycles and drive up the cost of goods sold for the final antibiotic.
In stark contrast, the novel approach detailed in this patent utilizes a completely different retrosynthetic logic that prioritizes atom economy and operational simplicity. By starting with L-glutamic acid or its sodium salt, the process bypasses the need for expensive protecting groups and exotic chain-extending reagents entirely. The core innovation lies in a sequential one-pot style transformation involving intramolecular condensation, hydrolysis decarboxylation, and esterification, which constructs the piperidine ring with high fidelity. This method eliminates the use of transition metal catalysts, thereby removing the need for costly and time-consuming metal scavenging steps downstream. The reaction selectivity is exceptionally high, with specific steps achieving near 100% selectivity due to the controlled pH and temperature conditions, ensuring that the desired isomer is formed preferentially without significant byproduct formation. This streamlined workflow not only enhances safety but also drastically simplifies the purification protocol, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Intramolecular Condensation and Chiral Resolution
The mechanistic elegance of this synthesis is best observed in the cyclization step, where the N-alkoxycarbonylmethyl-L-glutamic acid diester undergoes base-mediated intramolecular condensation. Under the influence of strong alkali bases like sodium methoxide or sodium hydride at controlled low temperatures, the ester enolate attacks the distal carbonyl group to form the six-membered piperidinone ring. Crucially, the subsequent hydrolysis and decarboxylation steps are managed in situ, preventing the isolation of unstable intermediates and driving the equilibrium toward the desired ketone product. The use of thionyl chloride or triphosgene in the esterification steps generates gaseous byproducts such as sulfur dioxide and hydrogen chloride, which spontaneously evolve from the reaction mixture, simplifying workup and improving the overall mass balance. This gas-evolving mechanism is a key feature that distinguishes this process from traditional liquid-liquid extraction heavy workflows, reducing the environmental footprint significantly.
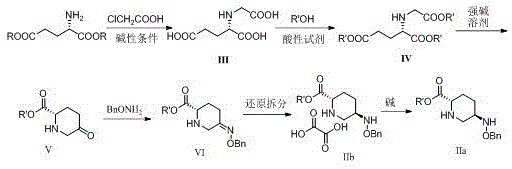
Impurity control is rigorously maintained through the strategic application of chiral resolution using oxalic acid in the final stages. Following the selective reduction of the benzyloxyimino intermediate, the resulting amine mixture is treated with oxalic acid to precipitate the target 5R-isomer as a crystalline oxalate salt. This crystallization-induced diastereomeric enrichment is highly effective, leveraging the solubility differences between the salt forms to achieve optical purities exceeding 99.5% as measured by chiral HPLC. The process parameters, including solvent ratios and cooling rates, are optimized to maximize the recovery of the correct enantiomer while leaving the unwanted isomer in the mother liquor. This level of stereochemical control is vital for ensuring the efficacy of the downstream Avibactam synthesis, as the biological activity is strictly dependent on the (1R, 2S, 5R) configuration. The ability to achieve such high purity without chromatographic separation underscores the robustness of this manufacturing route.
How to Synthesize 5R-Benzyloxyaminopiperidine-2S-Formate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-efficiency process in a pilot or production setting. The procedure begins with the alkylation of L-glutamic acid, followed by a telescoped esterification and cyclization sequence that minimizes intermediate handling. Operators must pay close attention to temperature control during the base addition phase to prevent exothermic runaways, ensuring the reaction remains within the optimal -20 to 30°C window. Detailed standard operating procedures regarding reagent addition rates and quenching protocols are essential for maintaining safety and consistency. For a comprehensive breakdown of the specific molar ratios, solvent choices, and reaction times required to replicate these results, please refer to the standardized guide below.
- Perform substitution of L-glutamic acid with chloroacetic acid under alkaline conditions to form N-carboxymethyl-L-glutamic acid.
- Execute esterification using thionyl chloride or triphosgene, followed by intramolecular condensation and hydrolysis to generate the piperidinone core.
- Condense with benzyloxyamine, followed by selective reduction and chiral resolution with oxalic acid to obtain the final target isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this new synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. The shift from scarce, high-cost starting materials to commodity chemicals like L-glutamic acid fundamentally alters the cost structure of the intermediate, providing a buffer against raw material price volatility. This stability is crucial for long-term contract negotiations and ensures a predictable cost of goods for the final antibiotic formulation. Furthermore, the elimination of heavy metal catalysts and hazardous reagents reduces the regulatory burden associated with impurity testing and validation, accelerating the timeline for technology transfer and regulatory filing. The simplified workflow also translates to shorter batch cycle times, allowing manufacturing facilities to increase throughput without requiring additional capital investment in new reactor trains.
- Cost Reduction in Manufacturing: The replacement of expensive N-protected pyroglutamic acid and iridium catalysts with inexpensive L-glutamic acid and common bases results in substantial raw material savings. The process avoids the use of trimethyl sulfoxide iodide, a costly and difficult-to-handle reagent, further driving down operational expenses. Additionally, the high atom economy and the removal of gaseous byproducts reduce the volume of waste solvents requiring disposal, leading to significant savings in waste treatment costs. The overall yield improvement from approximately 15% in prior art to over 60% in this new method means that less raw material is needed to produce the same amount of product, effectively halving the material cost per kilogram.
- Enhanced Supply Chain Reliability: Sourcing L-glutamic acid is far more secure than relying on specialized protected amino acid derivatives, which often have limited suppliers and long lead times. This abundance ensures that production schedules are not disrupted by raw material shortages, a critical factor for maintaining continuous supply of life-saving antibiotics. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry better than sensitive catalytic hydrogenations, reduces the risk of batch failures. Consequently, manufacturers can offer more reliable delivery commitments to their downstream API partners, strengthening the overall resilience of the pharmaceutical supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like esterification and crystallization that are well-understood in chemical engineering. The absence of high-pressure hydrogenation steps or cryogenic reactions simplifies the equipment requirements, allowing for production in standard glass-lined or stainless steel reactors. From an environmental perspective, the reduction in solvent usage and the elimination of heavy metal waste align with green chemistry principles, facilitating easier permitting and compliance with increasingly strict environmental regulations. This sustainability profile enhances the corporate social responsibility standing of the manufacturing partner, a key consideration for major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios. We encourage stakeholders to review these points carefully to appreciate the full scope of the technical advantages offered.
Q: What are the primary cost advantages of this new synthesis route compared to prior art?
A: The new method utilizes L-glutamic acid, a cheap and abundant starting material, replacing expensive N-protected pyroglutamic acid esters and eliminating the need for costly iridium catalysts or trimethyl sulfoxide iodide, significantly lowering raw material costs.
Q: How does this process improve environmental compliance and waste management?
A: The process generates gaseous byproducts like sulfur dioxide and carbon dioxide during esterification which are easily removed, reducing liquid waste discharge. Additionally, the high atom economy and elimination of heavy metal catalysts simplify three-waste treatment.
Q: What is the expected optical purity and yield of the final intermediate?
A: The patent data indicates a total yield exceeding 60% (up to 75%) based on L-monosodium glutamate, with chiral HPLC purity reaching 99.5% or higher after oxalic acid resolution, ensuring suitability for GMP API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5R-Benzyloxyaminopiperidine-2S-Formate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against bacterial resistance. Our team of expert chemists has thoroughly analyzed this patented route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this key Avibactam intermediate with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the highest international standards. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of this synthesis, guaranteeing a consistent and secure supply for your API development needs.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain and reduce your overall manufacturing costs. Let us be your partner in bringing safer, more affordable antibiotics to the market.
