Advanced Manufacturing of 1-Substituted-Hydantoins: Technical Breakthroughs and Commercial Scalability
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the relentless pace of innovation in both the pharmaceutical and agrochemical sectors. Patent CN1172804A introduces a transformative methodology for the preparation of 1-substituted-hydantoins, a critical structural motif found in numerous bioactive molecules. This technical disclosure outlines a robust synthetic pathway that leverages N-substituted-N-alkoxycarbonylamino-acetonitrile derivatives as key precursors, reacting them with alkali or alkaline earth metal hydroxides followed by acid treatment. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize supply chains for complex nitrogen-containing heterocycles. The process distinguishes itself through its operational simplicity, utilizing readily available inorganic bases such as sodium hydroxide and potassium hydroxide under mild thermal conditions ranging from -10 to 80°C. By circumventing the need for harsh cyclization reagents or expensive transition metal catalysts, this technology offers a compelling value proposition for manufacturers seeking to enhance process safety while maintaining stringent quality standards required for GMP production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing the hydantoin ring often suffer from significant drawbacks that impact both yield and environmental compliance. Conventional methods frequently rely on the reaction of amino acids with cyanates or the Bucherer-Bergs reaction, which can involve hazardous reagents like potassium cyanide and require extreme pH conditions that are difficult to control on a multi-ton scale. These legacy processes often generate substantial amounts of inorganic salt waste and necessitate complex purification steps, such as column chromatography, to remove trace impurities that could compromise the safety profile of the final active pharmaceutical ingredient. Furthermore, the thermal instability of certain intermediates in classical routes can lead to decomposition products, resulting in lower overall yields and increased production costs due to the loss of valuable starting materials. The reliance on stoichiometric amounts of toxic reagents also poses severe challenges for waste treatment facilities, forcing manufacturers to invest heavily in specialized effluent processing infrastructure to meet increasingly rigorous environmental regulations.
The Novel Approach
In stark contrast, the methodology described in CN1172804A utilizes a sophisticated N-alkoxycarbonyl protection strategy that fundamentally alters the reactivity profile of the acetonitrile precursor. By employing N-substituted-N-alkoxycarbonylamino-acetonitriles, the process enables a controlled hydrolysis and subsequent cyclization that proceeds smoothly under mild alkaline conditions. This approach effectively suppresses unwanted side reactions, such as polymerization or over-hydrolysis of the nitrile group, which are common pitfalls in less selective synthetic pathways. The use of common solvents like water, alcohols, and ethers further enhances the practicality of this method, allowing for flexible process design that can be adapted to existing reactor configurations without major capital expenditure. Additionally, the final acidification step not only triggers the ring closure but also facilitates the precipitation of the product, simplifying isolation and reducing the need for energy-intensive solvent removal steps. This streamlined workflow translates directly into reduced cycle times and lower operational expenditures for commercial manufacturing facilities.
Mechanistic Insights into Hydroxide-Mediated Cyclization
The core of this innovative synthesis lies in the precise manipulation of the electronic properties of the nitrile functionality through the adjacent carbamate protecting group. When the N-substituted-N-alkoxycarbonylamino-acetonitrile is exposed to a hydroxide source, the electron-withdrawing nature of the carbonyl group activates the methylene protons, facilitating nucleophilic attack on the nitrile carbon. This initiates a cascade of hydrolysis events that convert the nitrile group into an amide intermediate, which then undergoes intramolecular cyclization with the urea nitrogen to form the stable five-membered hydantoin ring. The choice of base is critical; alkali metal hydroxides like sodium hydroxide and potassium hydroxide provide the necessary nucleophilicity without introducing steric hindrance that could slow down the reaction kinetics. The reaction mechanism is further stabilized by the solvation effects of polar protic solvents such as water or tert-butanol, which help to dissolve the inorganic salts formed during the neutralization phase. This mechanistic clarity allows process chemists to fine-tune reaction parameters, such as molar ratios and addition rates, to maximize conversion efficiency and minimize the formation of open-chain urea byproducts.
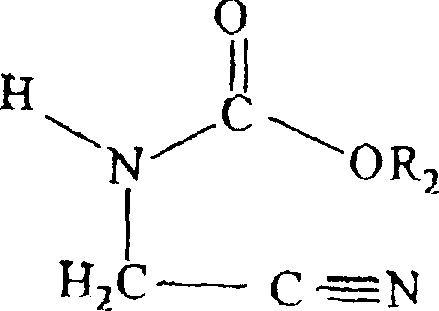
Impurity control is paramount in the synthesis of pharmaceutical intermediates, and this patent offers distinct advantages in managing the impurity profile throughout the reaction course. The use of specific alkoxycarbonyl groups, such as methoxycarbonyl or ethoxycarbonyl, ensures that the intermediate remains stable during the initial alkylation steps but becomes sufficiently labile under the cyclization conditions to allow for clean ring closure. Any unreacted starting material or hydrolyzed open-chain byproducts can be effectively removed through simple aqueous workups or recrystallization from solvents like acetonitrile or ethanol, as demonstrated in the patent examples. The absence of heavy metal catalysts eliminates the risk of metal contamination, a critical quality attribute for API intermediates that often requires costly scavenging steps in alternative catalytic methods. Furthermore, the mild reaction temperatures prevent thermal degradation of sensitive functional groups on the R1 substituent, preserving the structural integrity of diverse derivatives ranging from propargyl to benzyl variants. This high level of selectivity ensures that the final product meets the rigorous purity specifications demanded by regulatory bodies for downstream drug synthesis.
How to Synthesize 1-Substituted-Hydantoins Efficiently
To implement this synthesis effectively, manufacturers must adhere to a standardized protocol that balances reaction kinetics with safety considerations. The process begins with the preparation of the N-substituted-N-alkoxycarbonylamino-acetonitrile precursor, which can be synthesized via nucleophilic substitution of N-alkoxycarbonylamino-acetonitrile with appropriate alkyl halides or sulfonates in the presence of a base. Once the precursor is secured, the cyclization step involves dissolving the compound in a suitable solvent system, such as a mixture of water and dioxane, and adding the hydroxide solution dropwise to control exotherms. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety across different production scales.
- React N-substituted-N-alkoxycarbonylamino-acetonitrile with alkali or alkaline earth metal hydroxides in inert solvents at -10 to 80°C.
- Neutralize the resulting reaction mixture with mineral acids such as hydrochloric acid or sulfuric acid to induce cyclization.
- Isolate the final 1-substituted-hydantoin product through concentration, filtration, and recrystallization from aqueous or alcoholic solvents.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers substantial opportunities for cost optimization and supply chain resilience. The primary drivers of cost reduction stem from the utilization of commodity chemicals rather than specialized reagents, which stabilizes raw material pricing and reduces exposure to market volatility. The elimination of transition metal catalysts removes the need for expensive metal scavengers and the associated analytical testing for residual metals, significantly lowering the cost of goods sold. Moreover, the simplified workup procedure, which often relies on filtration and crystallization rather than distillation or chromatography, reduces utility consumption and waste disposal fees. These factors combine to create a more economically efficient manufacturing process that can withstand competitive pricing pressures in the global intermediates market.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by replacing expensive catalytic systems with inexpensive inorganic bases like sodium hydroxide and potassium hydroxide. By avoiding the use of precious metals or complex organocatalysts, manufacturers can drastically reduce the raw material bill of materials. Furthermore, the ability to perform the reaction in aqueous or semi-aqueous media minimizes the volume of organic solvents required, leading to lower solvent recovery costs and reduced fire hazard insurance premiums. The high selectivity of the reaction means that less material is lost to side products, improving the overall mass balance and yield per batch. This efficiency translates directly into a lower cost per kilogram of the final hydantoin intermediate, providing a competitive edge in tender negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the widespread availability of the key starting materials, such as chloroformates and aminoacetonitrile salts, which are produced by multiple global suppliers. Unlike proprietary reagents that may be subject to single-source bottlenecks, these commodity chemicals ensure that production schedules are not disrupted by raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, further enhances reliability by reducing the risk of batch failures due to minor variations in input quality. This flexibility allows supply chain managers to source materials from diverse geographic regions, mitigating geopolitical risks and logistics delays. Consequently, manufacturers can maintain consistent inventory levels and meet just-in-time delivery commitments to their pharmaceutical clients with greater confidence.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous reagents and the use of standard unit operations. The reaction exotherms are manageable, and the workup involves simple phase separations and filtrations that are easily automated in large-scale reactors. From an environmental standpoint, the process generates primarily inorganic salts and aqueous waste streams that are easier to treat than organic solvent-heavy effluents from traditional methods. This aligns with green chemistry principles and helps manufacturers meet increasingly strict environmental, social, and governance (ESG) targets. The reduced environmental footprint not only lowers compliance costs but also enhances the brand reputation of the manufacturer as a sustainable partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydantoin synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The insights provided here cover aspects ranging from reaction optimization to quality control measures.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization requires mild temperatures ranging from -10 to 80°C using alkali metal hydroxides like NaOH or KOH, ensuring high purity without degradation.
Q: How does this method improve impurity profiles compared to traditional routes?
A: By utilizing specific N-alkoxycarbonyl protection groups, the process minimizes side reactions and allows for simple recrystallization instead of complex chromatography.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common inorganic bases and aqueous workups eliminates the need for exotic catalysts, making it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Substituted-Hydantoins Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation for comprehensive impurity profiling. Our facility is designed to handle complex chemistries safely, adhering to international safety and quality standards that give our partners peace of mind.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and reduce your overall development costs. Let us be your trusted partner in bringing innovative therapies to market faster and more efficiently.
