Advanced Grignard-Based Synthesis for High-Purity 3,5-Bis(Trifluoromethyl)Benzyl Alcohol Manufacturing
Introduction to Advanced Intermediate Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and high-purity synthetic routes for critical intermediates, particularly those containing fluorinated motifs which are ubiquitous in modern drug design. A recent technological breakthrough, documented in patent CN113214045A, introduces a highly efficient preparation method for 3,5-bis(trifluoromethyl)benzyl alcohol, a pivotal building block for synthesizing various derivatives including benzyl halides and mesylates used in NK1 receptor antagonists and other therapeutic agents. This innovation addresses long-standing challenges in the field by leveraging a refined Grignard-based strategy coupled with specific formylation reagents, ultimately delivering a product with exceptional purity exceeding 99.5% and a maximum single impurity profile of less than 0.1%. For R&D directors and procurement specialists alike, this development represents a significant leap forward in process reliability, offering a pathway that mitigates the risks associated with moisture sensitivity and hazardous reagents found in legacy methods. The ability to source such high-quality intermediates is crucial for maintaining the integrity of downstream API synthesis, ensuring that final drug products meet rigorous regulatory standards without the burden of complex purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-bis(trifluoromethyl)benzyl alcohol has been plagued by inefficiencies and safety concerns inherent to older chemical pathways. One prominent legacy method, described in WO2005/035472, utilizes a Grignard reaction with solid paraformaldehyde. While conceptually straightforward, this approach is fundamentally flawed due to the high and uncontrollable water content present in solid paraformaldehyde. Given that Grignard reagents are notoriously sensitive to moisture, the presence of water leads to the premature quenching of the organometallic species, resulting in a suboptimal crude yield of approximately 76% and a complex mixture of by-products that are difficult to separate. Furthermore, alternative routes such as the one disclosed in US6596906 rely on the hydrogenation reduction of nitriles using nickel-aluminum alloys. This method not only depends on starting materials that are not readily available in the market but also introduces severe safety hazards associated with pyrophoric nickel catalysts and the generation of toxic, nickel-contaminated wastewater, posing significant environmental compliance burdens. Another pathway, EP1500641, employs lithium aluminum hydride (LiAlH4) for the reduction of the corresponding benzoic acid; however, this reagent is expensive, hazardous to handle on a large scale, and typically affords lower yields around 74%, making it economically unviable for commercial production.
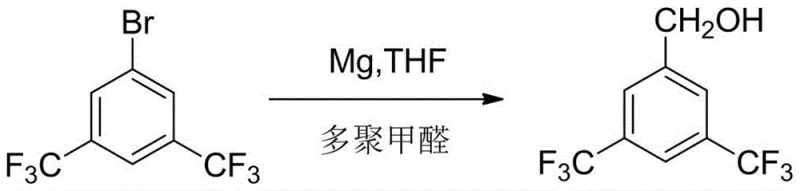
The Novel Approach
In stark contrast to these problematic legacy techniques, the novel process outlined in CN113214045A revolutionizes the synthesis by decoupling the carbon-carbon bond formation from the reduction step through a stable aldehyde intermediate. This innovative route begins with the formation of a Grignard reagent from 3,5-bis(trifluoromethyl)halobenzene and magnesium, but critically diverges by reacting this intermediate with specialized acylation reagents capable of introducing a formyl group, rather than unstable paraformaldehyde. By selecting optimal formylation agents such as N-methyl-N-phenylformamide or N,N-diethylformamide, the process achieves a highly selective conversion to 3,5-bis(trifluoromethyl)benzaldehyde with yields reaching up to 91%. This aldehyde intermediate is then subjected to a mild reduction using sodium borohydride, a reagent that is significantly safer, cheaper, and easier to handle than lithium aluminum hydride or nickel alloys. This strategic shift not only eliminates the moisture sensitivity issues of the paraformaldehyde route but also avoids the heavy metal contamination and safety risks of the nickel reduction pathway, resulting in a streamlined, two-step process that is inherently safer, more environmentally friendly, and capable of delivering the target alcohol with unprecedented purity levels suitable for direct use in sensitive pharmaceutical applications.
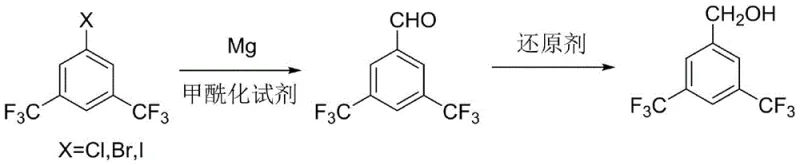
Mechanistic Insights into Grignard Formylation and Reduction
The core of this technical advancement lies in the precise control of the Grignard formation and the subsequent nucleophilic attack on the formylation reagent. The process initiates by reacting 3,5-bis(trifluoromethyl)halobenzene (where X can be Cl, Br, or I) with magnesium powder in a carefully selected ether solvent system, such as tetrahydrofuran (THF) or cyclopentyl methyl ether (CPME), at temperatures ranging from -10°C to 70°C. The choice of solvent is critical; experimental data indicates that while THF provides excellent results with yields around 91%, alternatives like CPME can push yields even higher to 93% while offering potential benefits in terms of solvent recovery and stability. Once the Grignard reagent is fully formed, indicated by the consumption of the halobenzene, the temperature is controlled between -10°C and 70°C for the addition of the formylation agent. Reagents like N-methyl-N-phenylformamide act as superior electrophiles compared to traditional sources, facilitating a clean nucleophilic attack by the Grignard carbon on the carbonyl carbon of the formamide. This step is meticulously managed to prevent over-addition or side reactions, ensuring that the resulting 3,5-bis(trifluoromethyl)benzaldehyde is generated with a GC purity of up to 99.4% before any further purification.
Following the isolation of the high-purity aldehyde, the second mechanistic phase involves a chemoselective reduction to the primary alcohol. Unlike the harsh conditions required for carboxylic acid reduction, the aldehyde functionality is readily reduced by metal borohydrides, specifically sodium borohydride (NaBH4), in alcohol solvents like methanol or ethanol at mild temperatures between -10°C and 45°C. This reduction proceeds via a hydride transfer mechanism where the borohydride anion delivers a hydride ion to the carbonyl carbon, forming an alkoxide intermediate which is subsequently protonated during the acidic workup. The beauty of this mechanism lies in its specificity; sodium borohydride reduces the aldehyde efficiently without affecting other potential sensitive functional groups that might be present in more complex analogues, although in this specific substrate, the trifluoromethyl groups remain inert. The final workup involves acidification with hydrochloric acid, solvent removal, and crystallization from water, a simple yet effective purification technique that leverages the solubility differences to exclude organic impurities, thereby locking in the final product purity of >99.5% with single impurities strictly controlled below 0.1%.
How to Synthesize 3,5-Bis(Trifluoromethyl)Benzyl Alcohol Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and solvent selection to maximize yield and safety. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to multi-ton industrial production without losing efficiency. Operators must ensure that the initial Grignard formation is complete before introducing the formylation reagent to prevent unreacted halide contamination. The subsequent reduction step is exothermic and requires controlled addition of the reducing agent to maintain thermal safety. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare the Grignard reagent by reacting 3,5-bis(trifluoromethyl)halobenzene with magnesium powder in a solvent like THF at -10°C to 70°C.
- React the Grignard reagent with a formylation agent (e.g., N-methyl-N-phenylformamide) to obtain 3,5-bis(trifluoromethyl)benzaldehyde.
- Reduce the resulting aldehyde using sodium borohydride in an alcohol solvent to yield the final high-purity alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By shifting away from expensive and hazardous reagents like lithium aluminum hydride and nickel-aluminum alloys, the new method drastically simplifies the procurement landscape, relying instead on commodity chemicals like magnesium, sodium borohydride, and readily available halobenzenes. This substitution not only lowers the raw material costs significantly but also reduces the logistical complexities associated with handling dangerous goods, thereby lowering insurance and transportation overheads. Furthermore, the elimination of heavy metal catalysts removes the need for costly and time-consuming metal scavenging steps, which are often required to meet strict residual metal limits in API production. This streamlining of the downstream processing workflow translates into shorter production cycles and reduced waste disposal costs, as the process generates benign by-products compared to the toxic sludge associated with nickel reductions.
- Cost Reduction in Manufacturing: The economic benefits of this route are substantial, driven primarily by the replacement of high-cost reducing agents and the improvement in overall process yield. By achieving yields exceeding 85% and often reaching above 90% for the intermediate aldehyde, the process minimizes raw material waste, which is particularly important given the value of fluorinated starting materials. Additionally, the use of sodium borohydride instead of lithium aluminum hydride represents a massive cost saving, as the former is a fraction of the price of the latter and does not require the same level of specialized containment infrastructure. The simplified purification protocol, which relies on crystallization rather than complex chromatography or distillation of sensitive intermediates, further reduces energy consumption and solvent usage, contributing to a leaner and more cost-effective manufacturing operation that enhances profit margins for the final API.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of widely available and stable raw materials. Unlike 3,5-bis(trifluoromethyl)benzonitrile, which can be difficult to source in bulk quantities with consistent quality, 3,5-bis(trifluoromethyl)bromobenzene is a commercially established intermediate with a robust global supply base. This availability ensures that production schedules are not disrupted by raw material shortages. Moreover, the mild reaction conditions and the stability of the intermediates allow for greater flexibility in production planning; the aldehyde intermediate can be isolated and stored if necessary, decoupling the two reaction steps and allowing for batch optimization. This flexibility is crucial for supply chain heads who need to manage inventory levels and respond quickly to fluctuating market demands without being constrained by the rigid timelines of continuous, hazardous processes.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, addressing the growing demand for green chemistry in the pharmaceutical sector. By avoiding the generation of nickel-containing wastewater and eliminating the use of pyrophoric catalysts, the facility's environmental footprint is significantly reduced, easing the burden on wastewater treatment plants and ensuring compliance with increasingly stringent environmental regulations. The solvents used, such as THF or CPME, are recoverable and recyclable, supporting a circular economy approach within the plant. The safety profile of the reaction, characterized by mild temperatures and the absence of high-pressure hydrogenation steps, reduces the risk of industrial accidents, ensuring business continuity and protecting the company's reputation. This alignment with ESG (Environmental, Social, and Governance) goals makes the supply of this intermediate more attractive to top-tier pharmaceutical companies that prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the process capabilities and limitations. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, ensuring that the information is grounded in verified scientific results. Understanding these nuances is essential for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: Why is the conventional paraformaldehyde route considered inferior for this synthesis?
A: Conventional routes using solid paraformaldehyde suffer from high water content which is difficult to control. Since Grignard reagents are extremely moisture-sensitive, this leads to lower crude yields (around 76%) and significant impurity formation, making purification difficult.
Q: What are the safety advantages of the new Grignard-Formylation route over Nickel-Aluminum reduction?
A: The Nickel-Aluminum alloy hydrogenation process involves significant safety hazards and generates nickel-containing wastewater which is environmentally unfriendly. The new method avoids heavy metal catalysts and uses milder conditions, enhancing operational safety and environmental compliance.
Q: What purity levels can be achieved with this new technical route?
A: The patented process consistently achieves a product purity of greater than 99.5%, with the maximum single impurity content controlled to less than 0.1%, meeting the stringent requirements for high-end pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Bis(Trifluoromethyl)Benzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just chemical knowledge; it demands engineering excellence and a commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel Grignard-based route are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle fluorinated chemistry safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3,5-bis(trifluoromethyl)benzyl alcohol meets the >99.5% purity benchmark required for high-end pharmaceutical synthesis. We understand the critical nature of this intermediate in your supply chain and are dedicated to providing a consistent, high-quality supply that supports your drug development timelines.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your COGS. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples, and let us help you secure a competitive advantage in the market with a reliable, high-purity supply of this critical fluorinated building block.
