Advanced Synthesis of Styrene-Terminated Tetrathiophene Derivatives for High-Performance OFET Commercialization
Advanced Synthesis of Styrene-Terminated Tetrathiophene Derivatives for High-Performance OFET Commercialization
The landscape of organic electronics is rapidly evolving, driven by the demand for flexible, lightweight, and cost-effective semiconductor materials. A pivotal advancement in this domain is documented in Chinese Patent CN101348491B, which discloses a novel class of styrene-terminated tetrathiophene derivatives. These compounds represent a significant leap forward in the design of organic field-effect transistors (OFETs), offering a unique combination of extended pi-conjugation and robust thermal stability. By strategically capping the tetrathiophene core with styrene groups, the inventors have successfully enhanced electron delocalization across the molecular backbone. This structural modification is not merely theoretical; it translates directly into tangible device performance metrics, including high carrier mobility and exceptional switching ratios. For R&D directors and procurement specialists in the optoelectronic sector, understanding the synthesis and application of these materials is critical for next-generation display and sensor technologies.
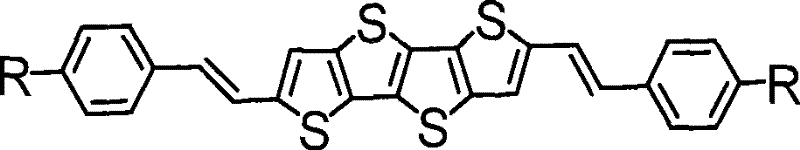
The core innovation lies in the specific molecular architecture shown in the structural formula, where the central fused thiophene system is flanked by vinyl-linked phenyl rings. This configuration effectively extends the conjugated system compared to traditional oligothiophenes, facilitating better charge transport properties. The patent highlights that these derivatives can serve as the active semiconductor layer in OFET devices, achieving mobilities that rival more complex and expensive alternatives. Furthermore, the versatility of the synthesis allows for various substituents at the R-position, enabling fine-tuning of solubility and energy levels without compromising the electronic performance. This adaptability makes the technology highly attractive for commercial scale-up of complex electronic chemicals, providing a reliable platform for developing customized organic semiconductor solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance oligothiophene-based semiconductors has been plagued by significant challenges regarding purity and processability. Traditional routes often involve harsh reaction conditions that can degrade the sensitive conjugated backbone, leading to defective materials with poor charge transport characteristics. Moreover, conventional methods frequently struggle to achieve the necessary regioregularity and end-group functionalization required for optimal device performance. The purification of these intermediates is another bottleneck, as standard recrystallization techniques often fail to remove trace metal catalysts or isomeric impurities that act as charge traps. Consequently, devices fabricated from such materials exhibit inconsistent mobility and low on-off ratios, limiting their viability in commercial applications. The reliance on multi-step syntheses with low overall yields further exacerbates cost issues, making widespread adoption in the competitive display market difficult.
The Novel Approach
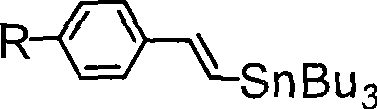
In contrast, the methodology described in CN101348491B offers a streamlined and efficient pathway to high-quality organic semiconductors. The novel approach utilizes a palladium-catalyzed cross-coupling reaction, specifically a Stille coupling, to attach the styrene termini to the tetrathiophene core. This reaction is conducted in toluene at moderate temperatures ranging from 80 to 100°C, which preserves the integrity of the conjugated system while ensuring high conversion rates. The use of (E)-beta-tributyltin styrene derivatives as coupling partners allows for precise control over the stereochemistry of the double bonds, ensuring the formation of the thermodynamically stable trans-isomer. This stereochemical purity is crucial for maximizing pi-orbital overlap and, consequently, charge mobility. The simplicity of the workup procedure, involving solvent removal and washing, significantly reduces processing time and waste generation compared to older methodologies.
Mechanistic Insights into Pd-Catalyzed Stille Cross-Coupling
The success of this synthesis hinges on the efficient operation of the palladium catalytic cycle, which facilitates the formation of carbon-carbon bonds between the aryl halide and the organotin reagent. The mechanism initiates with the oxidative addition of the dibromotetrathiophene to the zero-valent palladium species, forming a reactive organopalladium intermediate. This step is critical as it activates the relatively inert carbon-bromine bonds on the thiophene ring. Subsequently, transmetallation occurs, where the styryl group from the tributyltin reagent is transferred to the palladium center. The presence of the phosphine ligands on the catalyst stabilizes the intermediate complexes and prevents the aggregation of palladium black, which would otherwise deactivate the catalyst. Finally, reductive elimination releases the desired styrene-terminated product and regenerates the active palladium catalyst, allowing the cycle to continue. This catalytic efficiency is paramount for minimizing the loading of expensive precious metals, thereby contributing to cost reduction in electronic chemical manufacturing.
Beyond the reaction mechanism, the patent emphasizes a rigorous purification strategy that is essential for achieving electronic-grade purity. Trace impurities, particularly residual tin and palladium, can severely degrade the performance of OFET devices by introducing deep trap states within the bandgap. To address this, the process incorporates a multi-stage purification protocol. After the initial reaction, the crude solid is washed sequentially with methanol and acetone to remove soluble organic byproducts and excess reagents. However, the most critical step is the gradient sublimation performed in a specialized furnace. Sublimation is a phase transition technique that allows the pure compound to vaporize and re-condense, leaving behind non-volatile impurities and metal residues. This physical purification method is superior to chromatography for large-scale production as it avoids the use of large volumes of silica gel and solvents, aligning with green chemistry principles while ensuring the stringent purity specifications required for high-mobility semiconductor applications.
How to Synthesize Styrene-Terminated Tetrathiophene Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these advanced materials with high reproducibility. The process begins with the preparation of the key building blocks, specifically the dibromotetrathiophene core and the tributyltin-styrene derivatives. These precursors are combined in a toluene solvent system with a catalytic amount of bis(triphenylphosphine)palladium chloride. The reaction mixture is then heated to a temperature between 80 and 100 degrees Celsius and maintained for a period of 48 to 72 hours to ensure complete conversion. Following the reaction, the solvent is removed under reduced pressure, and the resulting solid undergoes the aforementioned washing and sublimation steps. This standardized approach minimizes variability and ensures that the final product meets the necessary quality standards for device fabrication. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve dibromotetrathiophene and (E)-beta-tributyltin p-R-styrene in toluene with a palladium catalyst.
- Heat the mixture to 80-100°C and maintain reaction for 48-72 hours under inert atmosphere.
- Purify the crude product through solvent washing and gradient sublimation to achieve electronic grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary advantage lies in the simplification of the manufacturing process, which directly translates to operational efficiency. By utilizing a one-pot coupling reaction that proceeds with high selectivity, the need for complex intermediate isolation and purification steps is significantly reduced. This streamlining of the workflow lowers the consumption of solvents and energy, resulting in substantial cost savings in organic semiconductor manufacturing. Furthermore, the use of commercially available starting materials, such as substituted phenylacetylenes and tributyltin hydride, ensures a stable and reliable supply chain. The ability to source raw materials from multiple vendors mitigates the risk of supply disruptions, enhancing the overall resilience of the production network.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in high loadings is a key driver for cost optimization. The patent describes the use of a palladium catalyst at relatively low molar ratios, which reduces the raw material cost per kilogram of product. Additionally, the high thermal stability of the final product allows for purification via sublimation, a method that is often more scalable and less labor-intensive than column chromatography. This efficiency in purification reduces the loss of product during processing, thereby improving the overall mass balance and yield. The combination of efficient catalysis and scalable purification creates a cost structure that is highly competitive for mass-market electronic applications.
- Enhanced Supply Chain Reliability: The synthetic route relies on robust chemical transformations that are well-understood in the fine chemical industry. The reagents involved, including the organotin coupling partners, are established commodities with mature supply chains. This maturity means that lead times for raw materials are predictable and generally short, facilitating just-in-time manufacturing strategies. Moreover, the reaction conditions are not overly sensitive to minor fluctuations in temperature or pressure, making the process robust against typical variations found in large-scale reactor environments. This robustness ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream device manufacturers who require strict material specifications.
- Scalability and Environmental Compliance: Scaling this chemistry from the laboratory to industrial production is facilitated by the use of common solvents like toluene and standard heating equipment. The process does not require exotic high-pressure or cryogenic conditions, which simplifies the engineering requirements for plant expansion. From an environmental perspective, the ability to recycle solvents and the reduction of hazardous waste through efficient purification align with increasingly stringent global environmental regulations. The high thermal stability of the product also implies a longer shelf life and reduced degradation during storage and transport, further minimizing waste. These factors collectively support a sustainable manufacturing model that appeals to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these styrene-terminated tetrathiophene derivatives. The answers are derived directly from the experimental data and claims presented in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating these materials into their existing product lines. The information covers aspects ranging from material stability to device performance metrics, providing a comprehensive overview for decision-makers.
Q: What is the thermal stability of styrene-terminated tetrathiophene derivatives?
A: According to patent CN101348491B, these derivatives exhibit excellent thermal stability with decomposition temperatures exceeding 350°C, making them suitable for vacuum deposition processes.
Q: What charge carrier mobility can be achieved with these organic semiconductors?
A: Devices fabricated using these materials have demonstrated hole mobilities up to 0.06 cm²/V·s and on-off ratios greater than 10^5, indicating strong potential for logic circuit applications.
Q: How is the purity of the final semiconductor material ensured?
A: The process utilizes a rigorous purification protocol involving sequential washing with methanol and acetone, followed by multiple cycles of gradient sublimation to remove metal residues and organic impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Styrene-Terminated Tetrathiophene Derivatives Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle the specific requirements of organic electronic materials, including inert atmosphere processing and gradient sublimation purification. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for OFET applications. Our commitment to quality assurance ensures that the materials you receive are ready for immediate integration into your device fabrication processes.
We invite you to collaborate with us to optimize your supply chain and reduce your time-to-market. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. By leveraging our expertise in process optimization, we can help you identify opportunities for further efficiency gains. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your strategic partner in bringing next-generation organic electronic devices to the global market.
