Advanced Manufacturing of 3-Carbonyl-4-Aza-5Alpha-Androstane Intermediates for Global Pharma Supply Chains
The pharmaceutical industry's relentless pursuit of effective treatments for Benign Prostatic Hyperplasia (BPH) has placed 5-alpha reductase inhibitors at the forefront of urological therapeutics. Central to the production of these life-changing medications, such as Finasteride, is the efficient and cost-effective synthesis of the key intermediate, 3-carbonyl-4-aza-5alpha-androstane. A pivotal advancement in this domain is detailed in patent CN101456897B, which discloses a novel preparation method that fundamentally shifts the paradigm from traditional high-pressure catalytic hydrogenation to a safer, more economical transfer hydrogenation protocol. This technological breakthrough addresses critical bottlenecks in the supply chain by replacing scarce and expensive precious metal catalysts with abundant formic acid derivatives, thereby unlocking new potential for cost reduction in API manufacturing. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at scale.
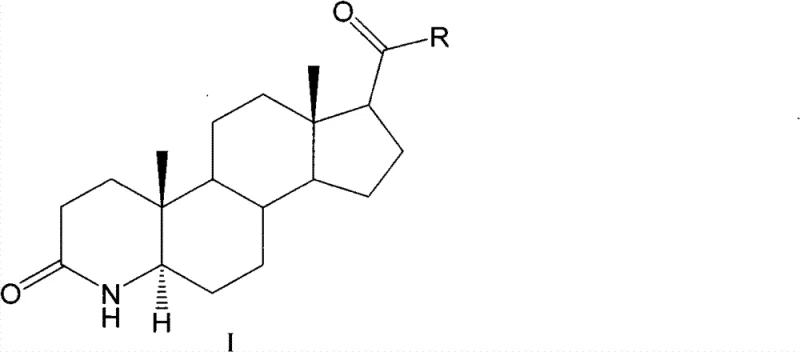
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-carbonyl-4-aza-5alpha-androstane derivatives has relied heavily on direct catalytic hydrogenation using noble metals. As illustrated in prior art such as U.S. Patent 4220775, the conventional route typically employs Platinum (Pt) catalysts in glacial acetic acid under normal pressure, or alternatively, 10% Pd/C under high hydrogen pressures exceeding 5 MPa. These legacy methods present severe logistical and economic challenges for modern chemical manufacturing. The reliance on precious metals like Platinum and Palladium introduces significant volatility in production costs, as the prices of these commodities are subject to global market fluctuations. Furthermore, the post-reaction processing is notoriously cumbersome; removing trace amounts of toxic heavy metal catalysts from the final product requires extensive purification steps to meet strict regulatory limits for pharmaceutical ingredients. Additionally, the use of high-pressure hydrogen gas necessitates specialized, expensive reactor infrastructure and rigorous safety protocols, creating a high barrier to entry for scalable production and increasing the overall lead time for high-purity pharmaceutical intermediates.
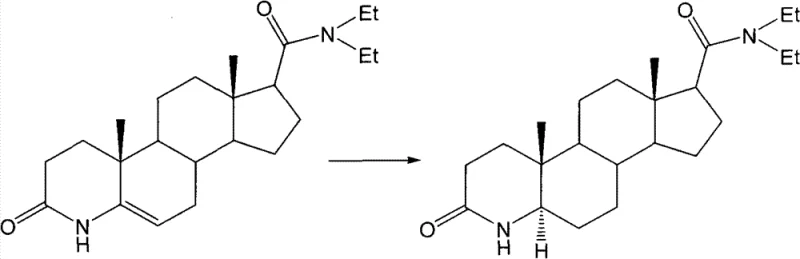
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN101456897B introduces a robust transfer hydrogenation strategy that elegantly circumvents the drawbacks of direct hydrogenation. This innovative approach utilizes formic acid in conjunction with a hydrogen donor agent, such as ammonium formate, potassium formate, or triethylammonium formate, to effect the reduction of the double bond in the 3-carbonyl-4-azepine-5-androstene precursor. The reaction proceeds smoothly at moderate temperatures ranging from 60°C to 101°C over a period of 5 to 24 hours, completely eliminating the need for external hydrogen gas cylinders or high-pressure autoclaves. By shifting to this liquid-phase hydrogen transfer system, manufacturers can achieve comparable or superior conversion rates while operating under atmospheric pressure, which dramatically simplifies the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. This transition not only enhances operational safety but also streamlines the workflow, allowing for a more continuous and efficient production cycle that is highly attractive to supply chain managers seeking reliability.
Mechanistic Insights into Formic Acid-Mediated Transfer Hydrogenation
The core of this technological advancement lies in the mechanistic efficiency of formic acid-mediated transfer hydrogenation. In this system, formic acid serves a dual role as both the solvent and the source of hydride equivalents. Upon heating, the formate species decomposes to release carbon dioxide and active hydrogen species directly onto the substrate surface, facilitating the stereoselective reduction of the C5-C6 double bond to the desired 5-alpha configuration. This mechanism avoids the heterogeneous surface adsorption issues often encountered with solid metal catalysts, where diffusion limitations can lead to incomplete reactions or the formation of unwanted isomers. The presence of the nitrogen atom at the 4-position of the androstane skeleton plays a crucial role in directing the stereochemistry, and the mild acidic environment provided by the formic acid helps maintain the protonation state necessary for high selectivity. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the molar ratio of substrate to formic acid (optimized between 1:5 and 1:40 g/ml), to maximize yield and minimize side reactions.
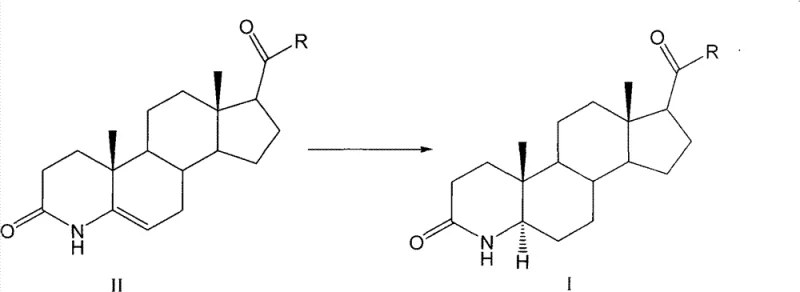
Furthermore, the impurity profile of the resulting intermediate is significantly improved through this method, primarily due to the absence of metal-catalyzed side reactions such as over-reduction or dehalogenation (if halogenated substrates were used). The crude product obtained after the removal of formic acid under reduced pressure is typically of high quality, requiring only a single recrystallization step to achieve pharmaceutical-grade purity. The patent specifies various recrystallization solvents, including ethyl acetate, acetone, and DMF, which effectively remove residual starting materials and minor byproducts. For instance, experimental data indicates that recrystallization from ethyl acetate can yield an alpha-isomer content of up to 99.1wt%, demonstrating the robustness of this purification strategy. This high level of impurity control is critical for R&D directors who must ensure that the final API meets the rigorous standards set by global health authorities, thereby reducing the risk of batch rejection and ensuring a consistent supply of high-purity OLED material or pharmaceutical precursors.
How to Synthesize 3-Carbonyl-4-Aza-5Alpha-Androstane Efficiently
Implementing this synthesis route in a production environment requires precise adherence to the optimized conditions described in the patent embodiments. The process begins with the careful selection of the hydrogen donor salt, with ammonium formate being a preferred choice due to its solubility and reactivity profile. Operators must monitor the reaction temperature closely, maintaining it within the 60°C to 101°C window to ensure complete conversion without thermal degradation of the sensitive steroid backbone. Following the reaction, the removal of excess formic acid is a critical step, typically achieved via vacuum concentration, followed by precipitation in ice water to isolate the crude solid. The detailed standardized synthetic steps for this procedure are provided in the guide below, ensuring reproducibility and compliance with GMP standards.
- Combine the 3-carbonyl-4-azepine-5-androstene precursor with formic acid and a hydrogen donor agent such as ammonium formate in a reaction vessel.
- Heat the mixture to a temperature between 60°C and 101°C and maintain stirring for a duration of 5 to 24 hours to complete the hydrogenation.
- Remove the formic acid under reduced pressure, precipitate the crude product in ice water, filter, dry, and finally recrystallize using ethyl acetate or similar solvents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transfer hydrogenation technology represents a strategic opportunity to optimize the cost structure and resilience of the supply chain for 5-alpha reductase inhibitors. The shift away from precious metal catalysts eliminates a major variable cost component, providing a buffer against market volatility in platinum and palladium prices. Moreover, the simplified operational requirements reduce the capital expenditure needed for plant upgrades, making it easier for contract manufacturing organizations to adopt this technology and offer competitive pricing. The enhanced safety profile of operating at atmospheric pressure also lowers insurance premiums and reduces the risk of production stoppages due to safety incidents, ensuring a more reliable pharmaceutical intermediate supplier partnership.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the substitution of expensive noble metal catalysts with commodity chemicals like formic acid and ammonium formate. In traditional processes, the cost of the catalyst and the subsequent recovery or disposal of heavy metals can account for a significant portion of the COGS (Cost of Goods Sold). By eliminating this requirement, the new method achieves substantial cost savings. Additionally, the energy consumption is optimized as the reaction proceeds at moderate temperatures without the need for energy-intensive compression of hydrogen gas. This qualitative shift in the cost base allows for more aggressive pricing strategies in the generic drug market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents and the complexity of logistics associated with hazardous materials. Formic acid and formate salts are widely available bulk chemicals with stable supply chains, unlike specialized catalysts which may have long lead times or single-source dependencies. The ability to source raw materials locally and reliably reduces the risk of production delays. Furthermore, the simplified process flow shortens the overall manufacturing cycle time, enabling faster turnaround from order to delivery. This agility is crucial for responding to sudden spikes in demand for BPH medications, ensuring that downstream API manufacturers never face a shortage of critical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers distinct advantages that facilitate easier permitting and scale-up. The absence of heavy metal waste streams simplifies wastewater treatment and reduces the burden of hazardous waste disposal, aligning with increasingly stringent global environmental regulations. The process generates carbon dioxide as a benign byproduct, which is easily vented, further minimizing the environmental footprint. This 'greener' profile not only appeals to environmentally conscious stakeholders but also reduces the administrative overhead associated with environmental compliance reporting. Consequently, scaling this process from pilot plant to multi-ton commercial production is straightforward, supporting the long-term growth objectives of pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios and assess the potential impact on their product lifecycle management.
Q: What are the primary cost advantages of this synthesis method compared to traditional catalytic hydrogenation?
A: The primary advantage is the elimination of expensive precious metal catalysts like Platinum (Pt) or Palladium on Carbon (Pd/C). By utilizing inexpensive formic acid and formate salts as hydrogen donors, the raw material costs are drastically reduced, and the complex post-reaction removal of heavy metal residues is no longer required.
Q: How does this process improve operational safety for large-scale manufacturing?
A: Traditional methods often require high-pressure hydrogen gas (e.g., >5 MPa), which poses significant explosion risks and requires specialized high-pressure equipment. This transfer hydrogenation method operates at atmospheric pressure and moderate temperatures (60-101°C), significantly lowering the safety barriers and capital expenditure for reactor infrastructure.
Q: What purity levels can be achieved with the described recrystallization process?
A: According to the patent data, the recrystallization step using solvents like ethyl acetate consistently yields products with high stereochemical purity. For instance, Example 1 demonstrates an alpha-isomer content of 98.5wt%, with other examples reaching up to 99.1wt%, ensuring the intermediate meets stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Carbonyl-4-Aza-5Alpha-Androstane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis methods described in CN101456897B for the global production of urological therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and uninterrupted supply of critical intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of transfer hydrogenation chemistry, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to leveraging our technical expertise to support the development and commercialization of next-generation 5-alpha reductase inhibitors.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full commercial potential of this advanced manufacturing route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your drug development pipeline.
