Breakthrough Synthesis of Thiophene Macrocycles for Commercial Scale-up and High-Purity Electronic Materials
Breakthrough Synthesis of Thiophene Macrocycles for Commercial Scale-up and High-Purity Electronic Materials
The landscape of organic electronic materials is constantly evolving, driven by the demand for higher charge mobility and superior conjugated properties in semiconductor applications. A pivotal advancement in this field is documented in patent CN101343280B, which details a novel preparation method for thiophene macrocyclic compounds, specifically focusing on eight-membered and twelve-membered ring structures. This technology addresses long-standing synthetic challenges by introducing a strategic trimethylsilyl (TMS) protection group methodology that drastically improves reaction yields and operational feasibility. For industry leaders seeking a reliable electronic chemical supplier, understanding this patented route is crucial as it transforms previously impractical laboratory curiosities into viable commercial commodities. The innovation lies not just in the final product but in the robust three-step sequence that ensures consistency and scalability, marking a significant leap forward for manufacturers of organic photoelectric transition materials and organic film FETs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiophene macrocyclic compounds has been plagued by notoriously low yields and complex purification hurdles, severely restricting their widespread adoption in high-performance electronics. As illustrated in prior art such as the Kauffmann method from 1978, the direct oxidative cyclization of 3,3'-dibromo-[2,2']-bithiophenes resulted in dismal yields, particularly for the twelve-membered ring variant which was obtained at merely 4%. 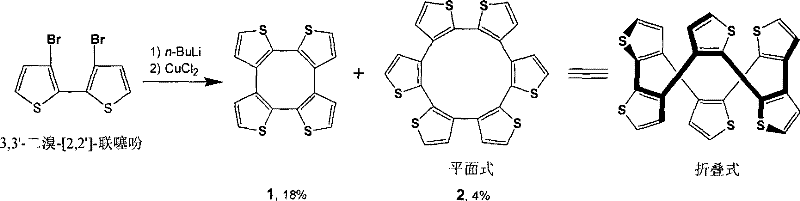 This inefficiency stems from uncontrolled side reactions at the active alpha-positions of the thiophene rings, leading to polymerization or the formation of undesired oligomers rather than the target macrocycle. Furthermore, the poor solubility of the intermediate organolithium species often necessitates harsh conditions that degrade product quality, creating a bottleneck for any procurement manager looking for cost reduction in electronic chemical manufacturing. These traditional pathways simply cannot support the volume or purity standards required by modern supply chains, rendering them obsolete for large-scale commercial endeavors.
This inefficiency stems from uncontrolled side reactions at the active alpha-positions of the thiophene rings, leading to polymerization or the formation of undesired oligomers rather than the target macrocycle. Furthermore, the poor solubility of the intermediate organolithium species often necessitates harsh conditions that degrade product quality, creating a bottleneck for any procurement manager looking for cost reduction in electronic chemical manufacturing. These traditional pathways simply cannot support the volume or purity standards required by modern supply chains, rendering them obsolete for large-scale commercial endeavors.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data introduces a sophisticated protection-deprotection strategy that fundamentally alters the reaction trajectory to favor macrocyclization. By initially masking the reactive alpha-protons with trimethylsilyl groups using lithium diisopropylamide (LDA), the synthesis effectively blocks parasitic side reactions that typically consume starting materials. This modification not only stabilizes the reactive intermediates but also significantly enhances their solubility in organic solvents, facilitating smoother processing and easier handling during the critical oxidative ring-closing step. The result is a dramatic improvement in efficiency, where the yield of the elusive twelve-membered thiophene ring jumps to 36%, representing a nearly seven-fold increase over the best historical methods. This breakthrough provides a clear pathway for the commercial scale-up of complex polymer additives and semiconductor precursors, ensuring that supply chain heads can rely on consistent output without the volatility associated with older, unpredictable chemistries.
Mechanistic Insights into TMS-Protected Oxidative Cyclization
The core of this technological advancement lies in the precise manipulation of electronic and steric factors through the TMS protection mechanism. The process begins with the deprotonation of 2,2'-dibromo-[3,3']-bithiophene at the alpha-position using LDA at low temperatures, followed immediately by quenching with trimethylchlorosilane to install the protecting groups. This step is critical as it converts the highly reactive thiophene protons into inert silyl ethers, preventing the formation of poly-thiophene chains during the subsequent lithiation. Following this protection, a bromine-lithium exchange is performed using butyllithium, generating a stable dilithio-intermediate that is perfectly poised for intramolecular coupling. The addition of anhydrous cupric chloride then triggers the oxidative cyclization, where the copper species mediates the formation of carbon-carbon bonds between the lithiated positions, closing the ring to form the macrocyclic skeleton with high regioselectivity. 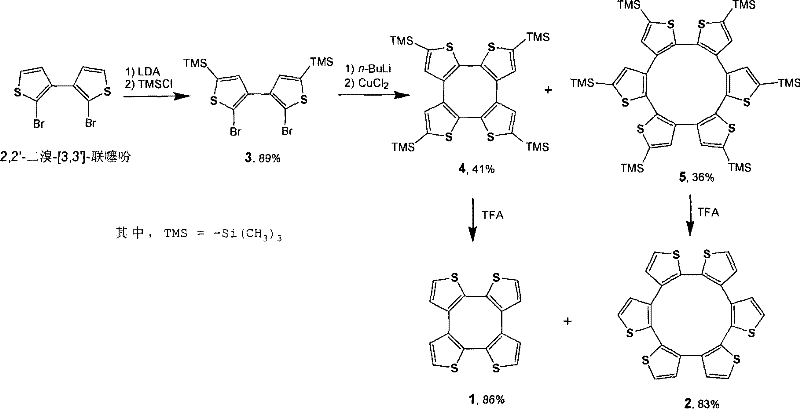 This mechanistic control ensures that the reaction proceeds through the desired twelve-membered transition state rather than collapsing into smaller, thermodynamically favored but less useful eight-membered rings or linear polymers.
This mechanistic control ensures that the reaction proceeds through the desired twelve-membered transition state rather than collapsing into smaller, thermodynamically favored but less useful eight-membered rings or linear polymers.
Beyond the cyclization event, the impurity control mechanism inherent in this design is equally vital for meeting the stringent purity specifications demanded by R&D directors in the semiconductor sector. The final step involves the removal of the TMS protecting groups using trifluoroacetic acid in chloroform, a mild yet effective deprotection condition that leaves the sensitive macrocyclic core intact. Because the intermediate macrocycles (compounds 4 and 5) are protected, they exhibit different polarity and solubility profiles compared to unprotected byproducts, allowing for highly efficient separation via silica gel column chromatography using standard chloroform and petroleum ether eluents. This ease of purification means that the final products, tetra(2,3-bithiophene) and hexa(2,3-bithiophene), can be isolated with exceptional purity levels, minimizing the presence of metal residues or oligomeric contaminants that could detrimentally affect charge mobility in finished electronic devices. The ability to consistently produce high-purity OLED material or semiconductor precursors through such a controlled mechanism is a decisive factor for quality assurance in advanced material manufacturing.
How to Synthesize Thiophene Twelve-Membered Cyclic Compound Efficiently
Implementing this synthesis route requires careful attention to anhydrous conditions and temperature control to maximize the benefits of the TMS protection strategy. The process is designed to be operationally robust, utilizing common reagents like n-Butyl Lithium and cupric chloride which are readily available in the global chemical market, thereby reducing lead time for high-purity electronic chemical procurement. The detailed standardized synthesis steps involve precise molar ratios, such as maintaining a 1:2 ratio between the dibromo-bithiophene derivative and butyllithium, to ensure complete conversion without excess reagent waste. For technical teams looking to replicate or license this technology, the patent provides a clear blueprint that balances reaction kinetics with practical workup procedures, ensuring that the theoretical yields observed in the lab can be translated to pilot and production scales.
- Protect the alpha-position protons of 2,2-dibromo-[3,3]-bithiophene using LDA and trimethylchlorosilane to form the bis-TMS intermediate.
- Perform bromine-lithium exchange with butyllithium followed by oxidative ring closure using anhydrous cupric chloride to generate the macrocyclic skeleton.
- Remove the trimethylsilyl protecting groups using trifluoroacetic acid in chloroform to isolate the final high-purity thiophene twelve-membered cyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly address the pain points of cost and reliability in the specialty chemical sector. The drastic improvement in yield translates directly into reduced raw material consumption per kilogram of finished product, which is a primary driver for lowering the overall cost of goods sold. By eliminating the need for extensive recycling of unreacted starting materials or complex separation of isomeric byproducts that plagued earlier methods, the process streamlines the manufacturing workflow significantly. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, a critical factor for procurement managers negotiating contracts for long-term supply agreements. Furthermore, the use of standard reagents and scalable reaction conditions reduces the dependency on exotic catalysts or proprietary equipment, enhancing the resilience of the supply chain against external disruptions.
- Cost Reduction in Manufacturing: The elimination of low-yield steps and the minimization of waste generation inherently lower the production costs associated with these high-value intermediates. By achieving a three-step total yield of 27% compared to the negligible yields of the past, the process reduces the effective cost per unit of active material significantly. This economic efficiency is further bolstered by the simplified purification process, which requires less solvent and stationary phase material, thereby reducing both material costs and environmental disposal fees. Consequently, this route enables a more sustainable and cost-effective production model that aligns with modern green chemistry initiatives while delivering financial value to the end buyer.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate standard industrial anhydrous and low-temperature setups, ensures that production schedules can be met with high predictability. Unlike fragile catalytic systems that might fail due to minor impurities in feedstocks, this stoichiometric approach is forgiving and consistent, guaranteeing a steady flow of materials to downstream customers. This reliability is paramount for supply chain heads who need to mitigate the risk of production stoppages in their own facilities due to delayed intermediate deliveries. The ability to source these complex macrocycles from a stable and proven manufacturing process adds a layer of security to the entire value chain of organic electronic device production.
- Scalability and Environmental Compliance: The synthesis is explicitly designed for large-scale preparation, avoiding the use of hazardous heavy metal catalysts that often require expensive and regulated removal steps. The oxidative cyclization uses copper chloride, a relatively benign and easily managed reagent, which simplifies waste treatment and compliance with environmental regulations. This ease of scale-up means that production volumes can be increased from 100 kgs to 100 MT annual commercial production without fundamental changes to the chemistry, ensuring that supply can grow in tandem with market demand. Additionally, the reduced solvent usage and higher atom economy contribute to a lower environmental footprint, satisfying the increasing corporate sustainability goals of major multinational purchasers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this thiophene macrocycle synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this material into your existing product lines or research pipelines.
Q: How does the TMS protection strategy improve the yield of thiophene twelve-membered rings?
A: The introduction of trimethylsilyl (TMS) groups at the alpha-positions prevents unwanted side reactions at these active sites during the lithiation process. This steric and electronic modulation significantly enhances the solubility of the organolithium intermediates and directs the oxidative cyclization towards the desired twelve-membered macrocycle, boosting yields from historical lows of 4% to 36%.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the reaction conditions involve anhydrous, oxygen-free, and low-temperature environments which are standard and manageable in industrial settings. The operability is high, and the significant increase in overall yield (up to 27% over three steps) makes the process economically viable for commercial scale-up compared to previous methods.
Q: What are the primary applications for these thiophene macrocyclic compounds?
A: These compounds exhibit high charge mobility and conjugated properties, making them ideal candidates for organic semiconductor materials, organic photoelectric transition materials, and organic film FETs. They also serve as host molecules in master-object chemistry for molecular and ionic identification applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiophene Macrocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the next generation of organic electronic materials. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory concept to market reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of thiophene macrocyclic compound meets the exacting standards required for high-performance semiconductor applications. We are committed to leveraging this advanced chemistry to provide our clients with a competitive edge through superior material quality and consistent availability.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic methodology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can drive innovation and efficiency in your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
