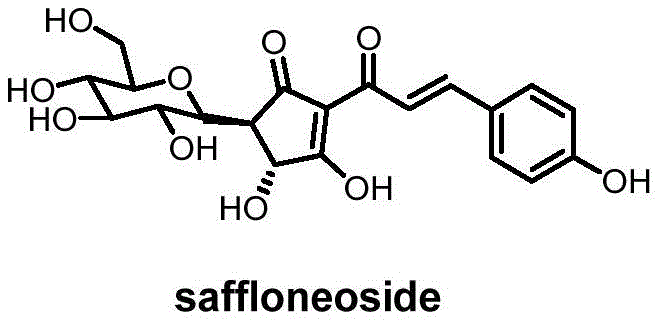
Scalable Total Synthesis of Saffloneoside for Cerebral Ischemia Drug Development
The pharmaceutical industry is constantly seeking robust supply chains for bioactive natural products, particularly those with potent neuroprotective properties. Patent CN111662260B introduces a groundbreaking total synthesis method for saffloneoside, a novel quinone chalcone C-glycoside isolated from safflower. This compound has demonstrated significant efficacy in treating cerebral ischemic injury, a leading cause of disability and mortality globally. Unlike traditional extraction methods which suffer from extremely low natural abundance and seasonal variability, this patented seven-step synthetic route offers a consistent and scalable alternative. By utilizing readily available starting materials like 2,4,6-trihydroxyacetophenone, the process overcomes the supply bottlenecks that have historically hindered the clinical advancement of this promising therapeutic agent. As a reliable saffloneoside supplier, understanding the technical nuances of this pathway is critical for securing long-term API availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of saffloneoside relied exclusively on isolation from traditional Chinese medicine sources, specifically safflower injections. This approach is fraught with significant logistical and economic challenges. The natural content of saffloneoside in the plant material is exceptionally low, necessitating the processing of massive quantities of biomass to obtain gram-scale amounts for research. Furthermore, the purification process from complex botanical matrices is labor-intensive and often results in variable purity profiles due to the presence of structurally similar analogs. Prior to this invention, there were no reported literature precedents for the total synthesis of quinone chalcone C-glycosides, leaving the field dependent on an unsustainable extraction model. This dependency creates severe risks for supply chain continuity, as crop yields are susceptible to environmental factors, pests, and geopolitical trade fluctuations, making cost reduction in pharmaceutical intermediates manufacturing nearly impossible through extraction alone.
The Novel Approach
The synthetic strategy outlined in CN111662260B represents a paradigm shift by constructing the complex molecular architecture from simple, commodity chemicals. The route is designed with scalability in mind, featuring stable intermediates that can be isolated and characterized at each stage, ensuring rigorous quality control. The seven-step sequence efficiently assembles the core cyclopentenone ring and the sensitive C-glycosidic bond without requiring cryogenic conditions or exotic reagents for the majority of transformations. By decoupling production from agricultural cycles, this method guarantees a steady supply of high-purity saffloneoside regardless of external harvest conditions. The ability to control stereochemistry and regioselectivity through specific catalytic steps means that the final product meets stringent regulatory standards for impurity profiles, a feat difficult to achieve with natural extracts.
Mechanistic Insights into Sc(OTf)3-Catalyzed C-Glycosylation and Organocatalysis
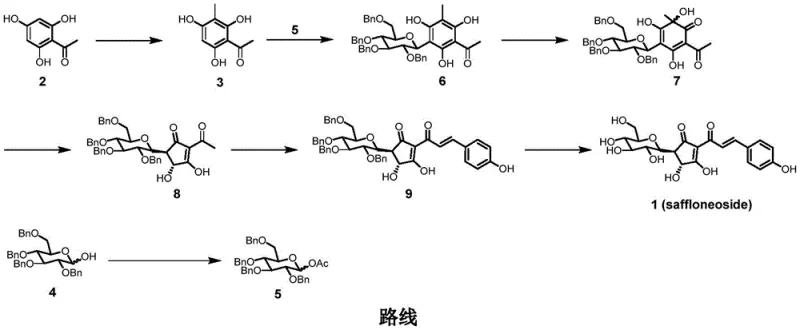
A critical highlight of this synthesis is the construction of the C-glycosidic bond, a notoriously difficult transformation due to the stability of the anomeric center and the potential for O-glycosylation side reactions. The process employs Scandium(III) triflate [Sc(OTf)3] as a Lewis acid catalyst to promote the coupling between the protected glucose donor and the polyhydroxyacetophenone acceptor. This catalytic system operates under mild thermal conditions (45 °C) in 1,2-dichloroethane, achieving an impressive yield of 95% for this key step. The Lewis acid activates the anomeric acetate, facilitating a nucleophilic attack by the electron-rich aromatic ring. This mechanism ensures the formation of the thermodynamically stable C-C bond rather than the kinetically favored O-glycoside, effectively locking the sugar moiety against hydrolytic cleavage in biological systems. Such precise control over the glycosylation topology is essential for maintaining the pharmacological activity of the final drug substance.
Furthermore, the installation of the chalcone side chain utilizes a sophisticated organocatalytic approach involving L-proline and triethylamine. This step involves an aldol condensation between the cyclopentenone intermediate and p-hydroxybenzaldehyde. The use of L-proline as a chiral organocatalyst not only drives the reaction forward under mild conditions but also helps in minimizing racemization and controlling the geometry of the resulting double bond. The subsequent dehydration leads to the conjugated enone system characteristic of chalcones. This metal-free catalytic cycle is particularly advantageous for pharmaceutical manufacturing as it eliminates the need for extensive heavy metal scavenging processes downstream. The combination of Lewis acid catalysis for glycosylation and organocatalysis for carbon-carbon bond formation exemplifies a modern, green chemistry approach to building complex natural product analogs.
How to Synthesize Saffloneoside Efficiently
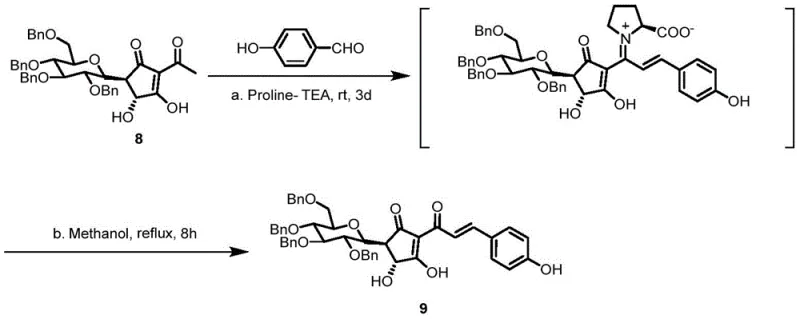
The execution of this synthesis requires careful attention to reaction parameters, particularly during the protection and deprotection phases. The initial methylation and subsequent glycosylation set the foundation for the molecule's stability, while the final oxidative and deprotection steps reveal the active pharmacophore. Operators must maintain strict temperature controls, especially during the final boron tribromide mediated deprotection which occurs at -40 °C to prevent degradation of the sensitive quinone system. The detailed standardized synthesis steps below outline the precise molar ratios, solvent systems, and workup procedures required to replicate the high yields reported in the patent documentation.
- Methylation of 2,4,6-trihydroxyacetophenone using methyl iodide and potassium carbonate to form 3-methyl-2,4,6-trihydroxyacetophenone.
- Preparation of 1-acetyl-2,3,4,6-tetrabenzylglucose via acetylation of tetrabenzylglucose with acetic anhydride.
- Scandium triflate catalyzed C-glycosylation to couple the sugar moiety with the acetophenone derivative.
- Oxidation to quinone structure followed by base-mediated ring contraction to form the cyclopentenone core.
- L-Proline catalyzed aldol condensation with p-hydroxybenzaldehyde to install the chalcone side chain.
- Final global deprotection of benzyl groups using boron tribromide to yield pure saffloneoside.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction to total synthesis offers transformative benefits regarding cost stability and risk mitigation. The reliance on botanical sources introduces uncontrollable variables such as weather patterns and land use changes, which can cause sudden price spikes and material shortages. By adopting this synthetic route, organizations can secure a predictable manufacturing timeline that aligns with commercial production schedules. The use of commodity starting materials like 2,4,6-trihydroxyacetophenone and protected glucose derivatives ensures that raw material costs remain low and sourcing is diversified across multiple global chemical suppliers. This strategic shift significantly reduces the lead time for high-purity saffloneoside, allowing pharmaceutical developers to accelerate their preclinical and clinical programs without the fear of raw material stockouts.
- Cost Reduction in Manufacturing: The synthetic route eliminates the massive overhead associated with agricultural sourcing and low-yield extraction processes. By utilizing high-yielding chemical transformations, such as the 98% yield in the glucose acetylation step and the 95% yield in the glycosylation step, the overall material throughput is maximized. The avoidance of chromatography-heavy purification in favor of crystallization or simple extractions where possible further drives down operational expenses. Additionally, the removal of transition metals in the final steps reduces the cost and complexity of purification, leading to substantial cost savings in the final API production budget.
- Enhanced Supply Chain Reliability: Synthetic manufacturing decouples the supply of saffloneoside from the seasonal harvest cycles of safflower crops. This independence ensures a continuous flow of materials, crucial for maintaining uninterrupted clinical trial supplies and commercial inventory. The stability of the protected intermediates allows for the stocking of key precursors, creating a buffer against unexpected demand surges. This reliability is paramount for partners seeking a reliable saffloneoside supplier who can guarantee delivery commitments over multi-year development timelines without exposure to agricultural volatility.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to scale-up, with most steps proceeding at or near ambient pressure and moderate temperatures. The use of organocatalysts like L-proline aligns with green chemistry principles, reducing the environmental footprint associated with heavy metal waste disposal. The process generates fewer hazardous byproducts compared to large-scale extraction which often requires vast volumes of organic solvents to process tons of plant matter. This efficiency facilitates the commercial scale-up of complex pharmaceutical intermediates while adhering to increasingly strict environmental regulations regarding solvent usage and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of saffloneoside. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy for R&D and procurement decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this molecule into their drug development pipelines.
Q: Why is synthetic saffloneoside preferred over natural extraction?
A: Natural extraction from safflower yields very low quantities, limiting clinical development. The synthetic route described in CN111662260B provides a reliable, scalable supply independent of agricultural variables.
Q: What are the key catalytic technologies used in this synthesis?
A: The process utilizes Scandium(III) triflate for stereoselective C-glycosylation and L-Proline for organocatalytic aldol condensation, ensuring high purity and specific stereochemistry without heavy metal contamination.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the patent highlights that all intermediates are stable and reaction conditions are easily controlled (mostly moderate temperatures), making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Saffloneoside Supplier
The successful translation of patent CN111662260B from laboratory bench to commercial reactor requires a partner with deep expertise in process chemistry and scale-up engineering. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereochemical requirements of saffloneoside are maintained at every ton. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch meets the exacting standards required for neuroprotective drug development. We understand the critical nature of cerebral ischemia therapeutics and are committed to delivering materials that support life-saving research.
We invite pharmaceutical innovators to collaborate with us to optimize this synthesis for your specific needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, identifying further opportunities for process intensification. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of saffloneoside to advance your pipeline from discovery to market.
