Scalable Synthesis of Next-Generation Lipid Droplet Fluorescent Probes for Biomedical Imaging
Scalable Synthesis of Next-Generation Lipid Droplet Fluorescent Probes for Biomedical Imaging
The rapid advancement of cellular biology and disease diagnostics has created an urgent demand for high-performance imaging reagents capable of distinguishing specific intracellular structures with precision. Patent CN112174946A introduces a groundbreaking lipid droplet fluorescent probe that addresses critical limitations found in traditional imaging agents. This novel compound, characterized by a donor-acceptor (D-A) molecular architecture, offers exceptional specificity for neutral lipids within cells, making it an invaluable tool for researching metabolic disorders such as atherosclerosis and fatty liver disease. For procurement leaders and R&D directors in the biomedical sector, understanding the synthetic accessibility and performance metrics of this molecule is paramount. The technology leverages a robust palladium-catalyzed cross-coupling strategy, ensuring that the transition from laboratory discovery to commercial supply is both technically feasible and economically viable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the visualization of lipid droplets in biological systems has relied heavily on commercial dyes such as BODIPY 493/503 and Nile Red. While these reagents have served the scientific community for decades, they possess inherent drawbacks that compromise data integrity in high-resolution imaging applications. A significant issue is their tendency to exhibit substantial background fluorescence in aqueous environments, which obscures the specific signal originating from the lipid droplets themselves. This lack of environmental sensitivity results in a poor signal-to-noise ratio, making it difficult to accurately quantify lipid content or observe morphological changes in live cells. Furthermore, many existing probes suffer from photobleaching and limited stability, restricting their utility in long-term time-lapse studies. From a supply chain perspective, the complex multi-step synthesis required for some of these legacy fluorophores often leads to high costs and inconsistent batch-to-batch quality, creating bottlenecks for large-scale research projects.
The Novel Approach
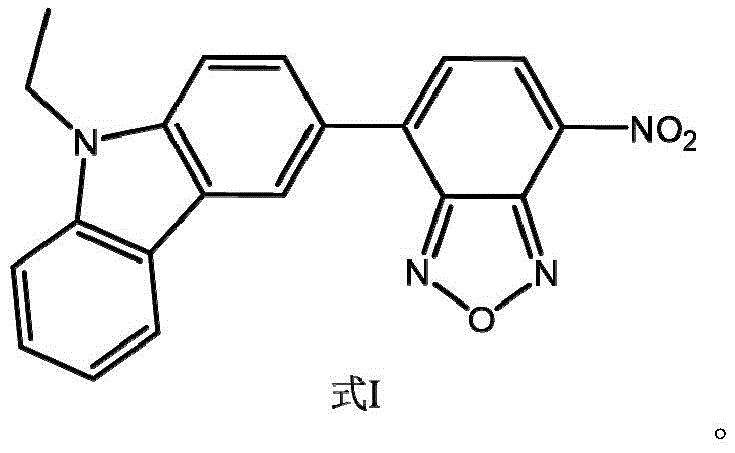
The synthesis method disclosed in the patent represents a paradigm shift by utilizing a push-pull electronic system designed specifically for environment-sensitive fluorescence. By coupling an electron-donating N-ethyl carbazole group with an electron-withdrawing 7-nitrobenzo-2-oxa-1,3-diazole (NBD) moiety, the resulting molecule remains virtually non-fluorescent in polar solvents like water. However, upon entering the hydrophobic environment of a lipid droplet, the probe undergoes a conformational restriction that triggers intense fluorescence emission. This "turn-on" mechanism effectively eliminates background noise, providing crystal-clear imaging of intracellular lipid stores. The synthetic route is streamlined, relying on a direct coupling of two commercially accessible precursors. This simplicity not only accelerates the R&D timeline but also drastically reduces the complexity of purification, offering a clear pathway for cost reduction in biomedical reagent manufacturing without sacrificing the high purity required for sensitive biological assays.
Mechanistic Insights into Pd-Catalyzed Suzuki-Miyaura Coupling
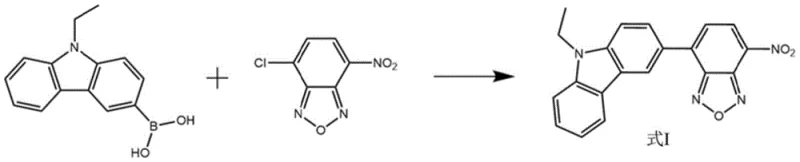
The core of this synthesis lies in the palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, a cornerstone of modern organic synthesis known for its tolerance of functional groups and mild reaction conditions. In this specific transformation, the oxidative addition of the palladium(0) catalyst to the carbon-chlorine bond of the 4-chloro-7-nitrobenzoxadiazole initiates the catalytic cycle. This step is followed by transmetallation with the 9-ethyl-3-carbazole boronic acid, facilitated by the presence of a base such as sodium carbonate or cesium carbonate. The base activates the boronic acid species, forming a reactive boronate complex that readily transfers the carbazole group to the palladium center. Finally, reductive elimination releases the desired biaryl product and regenerates the active palladium(0) catalyst, allowing the cycle to continue. The choice of ligand and palladium source is critical; the patent highlights the efficacy of tetrakis(triphenylphosphine)palladium, which provides a stable yet reactive species capable of driving the reaction to high conversion rates even with sterically hindered substrates.
Controlling impurities in this reaction is essential for ensuring the probe's performance in biological systems, where trace metal contaminants or side products could induce cytotoxicity. The reaction conditions specified, typically involving temperatures between 80°C and 120°C in mixed solvent systems like toluene and ethanol, are optimized to minimize homocoupling of the boronic acid and hydrolysis of the sensitive NBD chloride. The use of inert gas protection, such as nitrogen or argon, prevents the oxidation of the phosphine ligands and the palladium catalyst, thereby maintaining catalytic efficiency throughout the extended reaction times of 10 to 50 hours. Post-reaction workup involves simple filtration and solvent removal, followed by flash column chromatography. This purification strategy effectively removes residual palladium species and unreacted starting materials, yielding a product with the stringent purity specifications necessary for reliable fluorescent imaging and consistent experimental results.
How to Synthesize 9-Ethyl-3-(7-nitrobenzo[c][1,2,5]oxadiazol-4-yl)-9H-carbazole Efficiently
Executing this synthesis requires careful attention to stoichiometry and atmospheric control to maximize yield and reproducibility. The process begins by charging the reactor with the boronic acid and the chloro-NBD derivative under an inert atmosphere to prevent moisture interference. A suitable palladium catalyst and base are added, followed by a solvent system that balances solubility with reaction kinetics, such as a mixture of toluene, ethanol, and water. The reaction mixture is then heated to reflux, typically around 110°C, and maintained for approximately 24 hours to ensure complete consumption of the limiting reagent. Upon completion, the mixture is cooled, filtered to remove insoluble salts and catalyst residues, and concentrated. The crude solid is then subjected to chromatographic purification to isolate the pure reddish-brown fluorescent probe. For detailed operational parameters and safety considerations, please refer to the standardized protocol below.
- Charge 9-ethyl-3-carbazole boric acid and 4-chloro-7-nitrobenzo-2-oxa-1,3-diazole into a reactor under inert gas protection.
- Add palladium catalyst, base additive, and organic solvent mixture (e.g., toluene/ethanol/water) and heat to 80-120°C.
- After reaction completion, cool, filter, remove solvent, and purify the crude product via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding cost stability and sourcing reliability. The primary advantage lies in the accessibility of the starting materials; both 9-ethyl-3-carbazole boric acid and 4-chloro-7-nitrobenzo-2-oxa-1,3-diazole are established intermediates available from multiple global suppliers, mitigating the risk of single-source dependency. This competition among raw material vendors naturally drives down input costs and ensures a steady flow of inventory. Furthermore, the reaction utilizes common, low-cost solvents like toluene and ethanol rather than exotic or highly regulated chemicals, simplifying waste management and reducing the overall environmental compliance burden associated with production. The robustness of the Suzuki coupling means that the process is less prone to failure due to minor variations in conditions, leading to higher batch success rates and reduced material waste.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metals beyond standard palladium catalysts, which can often be recovered or used in low loadings. By avoiding complex multi-step sequences and protecting group strategies common in other fluorophore syntheses, the overall number of unit operations is significantly reduced. This streamlining translates directly into lower labor costs, reduced energy consumption for heating and cooling, and minimized solvent usage. Additionally, the high selectivity of the reaction reduces the formation of difficult-to-separate byproducts, lowering the cost and time associated with downstream purification processes like chromatography.
- Enhanced Supply Chain Reliability: The flexibility in catalyst selection allows manufacturers to switch between different palladium sources (e.g., Pd(PPh3)4 vs. Pd(OAc)2) based on market availability and price fluctuations without compromising the reaction outcome. This adaptability is crucial for maintaining continuous production schedules in the face of global supply chain disruptions. Moreover, the reaction tolerates a wide range of temperatures and solvent ratios, providing process engineers with the leeway to optimize for throughput or equipment constraints. Such operational flexibility ensures that delivery timelines for high-purity fluorescent probes remain consistent, supporting the rigorous scheduling needs of pharmaceutical and academic research clients.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is straightforward due to the homogeneous nature of the reaction and the absence of hazardous reagents like strong acids or pyrophoric organolithiums. The use of aqueous bases and alcohol co-solvents aligns well with green chemistry principles, facilitating easier wastewater treatment and solvent recovery. The simplified workup procedure, involving filtration and concentration, is easily adaptable to large-scale reactors, enabling the commercial scale-up of complex fluorescent intermediates without requiring specialized high-pressure or cryogenic equipment. This ease of scale-up ensures that supply can rapidly expand to meet surging demand in the biomedical imaging sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this lipid droplet probe. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this reagent into their supply chains or research workflows. The answers reflect the balance between high-performance imaging requirements and practical manufacturing constraints.
Q: What is the primary advantage of this probe over BODIPY 493/503?
A: Unlike BODIPY which often suffers from background fluorescence in aqueous environments, this carbazole-based probe exhibits negligible fluorescence in water but strong emission in lipid environments, significantly improving the signal-to-noise ratio for specific lipid droplet imaging.
Q: What catalysts are suitable for this synthesis?
A: The patent specifies a wide range of palladium catalysts including tetrakis(triphenylphosphine)palladium, palladium acetate, and various phosphine-ligated palladium complexes, allowing flexibility based on cost and availability.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes common organic solvents like toluene and ethanol, operates at moderate temperatures (e.g., 110°C), and uses readily available starting materials, making it highly amenable to kilogram-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lipid Droplet Fluorescent Probe Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality imaging reagents play in advancing biomedical research and drug discovery. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for pilot studies or full-scale clinical trial material. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our fluorescent probes delivers the consistent performance and signal-to-noise ratio demanded by top-tier research institutions. Our commitment to quality assurance means that you can rely on our materials to produce reproducible and publishable data.
We invite you to collaborate with us to optimize your supply chain for advanced imaging reagents. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume needs and purity requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of this next-generation lipid droplet probe, empowering your research with superior imaging tools.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
