Strategic Synthesis of Phenanthrene Alkaloid H4 for Commercial NAFLD Therapeutics
The pharmaceutical industry is currently facing a critical demand for effective treatments against Nonalcoholic Fatty Liver Disease (NAFLD), a condition affecting a significant portion of the global population. In response to this urgent medical need, patent CN113416221B discloses a groundbreaking total synthesis method for Compound H4, a novel phenanthrene alkaloid glycoside with potent hepatoprotective properties. Unlike traditional methods that rely on the cumbersome and low-yield extraction of this bioactive molecule from the plant Sinomenium acutum, this innovative chemical approach utilizes 3-methoxy-4-hydroxyphenethylamine hydrochloride as a readily available starting material. The synthetic strategy encompasses eleven distinct reaction steps, meticulously designed to construct the complex polycyclic framework and install the essential glycosidic moiety with high stereocontrol. By shifting from biological extraction to chemical synthesis, manufacturers can secure a stable supply of this high-value intermediate, bypassing the ecological and logistical constraints of natural sourcing. The structural integrity of the final product is paramount for its biological activity, as illustrated by the precise molecular architecture required for NAFLD intervention.
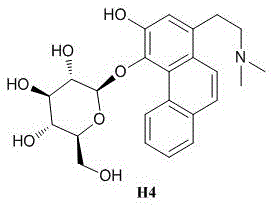
This transition to a fully synthetic route represents a paradigm shift in the manufacturing of complex alkaloid derivatives, offering a reliable phenanthrene alkaloid supplier pathway that is independent of agricultural variables. The ability to produce Compound H4 through a defined chemical sequence ensures that impurity profiles can be strictly monitored and controlled, which is a prerequisite for regulatory approval in major markets such as the US and EU. Furthermore, the synthetic route described in the patent demonstrates robustness across multiple experimental examples, indicating that the process parameters are well-optimized for reproducibility. For R&D directors and procurement managers alike, this development signals a new era of accessibility for this promising therapeutic candidate, potentially accelerating the timeline for clinical trials and eventual commercialization of NAFLD drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of rare natural products like Compound H4 has been plagued by severe inefficiencies inherent to botanical extraction processes. The concentration of this specific phenanthrene alkaloid within the host plant Sinomenium acutum is exceptionally low, necessitating the processing of massive quantities of biomass to isolate mere grams of the active ingredient. This low abundance not only drives up the cost of goods significantly but also introduces substantial variability in the quality of the raw material due to factors such as soil composition, climate conditions, and harvest timing. Moreover, the separation of H4 from the complex matrix of co-extracted plant metabolites requires extensive chromatographic purification, which consumes large volumes of organic solvents and generates considerable chemical waste. From a supply chain perspective, reliance on natural sources creates a fragile dependency on crop yields, making it nearly impossible to guarantee the continuous, large-scale supply required for late-stage clinical development or commercial drug launch. These cumulative disadvantages render natural extraction economically unviable for meeting the growing global demand for NAFLD therapeutics.
The Novel Approach
In stark contrast to the unpredictability of nature, the total synthesis method outlined in patent CN113416221B offers a deterministic and highly controllable alternative for producing Compound H4. By employing a linear 11-step sequence starting from inexpensive commodity chemicals, this approach decouples production from agricultural cycles and environmental fluctuations. The synthetic route is characterized by the use of standard organic transformations, such as amide coupling, cyclization, and palladium-catalyzed cross-coupling, which are well-understood and easily scalable in a GMP environment. Each intermediate in the sequence is stable and can be rigorously characterized, allowing for precise quality control checkpoints throughout the manufacturing process. This level of process control enables cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and maximizing throughput efficiency. Furthermore, the synthetic pathway allows for the strategic optimization of individual steps to improve overall yield, a flexibility that is simply unavailable when working with fixed biological concentrations. Ultimately, this novel approach transforms Compound H4 from a scarce natural curiosity into a commercially viable drug substance.
Mechanistic Insights into Pd-Catalyzed Cyclization and Glycosylation
The cornerstone of this synthetic strategy lies in the construction of the rigid phenanthrene core, which is achieved through a sophisticated intramolecular Heck reaction. In this critical step, a brominated tetrahydroisoquinoline precursor undergoes palladium-catalyzed carbon-carbon bond formation to close the third ring of the phenanthrene system. The reaction utilizes a specialized ligand system, specifically XPhos-PdG2, which facilitates the oxidative addition and reductive elimination cycles necessary for efficient coupling under relatively mild conditions. This mechanistic choice is vital for preserving the sensitive functional groups present on the aromatic rings while ensuring high regioselectivity for the desired ring closure. Following the formation of the carbocyclic skeleton, the synthesis proceeds through a series of functional group manipulations, including Eschweiler-Clark methylation and Hofmann elimination, to establish the correct nitrogen substitution pattern. The precision of these transformations ensures that the final alkaloid scaffold matches the natural product exactly, avoiding the formation of inactive regioisomers that could complicate downstream purification.
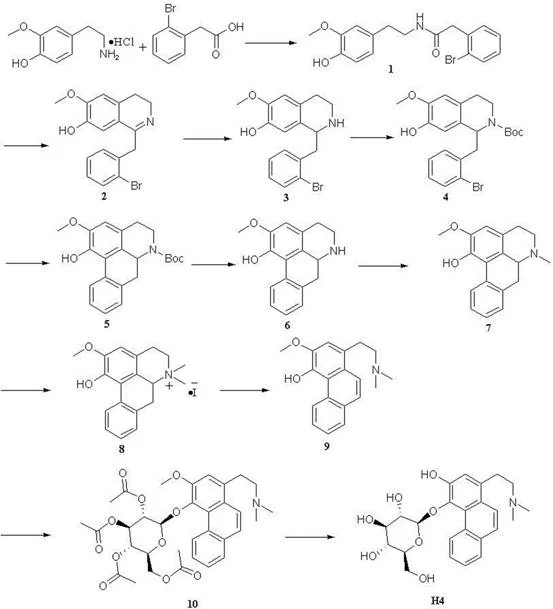
The final stage of the synthesis involves the installation of the glucose moiety via a Koenigs-Knorr glycosylation, a reaction that demands strict stereochemical control to ensure biological efficacy. The process employs a peracetylated glucosyl bromide donor and a phase-transfer catalyst to couple the sugar unit to the phenolic oxygen of the phenanthrene aglycone. This step is followed by a careful deprotection sequence using magnesium iodide and sodium methoxide to remove the acetyl groups without hydrolyzing the sensitive glycosidic bond. The success of this glycosylation is critical, as the sugar component often plays a key role in the solubility and pharmacokinetic profile of the drug. By mastering these complex mechanistic challenges, the synthetic route delivers high-purity Compound H4 with a defined impurity profile that meets stringent pharmaceutical standards. This deep understanding of the reaction mechanisms allows process chemists to troubleshoot potential issues and optimize conditions for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Compound H4 Efficiently
The synthesis of Compound H4 is a multi-stage process that requires careful attention to reaction conditions and purification techniques to achieve optimal yields. The procedure begins with the condensation of the starting amine with o-bromophenylacetic acid, followed by cyclization to form the isoquinoline core. Subsequent steps involve protection strategies, ring-closing metathesis, and functional group interconversions that build up the molecular complexity. The detailed operational parameters, including specific molar ratios, solvent choices, and temperature controls for each of the 11 steps, are essential for replicating the high yields reported in the patent literature. For process development teams, adhering to these standardized protocols is crucial for minimizing side reactions and ensuring the consistency of the final active pharmaceutical ingredient. The complete technical breakdown of these operations provides a clear roadmap for translating laboratory success into industrial reality.
- Construct the isoquinoline core via acid-amine condensation, cyclization with POCl3, and subsequent reduction and Boc protection.
- Form the phenanthrene skeleton using a palladium-catalyzed Heck reaction followed by deprotection and Eschweiler-Clark methylation.
- Complete the synthesis via Hofmann elimination, stereoselective Koenigs-Knorr glycosylation, and final deacetylation to yield Compound H4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route for Compound H4 offers transformative benefits that directly impact the bottom line and operational resilience. The primary advantage is the elimination of supply risk associated with natural product sourcing, where crop failures or geopolitical issues can disrupt availability overnight. By switching to a synthetic supply chain, companies can secure long-term contracts for raw materials that are produced by the global petrochemical industry, ensuring stability and predictability in lead times. Additionally, the synthetic process allows for significant cost reduction in manufacturing by removing the expensive and labor-intensive steps of plant harvesting, drying, and initial extraction. The ability to produce the compound on demand also reduces the need for large inventory buffers, freeing up working capital and storage space. These factors combined create a more agile and cost-effective supply chain model that is better suited for the fast-paced environment of modern drug development.
- Cost Reduction in Manufacturing: The synthetic route leverages commodity chemicals and standard reagents, avoiding the premium pricing of rare botanical extracts. By utilizing efficient catalytic processes like the Heck reaction, the method minimizes the consumption of expensive metals and reduces the number of purification cycles required. This streamlined approach lowers the overall cost of goods sold, making the final therapy more accessible to patients while improving profit margins for the manufacturer. The removal of agricultural processing steps further contributes to substantial cost savings by reducing energy consumption and waste disposal fees associated with biomass handling.
- Enhanced Supply Chain Reliability: Synthetic manufacturing decouples production from seasonal variations and environmental risks, guaranteeing a consistent year-round supply of high-quality intermediates. This reliability is critical for maintaining clinical trial timelines and ensuring that commercial launches are not delayed due to raw material shortages. The use of established chemical suppliers for starting materials adds an additional layer of security, as multiple sources can be qualified to prevent single-point failures. Consequently, pharmaceutical companies can plan their production schedules with greater confidence and accuracy.
- Scalability and Environmental Compliance: The 11-step synthesis is designed with scalability in mind, utilizing reaction conditions that can be safely transferred from kilogram to tonne scale. The process avoids the use of exotic or highly toxic reagents that would complicate regulatory filings and waste management. Furthermore, the improved atom economy of the synthetic route compared to extraction results in a lower environmental footprint, aligning with the sustainability goals of modern pharmaceutical corporations. This compliance with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate social responsibility profile of the drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Compound H4. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology. Understanding these details is essential for making informed decisions about integrating this synthetic intermediate into your drug development pipeline. The information covers aspects of purity, scalability, and regulatory considerations that are top of mind for industry professionals.
Q: What are the primary advantages of synthesizing H4 versus extracting it from natural sources?
A: Synthetic production eliminates the variability associated with plant cultivation and seasonal harvesting, ensuring consistent batch-to-batch purity and supply continuity that natural extraction cannot guarantee.
Q: How does the Heck reaction step impact the overall scalability of the process?
A: The use of robust palladium catalysis in the ring-closing step allows for efficient formation of the complex phenanthrene core under controlled conditions, facilitating easier scale-up compared to multi-step natural isolation.
Q: Is the glycosylation step compatible with large-scale manufacturing requirements?
A: Yes, the Koenigs-Knorr glycosylation utilized in this route employs standard reagents and phase-transfer conditions that are well-established in industrial organic synthesis, supporting reliable commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Compound H4 Supplier
As the demand for effective NAFLD treatments continues to rise, securing a dependable source of high-quality intermediates like Compound H4 is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet the rigorous needs of global pharmaceutical partners. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of Compound H4 meets the highest international standards. We understand the complexities involved in bringing novel alkaloid therapeutics to market and are committed to providing a seamless supply chain solution that supports your clinical and commercial goals.
We invite you to collaborate with us to explore the full potential of this innovative synthetic route for your specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating how our manufacturing capabilities can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in transforming this promising scientific discovery into a life-saving reality for patients worldwide.
